CMMB 343 Lab Practical Exam 2 (Labs 5-8)
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
LAB 5
Oxygen Tolerance & Aerotolerance (4)
Molecular oxygen (O2) is important in aerobic respiration because it acts as the final electron accepter in the ETC
Reducing O2 to H2O can generate toxic oxygen by-products (O2-, H2O2, OH·)
If not removed, organic cellular components are oxidized and the cell dies
Microorganisms differ in their ability to detoxify reactive oxygen species, so they differ in the level of oxygen used for metabolism; some cannot tolerate oxygen at all
Testing Oxygen Tolerance: Deep Shake Culture (3)
Molten tryptose agar is inoculated and allowed to solidify
As oxygen diffuses from the top, an oxygen gradient forms
The area where bacteria grow indicates oxygen preference
Testing Oxygen Tolerance: Thioglycolate Medium (3)
Thioglycolate removes oxygen from the medium, creating a gradient (top of the tube = oxygen presence; bottom = anaerobic)
A pink layer at the top indicates the oxygenated region
Growth patterns reveal oxygen tolerance
Oxygen Tolerance: Interpreting Results - 4 Things to Look For
Location of Growth
Amount of Turbidity
Presence or Absence of Gas
Relationship of Growth to Oxygen Gradient
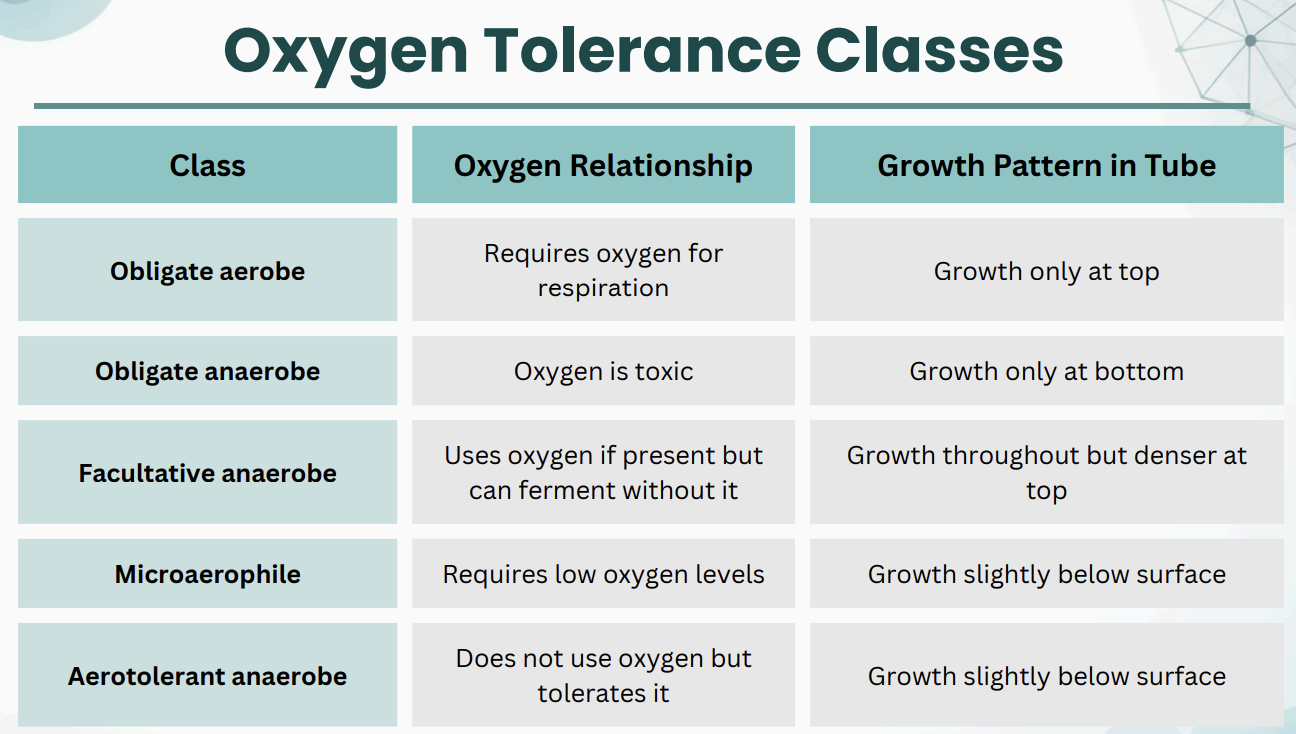
Oxygen Tolerance Classes
*Reference Midterm 1 Flashcards?
