Exam 3: Ch. 5 & 6
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
What atmospheric component is responsible for the natural acidity of rain?
nitrogen
ozone
oxygen
carbon dioxide
carbon dioxide
What anthropogenic pollutants are implicated in the formation of most acidic precipitation?
-carbon oxides
-phosphoric acid and hydrochloric acid
-ozone and carbon monoxide
-nitrogen oxides and sulfur oxides
nitrogen oxides and sulfur oxides
Sulfur oxides have been implicated as important contributors to the problem of acid rain. What is the principal anthropogenic source of these compounds?
A. transportation
B. coal fired power plants
C. lightning
D. volcanoes
coal fired power plants
The formation of nitric oxide (NO, a precursor to nitric acid) from oxygen and nitrogen is ordinarily a very slow process. What accounts for its more rapid formation in automobile exhaust?
the extreme conditions involved in gasoline combustion
the presence of nitrogen compounds in gasoline
catalysts present in fuel that facilitate its formation
well-designed catalytic converters
the extreme conditions involved in gasoline combustion
Lakes surrounded by___ have very little acid-neutralizing capacity (can choose more than one)
-marble
-granite
-limestone
granite (II only)
The acid neutralizing capacity of a lake or stream most often derives from the presence of ____ in the surrounding soil or rock
-CaCO3
-HNO3
-NaCl
-Sr(OH)2
CaCO3
Cars and trucks produce most of the NOx emissions and they also suffer the deleterious effects of acid rain. Which negative effects of acid rain affect cars and trucks? ( can choose more than one)
-decreased gas mileage
-damage to paint and finish
-increased susceptibility to rust
-reduced radio reception
damage to paint and finish
increased susceptibility to rust (II & III)
Carbon dioxide is the acid anhydride of which compound?
-CH2O3
-CO
-CH2O
-CHO2
CH2O3
Why have efforts to reduce sulfur oxide emissions met with greater success than those directed at nitrogen oxide emissions reductions?
Sulfur oxides have higher molar masses than nitrogen oxides.
Sulfur oxide emissions come from a limited number of point sources.
Nitrogen oxides are only produced at power plants.
Nitrogen reacts more readily with oxygen than sulfur.
Sulfur oxide emissions come from a limited number of point sources.
The primary product in the combustion of sulfur is:
-H2S
-H2SO4
-H2SO3
-SO2
SO2
What is the current expert consensus concerning the role of acid rain on the health of forests?
-acid rain is indisputably responsible for the declining health of many forests
-acid rain is not responsible for any of the decline observed in many forests
-acid rain weakens trees and the surrounding soil, leaving them susceptible to disease and insects
-there is not sufficient evidence that acid rain has caused an appreciable decline in the health of any forests
acid rain weakens trees and the surrounding soil, leaving them susceptible to disease and insects
The acidity of rain was first studied in
-the mid-1750s
-the mid-1850s
-1900
-the mid-1950s
the mid-1850s
Sulfur dioxide, the primary product of sulfur combustion, further reacts with oxygen as shown:
2 SO2 + O2 --> 2 SO3
What is the final step in the formation of the acidic aerosol?
-reaction of sulfur trioxide with nitrogen oxide
-reaction of sulfur trioxide with water
-reaction of sulfur trioxide with sodium hydroxide
-reaction of sulfur trioxide with nitrogen gas
reaction of sulfur trioxide with water
Which nitrogen-containing substance is generally unreactive?
-NO
-NaNO2
-N2
-NH3
N2
Soil nitrification refers to which process?
-conversion of NO to NO2
-conversion of nitrogen oxides to nitric acid
-conversion of N2 to NO2-
-conversion of NH4+ to NO3
conversion of NH4+ to NO3
Most of the sulfur in coal originates from
decaying vegetation in swamps and peat bogs.
sulfate ions naturally present in sea water.
sulfur deposits located adjacent to coal deposits.
the natural sulfur content of the plants making up the coal.
sulfate ions naturally present in sea water.
Why did the industrial-scale production of ammonia from nitrogen and hydrogen present a difficult challenge?
-ammonia is explosive
-nitrogen is difficult to purify
-nitrogen is unreactive
-hydrogen is unreactive
nitrogen is unreactive
In addition to fossil fuel combustion, what other facet of modern industrial society contributes to the formation of acid rain?
the use of chlorofluorocarbons
deforestation
fertilizer production
the use of river water in cooling towers
fertilizer production
The pH of rain water falling through an unpolluted atmosphere is closest to:
4.7
5.4
7.0
8.7
5.4
Which reactive nitrogen compound is produced using the Haber-Bosch process?
