Topic 2 - Covalent bonding
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
what does covalent bonding occur between?
two non-metals
what is covalent bonding?
the electrostatic attraction between nuclei of two atoms and the bonding electrons of their outer shells
are electrons transferred or shared in covalent bonding?
shared
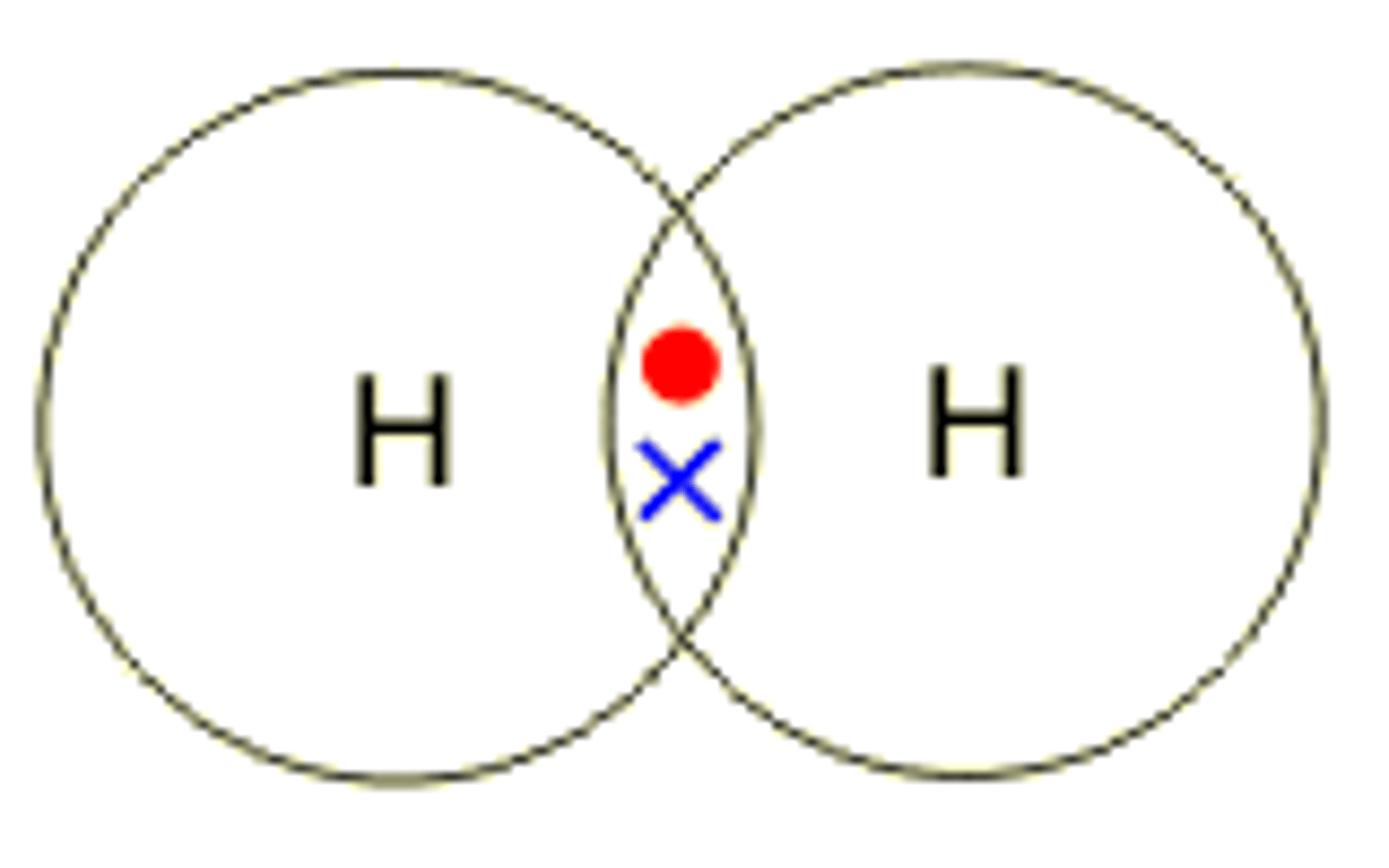
draw the dot and cross diagram for hydrogen
draw the dot and cross diagram for chlorine
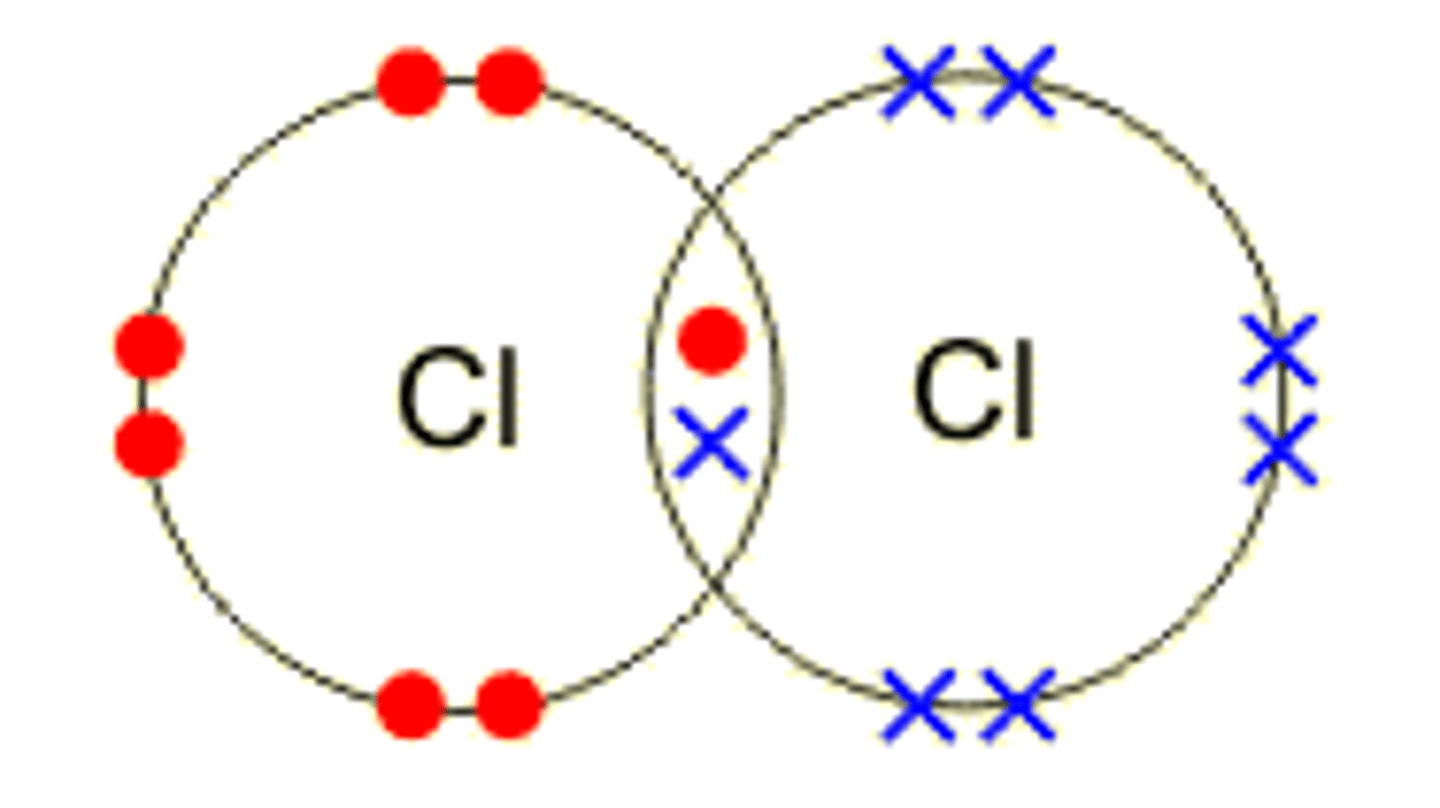
describe the bonding in chlroine
chlorine atoms have 7 electrons in their outer shells, a shared pair of electrons forms a single covalent bond and each chlorine atom has 8 electrons in its outer shell which makes it stable