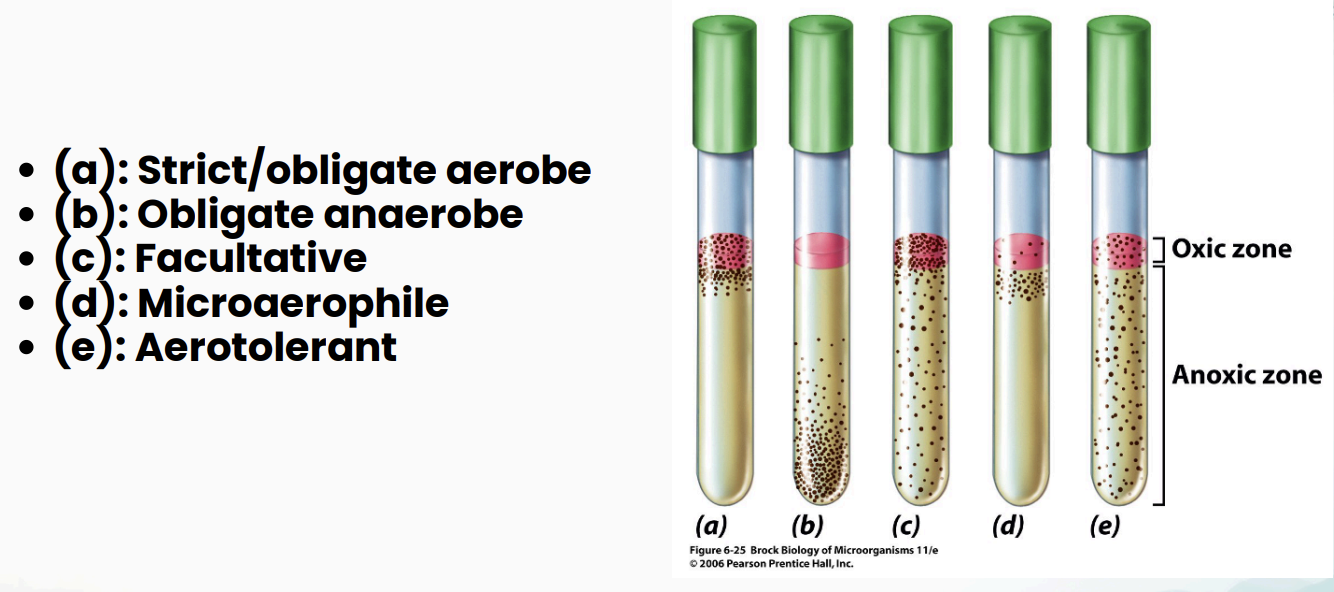
Interpreting Deep Shake Culture Results: What to Record (3)
Where is the turbidity?
Is growth only at the top, throughout the tube, or deeper in the agar?
Is there cracking in the agar?
