Ch 15: Benzene and Aromatic Compounds
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What is the simplest aromatic hydrocarbon?
Benzene (C6H6)
What is the degree of unsaturation in benzene?
Four degrees of unsaturation
What affect does benzene's structure have on its reactivity?
It does not readily undergo electrophilic addition reactions
Describe benzene's structure
1. A six membered ring and three additional degrees of unsaturation
2. All C-C bond lengths are equal
3. Planar
How can the length between every C-C bond in benzene be the same?
They are intermediates caused by each C-C bond being single in one resonance structure and double in the other
Describe the hybridization and molecular geometry of carbon atoms in benzne
Every carbon is sp² hybridized and trigonal planar with 120° bond angles
How many π electrons does benzene have?
six
Why does benzene have a conjugated system?
Its six adjacent orbitals overlap resulting in delocalization of the six electrons over the six carbons of the ring
Is benzene an electrophile or nucleophile? Why?
Nucleophile due to its six π electrons
What is the nomenclature for monosubstituted benzenes?
Add the word 'Benzene' after the substituent name.
What are the three positions for disubstituted benzenes?
ortho (o), meta (m), para (p)
Ortho means the benzene is...
1,2-disubstituted
Meta means the benzene is...
1,3-
disubstitued
Para means the benzene is...
1,4 disubstituted
How do you name polysubstituted benzenes?
Number to give the lowest possible number to each substituent and alphabetize the names.
What four structural criteria must be satisfied for a compound to be aromatic?