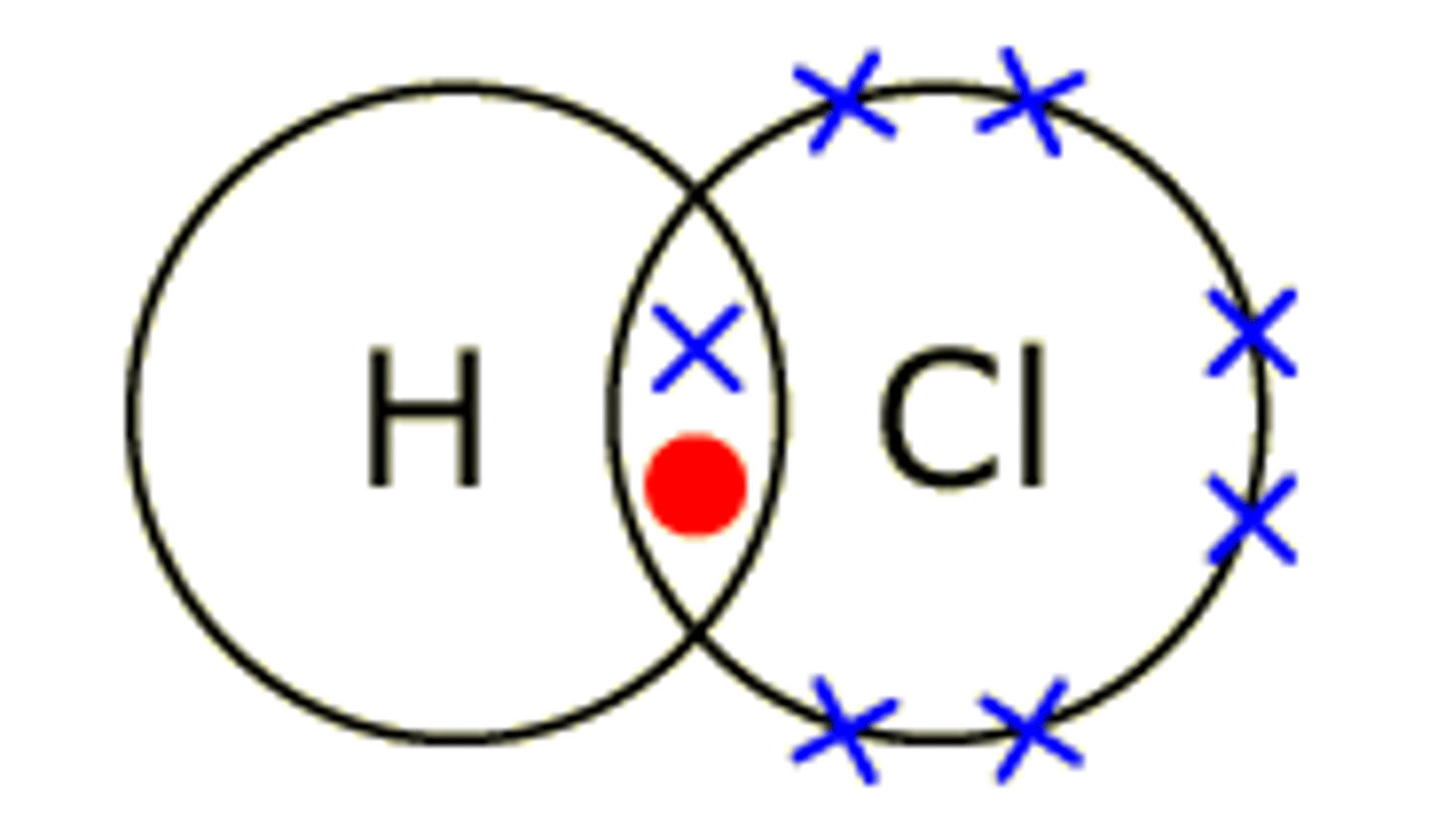
draw the dot and cross diagram for HCl
draw the dot and cross diagram for ammonia
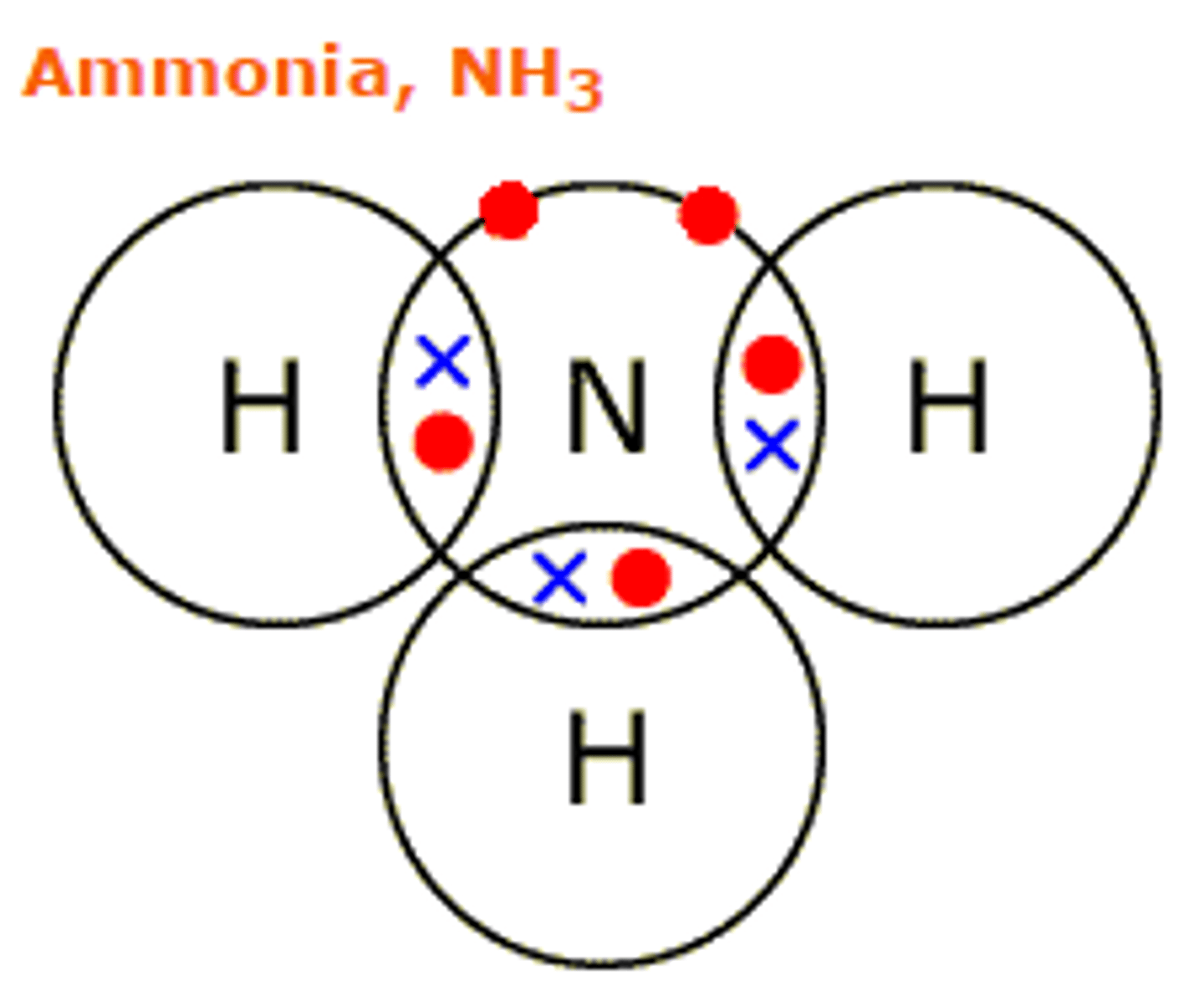
describe the bonding in ammonia
each hydrogen atom can donate an electron to form a covalent bond. nitrogen is in group 5 so it requires 3 more electrons