If gas was produced in the anaerobic portion of the tube, cracking of the agar may be observed
Interpreting Thioglycolate Medium Results: Pink Layer (2)
The pink layer at the top marks the oxygenated region (Resazurin redox indicator)
This layer is pink when oxidized and colourless when reduced
Interpreting Thioglycolate Medium Results: Growth Relative to Pink Layer (2)
If growth is below the pink layer, the organism is growing away from the oxygen-rich region
If the pink layer disappears, oxygen may have been consumed and the indicator was reduced
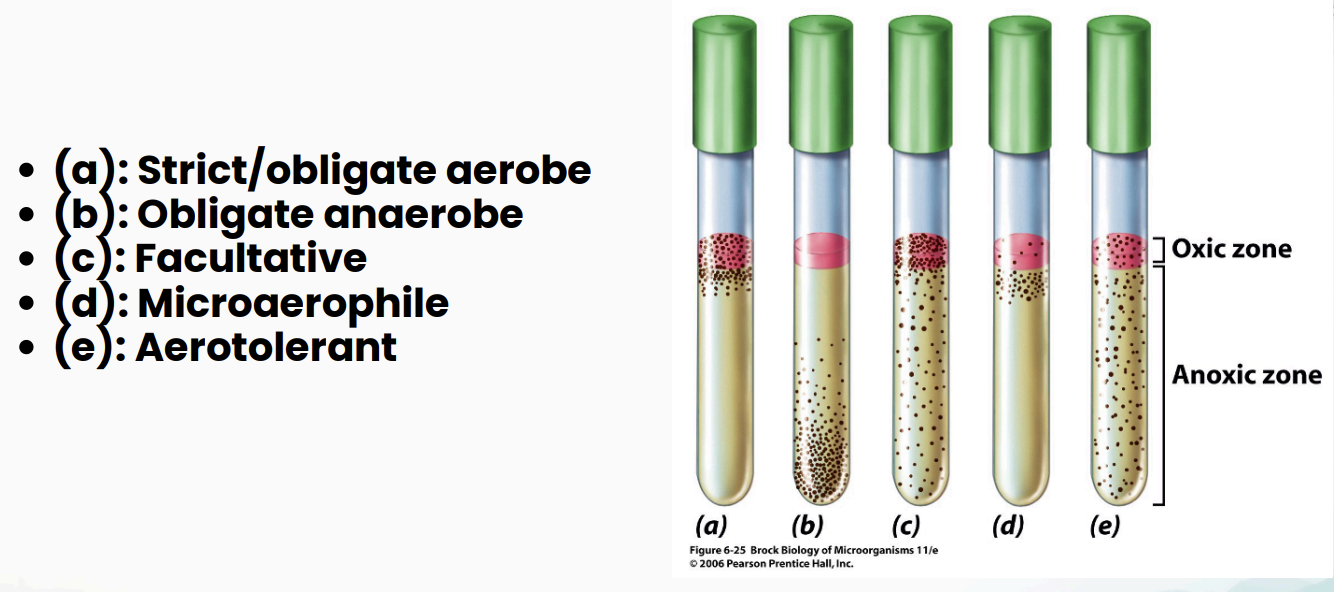
Interpreting Thioglycolate Medium Results: What to Record (3)
Location of growth
Relationship of growth relative to the pink layer
Turbidity pattern
Metabolic Diversity: Testing Glucose (3)
Glucose is a central substrate in microbial metabolism; many microbes use glucose to generate energy, but not all use it in the same way
Depending on the organism and oxygen availability, glucose may be used in: aerobic respiration, anaerobic respiration, fermentation
The key idea is that different organisms can process the same substrate using different metabolic pathways
Testing Glucose: Hugh & Leifson (H&L) Glucose Medium (3)
Two H&L glucose tubes are stab inoculated with the same organism: one tube remains open to air, and the other is overlaid with mineral oil
Mineral oil limits oxygen exposure to create more anaerobic conditions
The purpose of the test is to compare glucose metabolism in the presence vs. absence of oxygen
Hugh & Leifson (H&L) Glucose Medium: What This Test Asks (3)
Does The Organism Use Glucose:
Only when oxygen is available
In both aerobic and anaerobic conditions
Or not at all
*Distinguishes oxidative, fermentative, and non-utilizing organisms
Hugh & Leifson (H&L) Glucose Medium: Interpreting The Indicator (3)
H&L glucose medium contains bromothymol blue, a pH indicator
If the organism metabolizes glucose, acidic end products are released into the medium (lowers pH)
If the organism does not utilize glucose, it may instead use peptones in the medium which produces alkaline end products (raises pH)
Hugh & Leifson (H&L) Glucose Medium: Interpreting The Indicator - Colour Change (3)
Green → Yellow: Positive for carbohydrate utilization
Green → Blue: Negative for glucose utilization
Green: Neutral/No Major Change
Four Common H&L Result Patterns
Both Tubes Green
Negative/No glucose utilization
Only Open Tube Yellow
Oxidative metabolism/aerobic glucose use
Positive for respiration
Both Tubes Yellow
Fermentation/Facultative Glucose Use
Positive for fermentation
Blue Colour Develops
Peptone utilization/alkaline end products
Positive for peptone utilization
*Blue colour developing is still considered a negative result

Fermentation
Fermentation is a way for heterotrophic microorganisms to obtain energy from carbohydrates without using an ETC
4 Key Features of Fermentation
Occurs in the absence of oxygen
No energy-producing electron transport
Less energy-efficient than respiration
Metabolic intermediates or fermentation end products are produced
Fermentation: Two Stages
Oxidation of Glucose
Glucose is split and oxidized; pyruvate is produced; reducing power is generated
Reduction of Pyruvate-Derived Compounds
Pyruvate or its derivatives are reduced; regenerates oxidized cofactors needed to continue glycolysis; characteristic products are formed