A compound must be cyclic, planar, completely conjugated, and follow Hückel's rule.
Why must an aromatic compound be cyclic?
Each p orbital will overlap with p orbitals on two adjacent atoms
Why must an aromatic compound be planar?
All adjacent p orbitals are
able to be aligned so that
the electron density can
be delocalized.
Why must an aromatic compound be completely conjugated?
There will be a p orbital on every atom in the ring
What is Hückel's rule?
an aromatic compound must contain 4n+2 π electrons.
What is an antiaromatic compound?
A cyclic, planar, completely conjugated compound with 4n π electrons.
What is a nonaromatic compound?
A compound that lacks one or more requirements for aromaticity.
What is an annulene?
Hydrocarbons containing a single ring with alternating double and single bonds.
As the number of fused benzene rings increases, so does...
the number of resonance structures.
For a charged aromatic compound to be aromatic, what two things are required?
1. The charged carbon atom must be sp² hybridized
2. The non-bonded electron pair must occupy a p orbital.
Where is the lone pair of a heteroatom located in a heterocycle if the heteroatom is already part of a double bond
it occupies an sp² orbital so it cannot be delocalized over the ring
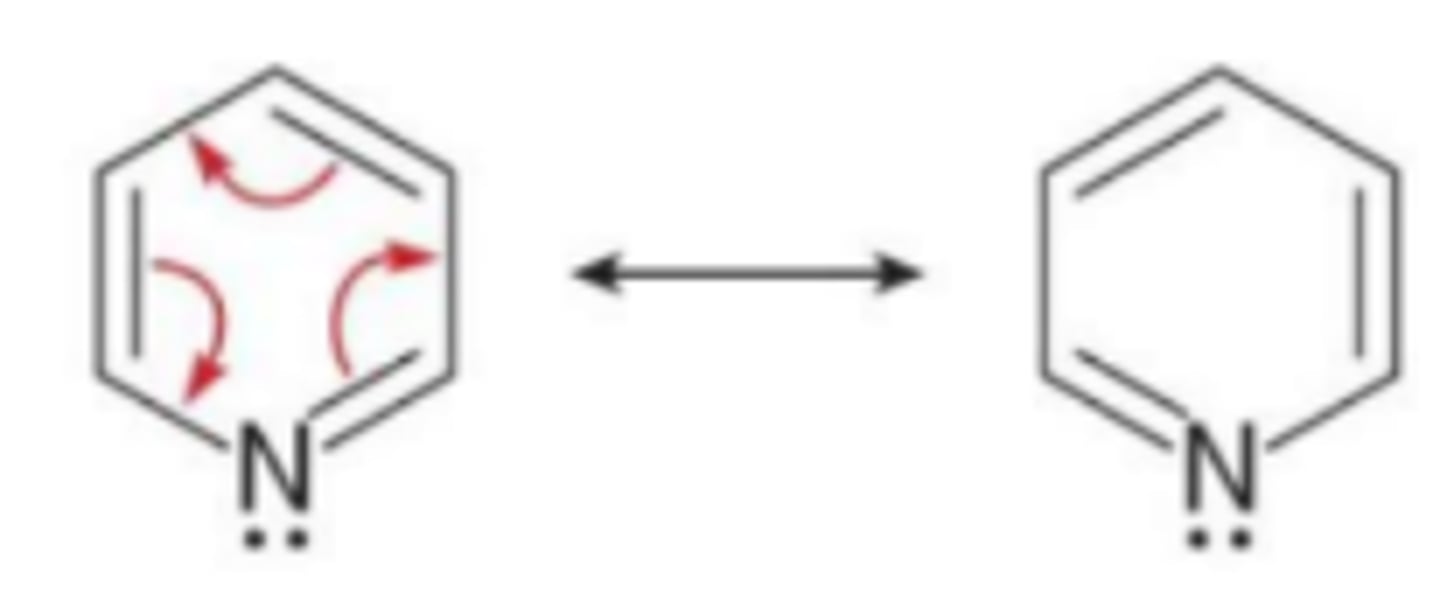
Where is the lone pair of a heteroatom located in a heterocycle if the heteroatom is not part of a double bond
it occupies a p orbital and is delocalized over the ring
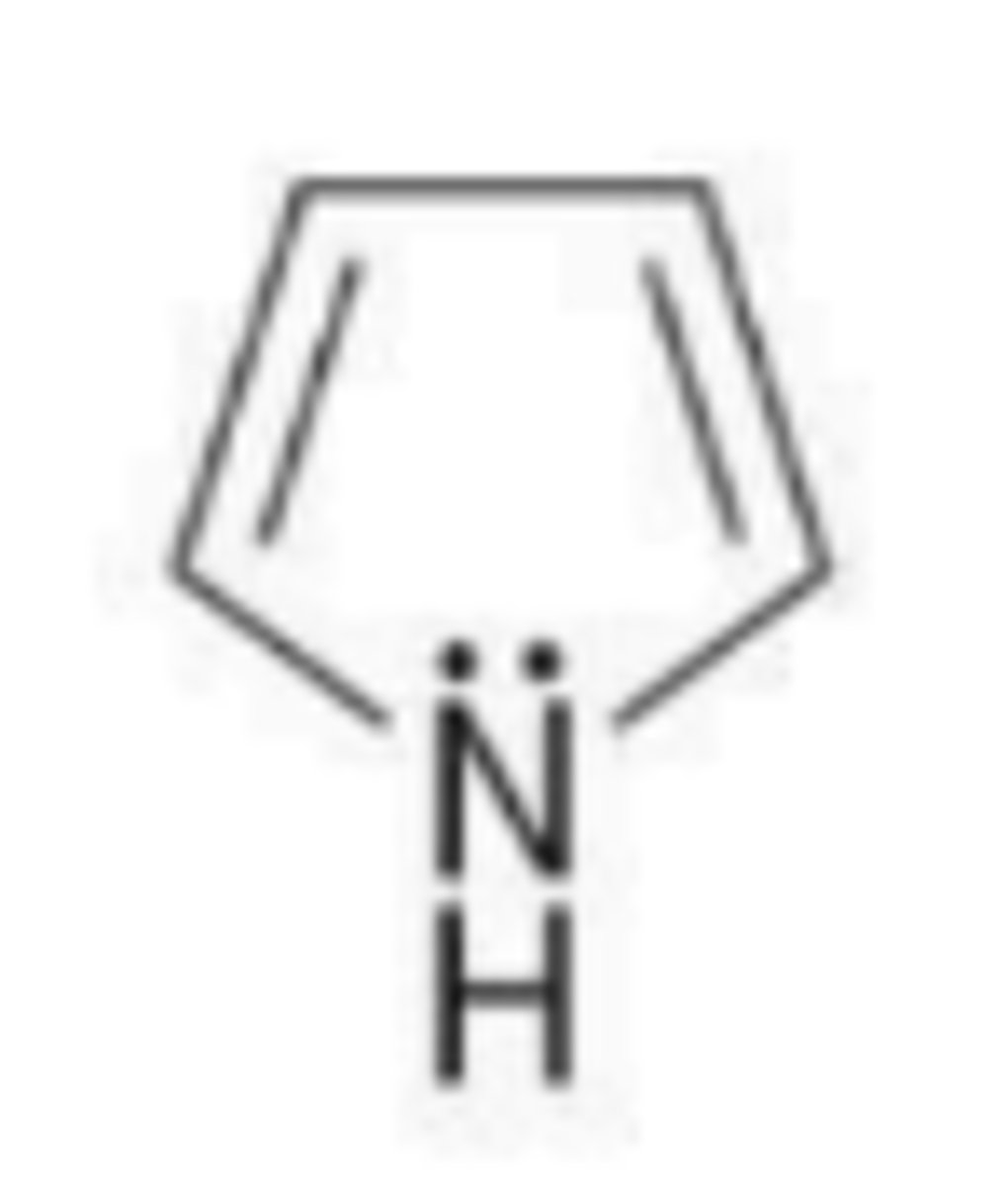
What is the significance of the overlap of p orbitals in benzene?
It creates two rings of electron density above and below the plane of the benzene ring.