to complete its outer shell, nitrogen has one lone pair and forms 3 single bonds with each of the hydrogen atoms
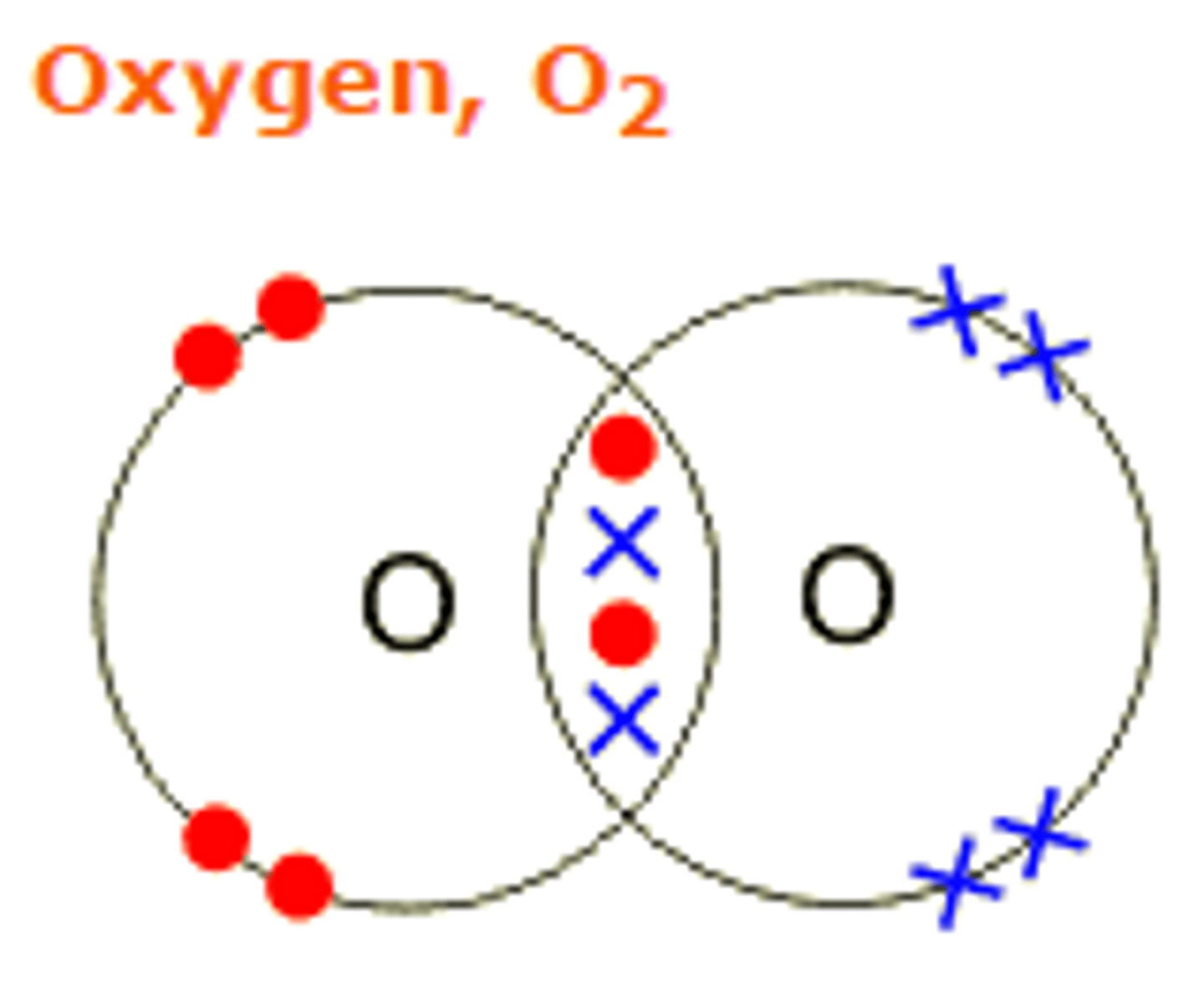
what type of bonds does O2 have?
double covalent bonds
draw the dot and cross diagram for oxygen
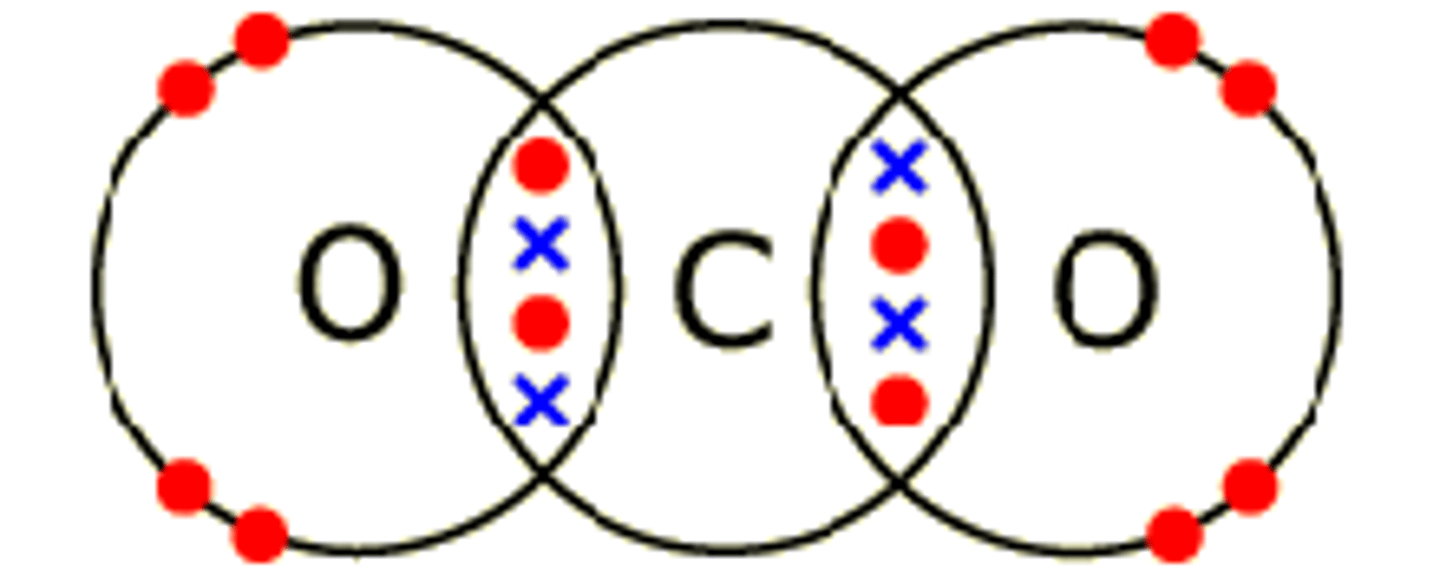
does carbon dioxide have single or double covalent bonds?