Distinguishing Bacteria Via Fermentation
Different bacteria produce different end products, and these products help distinguish species
Homolactic Fermentation (3)
In homolactic fermentation, carbohydrate (glucose, lactose, etc.,) is first converted to pyruvate via the EMP pathway
Pyruvate is then reduced almost entirely to lactic acid
Lactic acid production changes the chemistry of the medium and can be used to detect fermentation activity
Homolactic Fermentation: Examples (2)
Streptococcus
Lactobacillus
Testing Homolactic Fermentation: Litmus Milk (3)
Litmus milk is used to detect lactose fermentation and protein metabolism produced by microorganisms growing in milk-based medium
Litmus milk is a pH indicator; several different metabolic reactions can occur in litmus milk
The medium contains: skim milk (source of lactose and casein protein) and litmus dye (acts as pH indicator and redox indicator)
The initial colour of the litmus milk medium is ___
Light purple (lilac)
Litmus Milk: Interpreting Results - 5 Potential Metabolic Reactions
Acid Production
Litmus turns pink
Indicates lactose fermentation (acidic end products are produced)
Alkaline Reaction
Litmus turns purple or blue
Indicates casein digestion (ammonia is produced)
Reduction
Litmus turns white
Occurs when litmus dye is chemically reduced during fermentation
Coagulation
Medium solidifies
Acid causes casein precipitation
Gas production may create cracks or fissures
Peptonization
Casein is digested
Medium becomes translucent or watery

Anaerobic Respiration
Respiration does not always require oxygen; some microorganisms can use alternative terminal electron acceptors (e.g. NO3-, SO42-, CO2, Fe3+, fumarate)
Anaerobic (Nitrate) Respiration (2)
Two Different Ways Nitrate Can Be Reduced:
Assimilative Nitrate Reduction
Dissimilative Nitrate Reduction
Anaerobic (Nitrate) Respiration: Assimilative Nitrate Reduction - Biosynthesis (3)
Nitrate is reduced for use as a nutrient source (providing nitrogen for amino acids and proteins - typically ammonia)
Can occur under aerobic or anaerobic conditions
Assimilative nitrate reductases are soluble proteins that are ammonia repressed
Anaerobic (Nitrate) Respiration: Dissimilative Nitrate Reduction - Energy Metabolism (3)
Nitrate is used as a terminal electron acceptor in energy metabolism (anaerobic respiration)
Restricted to bacteria
Dissimilative nitrate reductases are membrane-bound proteins that are repressed by O2 and synthesized under anaerobic conditions only
Nitrate Reduction (2)
Ability to reduce nitrate to nitrite in the absence of oxygen (anaerobic respiration)
This process is present in many bacteria
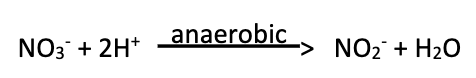
Denitrification (3)
Ability to further reduce nitrate beyond nitrite to gaseous products
This process is limited to a smaller number of genera
Denitrification is considered a detrimental process as gases can escape into the atmosphere, removing biologically available nitrogen from the environment (N2 less readily available)
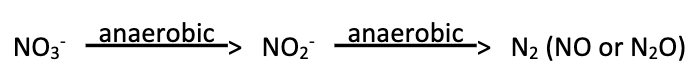
Testing Nitrate Respiration: Nitrate Reduction Broth Test - Interpreting Results (3)
Turbidity in the tube indicates organism growth, but turbidity by itself does not prove nitrate reduction
A positive result is the presence of a bubble in the Durham tube (indicates formation of nitrogenous gas - denitrification)