double
draw the dot and cross diagram for CO2
draw the dot and cross diagram for nitrogen
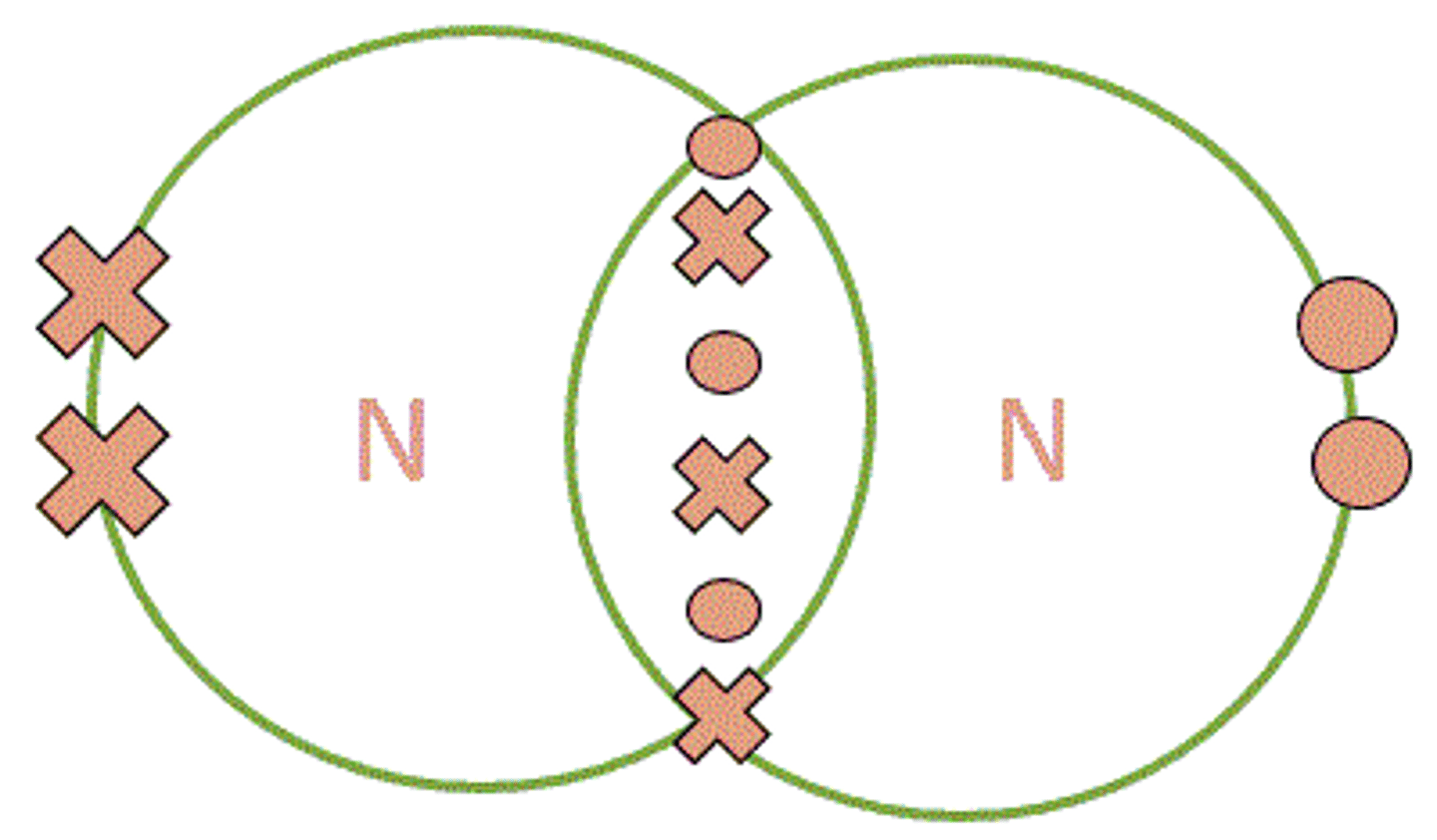
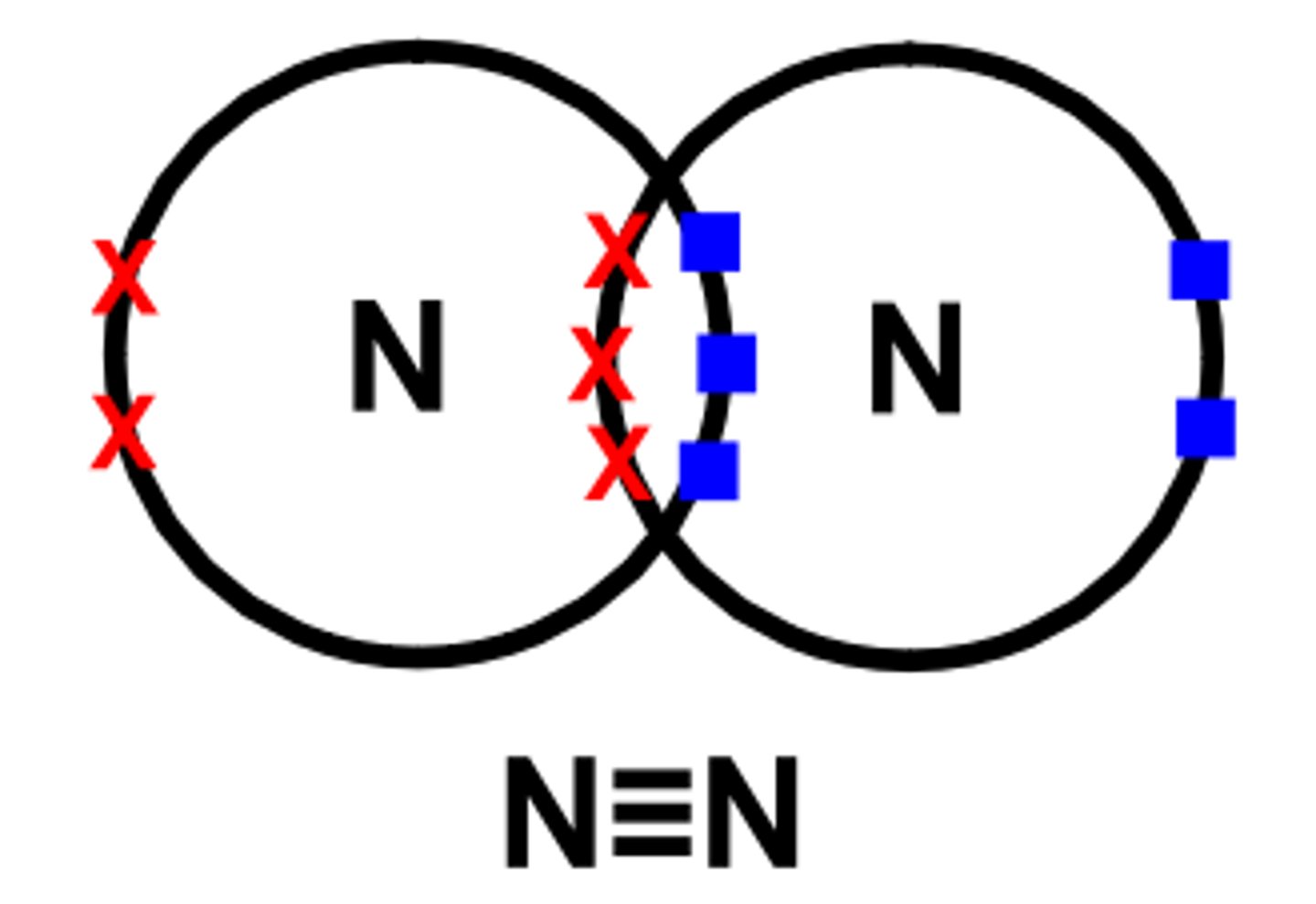
describe the bonding in nitrogen
each nitrogen atom has 5 electrons in its outer shell. each nitrogen atom shares 3 electrons to form a triple bond
does nitrogen N2 have single, double or triple covalent bonds?
triple
draw the dot and cross diagram for sulfur dioxide
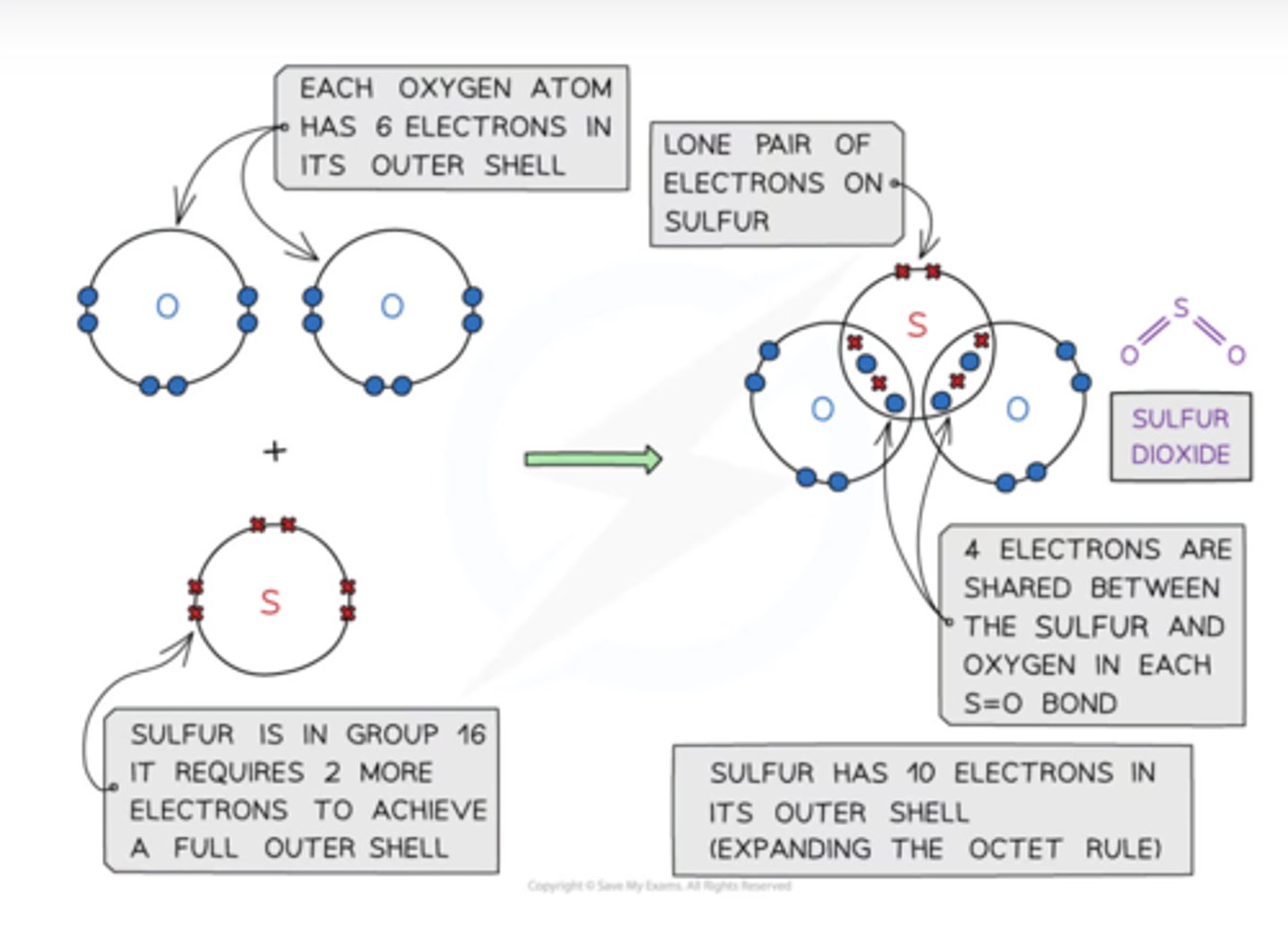
describe the structure of sulfur dioxide
sulfur has 10 electrons in its outer shell (expanding the octet rule) 4 electrons are shared between the sulfur and oxygen in each S=O bond. there is also lone pair of electrons on sulfur
describe the structure of phosphorus pentachloride
2 electrons are shared between the phosphorus and chlorine atoms in the P-Cl bond. Phosphorus has 10 electrons in its outer shell (expanding the octet rule)
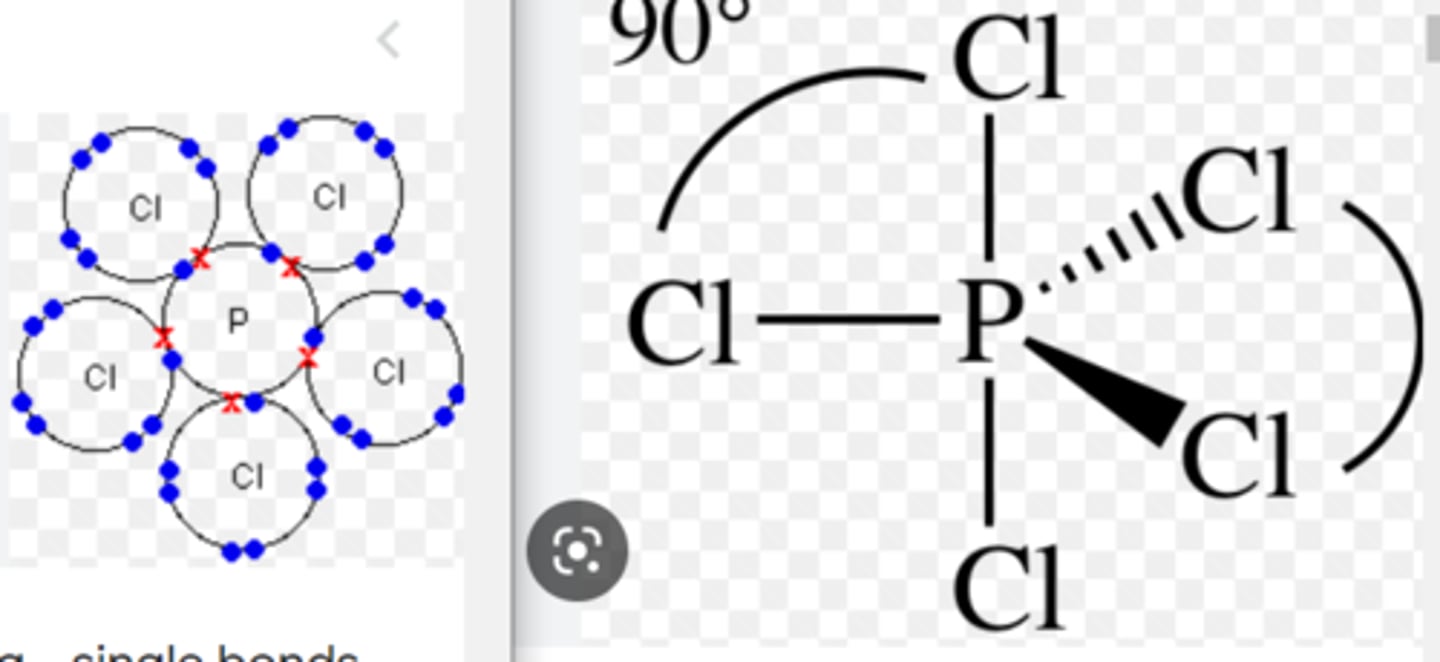
double, single or triple bonds in phosphorus pentachloride
single
describe the structure of sulfur hexafluoride
2 electrons are shared in each S-F bond. Sulfur has 12 electron in its outer shell (expanding the octet rule)

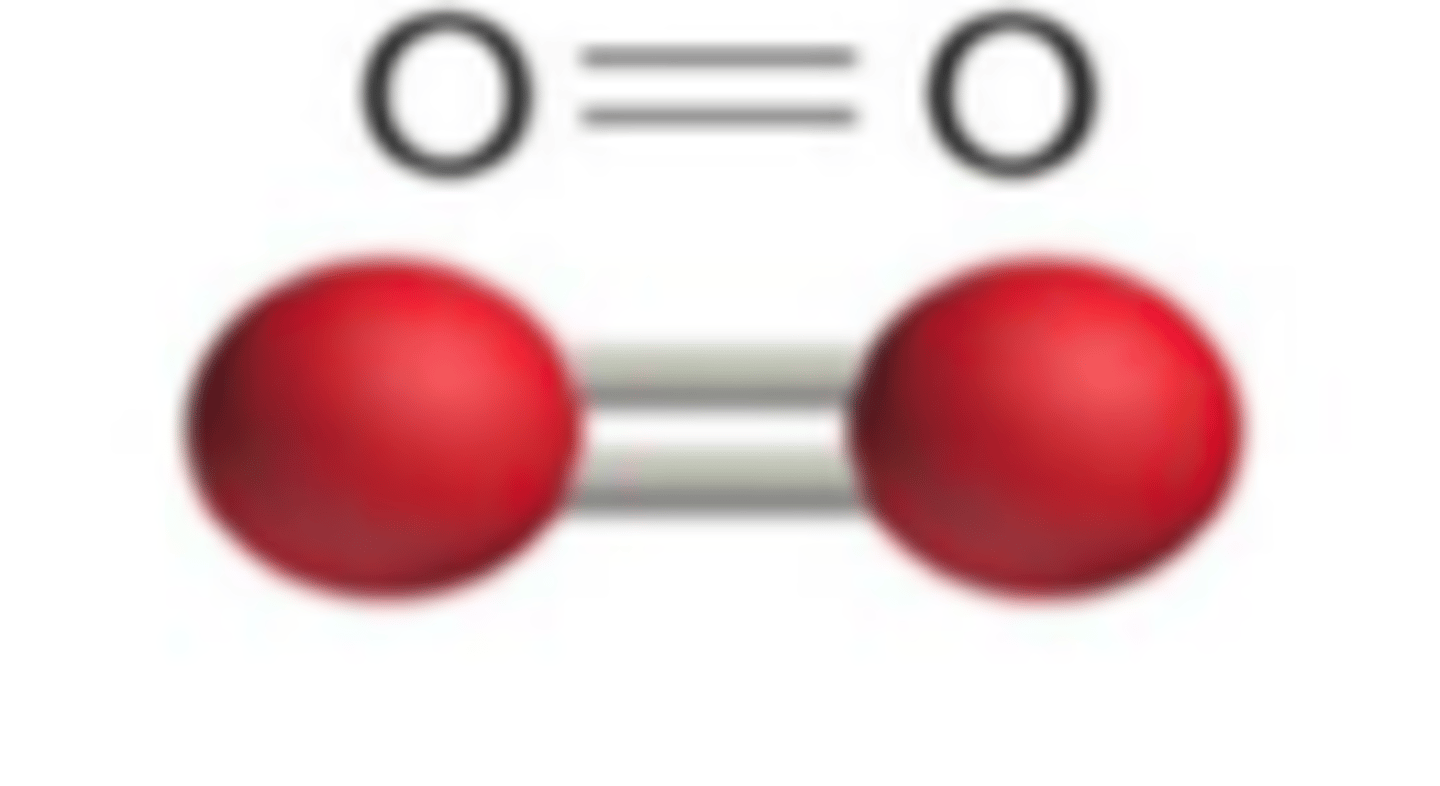
double, single, or triple bonds in O2 ?
double
double, single, or triple bonds in N2?
triple
double, single, or triple bonds in CO2?
double
describe the bonding of an ammonium ion
3 hydrogens form single covalent bonds with the hydrogens and then the last hydrogen is formed with a dative covalent bond
at a high temperature, what does aluminium chloride exist as?
a monomer AlCl3
what happens to aluminium chloride at lower temperatures?
two molecules of AlCl3 join together to form a dimer Al2Cl6
how does Al2Cl6 exist ?
two molecules of Al2Cl6 combine because lone pairs of electrons on two of the chlorine atoms forms two dative bonds with the aluminium atoms
what is bond energy?
the energy required to break one mole of a particular covalent bond in the gaseous states
what does a large bond energy show about the strength of a covalent bond?
stronger covalent bond
what is bond length?
the internuclear distance of two covalently bonded atoms
what are the shortest and strongest type of covalent bonds?
triple bonds