A negative result is no bubble in Durham vial; no evidence of gas production; organism does not fully reduce nitrate (unknown result)

Effect of Chemical Agents on Bacterial Growth: 3 Mechanisms
Cidal
Concerned with the killing of microorganisms (bactericidal, fungicidal)
Static
Inhibits growth (bacteriostatic)
Lytic
Causes cell lysis
Antiseptics (2)
Antimicrobial agents safe for living tissue (used on skin or other tissues)
May be cidal, static, or lytic
Disinfectants (2)
More potent antimicrobial agents used on non-living surfaces (unsafe for living tissues due to toxicity)
Usually cidal or lytic
Evaluating Claims of Commercially Prepared Antibacterial Agents: Experiment (3)
Many commercial preparations claim to be antibacterial; experiment will evaluate these claims and compare effectiveness of competitive products
The effectiveness of products is tested against both Gram-positive bacterium (Staphylococcus), and Gram-negative bacterium (Enterobacteriaceae)
Each test solution is applied to a paper disc on a bacterial lawn; after incubation the inhibition of growth around the disc can be compared
Evaluating Claims of Commercially Prepared Antibacterial Agents: Products Tested (4)
Tea Tree Oil
BandAid
Pine Sol
Mr. Clean
Effect of Chemical Agents on Bacterial Growth: Kirby-Bauer Disc Diffusion (2)
Chemotherapeutic agents are antimicrobial compounds that can be administered internally (important in both human and veterinary medicine)
The Kirby-Bauer test is used to evaluate how sensitive a bacterium is to different antibiotics (standardized method to compare antibiotic susceptibility)
Kirby-Bauer Disc Diffusion: Experiment (4)
Each antibiotic disc contains a specific amount of antibiotic and is placed on a bacterial lawn
The antibiotic diffuses outward into the agar to create a concentration gradient; antibiotic concentration is highest near the disc and decreases with distance
If the bacterium is inhibited, a zone of inhibition forms around the disc
The zone diameter can be compared to a standard chart to classify the organism as: susceptible, intermediate, resistant
Kirby-Bauer Disc Diffusion: Antibiotics Tested (3)
Erythromycin
Vancomycin
Penicillin
Effect of Chemical Agents on Bacterial Growth: Phenol (2)
Phenol was one of the first disinfectants widely used in Western medicine
Because phenol became an early standard disinfectant, other chemical agents are compared against it
Phenol Coefficient (2)
The phenol coefficient is the ratio of the test disinfectant’s effectiveness to phenol’s effectiveness (reciprocal effective dilution of test disinfectant/reciprocal effective dilution of phenol)
Both agents are tested against the same organism in the same conditions

Phenol Coefficient: Interpretation (3)
Coefficient > 1: Test disinfectant is more effective than phenol
Coefficient < 1: Test disinfectant is less effective than phenol
Coefficient = 1: Equally effective
Phenol Coefficient: Effective Dilution (2)
An effective dilution is the dilution of an agent that completely inhibits growth after 10 minutes, but does not completely inhibit growth after 5 minutes
This gives a standard way to compare disinfectants fairly
Phenol Coefficient: Effective Dilution - Why 5 & 10 Minutes (2)
Because effectiveness depends on both concentration and exposure time
A disinfectant may not kill quickly at 5 minutes, but becomes effective by 10 minutes
LAB 6