A. NaNO3
B. KNO3
C. NH3
D. NO2
NH3
Bases are substances that increase the hydroxide ion concentration in aqueous solution. Why does ammonia (NHs), which does not contain a hydroxide group, act as a base?
Ammonia acts as a base only in the presence of hydroxide ion-containing compounds.
Ammonia molecules remove protons from water molecules, forming hydroxide ions.
Ammonia molecules donate protons to water molecules, forming hydroxide ions.
Ammonia acts as a base only in the presence of very strong acids.
Ammonia molecules remove protons from water molecules, forming hydroxide ions.
Most aquatic life in lakes cannot survive in water with a pH less than
-9
-8
-6
-5
5
A substance that is hygroscopic
-absorbs and releases moisture from the atmosphere
-releases water to the atmosphere
-purifies water by removing nitrate and sulfate ions
-prevents atmospheric gases from forming aerosols
absorbs and releases moisture from the atmosphere
The production of nickel and copper metals from their ores involves sulfur dioxide emissions because
the ores of these elements are composed primarily of NiSOz and CuSO2.
the ores of these elements are composed primarily of NiS and CuS.
to release the metals from their ores, the ores are heated with sulfur-containing coal.
the release of metals from their ores involves the reaction of SOs with the ore.
the ores of these elements are composed primarily of NiSOz and CuSO2.
Which of the following compounds contributes to "dry" acid deposition?
A. Calcium carbonate
B. Ammonium nitrate
C. Sodium hydroxide
D. All of the above
Ammonium nitrate
Why are automobiles a contributor to acid rain when there is no sulfur in gasoline to make SOx?
A. This is incorrect because there is sulfur in gasoline.
B. Catalytic converters contain sulfur compounds.
C. Hydrocarbons contribute to acid rain, also.
D. Nitrogen from the atmosphere combines with oxygen in the hot engine to make NOx.
Nitrogen from the atmosphere combines with oxygen in the hot engine to make NOx.
Which of the following is not carried out by bacteria in soil?
A. Atmospheric nitrogen is fixed in soils.
B. Ammonium is converted into nitrites.
C. Nitrites are converted to nitrates.
D. All of these are carried out by bacteria.
All of these are carried out by bacteria.
Which of the following countries is the largest consumer of coal (by total mass)?
A. China
B. United States
C. India
D. Indonesia
China
Percentage of Earth's water in oceans
-97.4%
-50%
-2.59%
-0.014%
97.4%
What is an aquifer?
-holding tank for disinfected water at municipal water works
-Roman water purification system
-large pool of water trapped in sand and gravel below surface of Earth
-solution in which solvent is water
large pool of water trapped in sand and gravel below surface of Earth
definition of groundwater
any water taken from aquifers
The water with the least harmful contaminants is
-ocean water
-surface water from lakes
-groundwater from aquifers
-surface water from rivers
groundwater from aquifers
Definition of specific heat:
-quantity of heat energy that must be absorbed to cause one gram of a liquid substance to boil
-quantity of heat energy that must be absorbed to increase temp. of one gram of a substance one degree Celsius
-boiling point of a liquid substance in Celsius
-difference between freezing and boiling point
quantity of heat energy that must be absorbed to increase temp. of one gram of a substance one degree Celsius
Electronegativity is...
-a relative measure of an atom's attraction for electrons it shares in a covalent bond
-the total negative charge on a polyatomic anion
-the same for all the elements in a family or group
-decreases from left to right across a period on the periodic table
a relative measure of an atom's attraction for electrons it shares in a covalent bond
The fact that carbon (C) is less electronegative than nitrogen (N) means that in a C — N bond, the
shared electrons are closer to the N atom than to the C atom.
A polar covalent bond is created when
-a metallic element reacts with a nonmetallic element
-two atoms share their bonding electrons unequally
-two atoms share three or more electrons'
-two atoms of same element form double bonds
two atoms share their bonding electrons unequally
Which covalent bond is most polar?
H-H
H-O
H-N
H-S
H-O
Drawing of two molecules; A points to solid line bond between O and H, B points to dotted line bond between H and O in different molecules. Which statement is correct?
-A:covalent, B: hydrogen bond, B is stronger
-A:hydrogen, B: covalent, A is stronger
-A: covalent, B: hydrogen, A is stronger
-A: hydrogen, B: covalent, B is stronger
A: covalent, B: hydrogen bond
A is stronger
(covalent bonds are stronger than hydrogen bonds)
Hydrogen bonds are
-ten times stronger than typical covalent bonds
-are 1/10 as strong as covalent bonds
-found in every molecule containing more than one H
-found only between H atoms
are 1/10 as strong as covalent bonds
Which best symbolizes the hydrogen bonding between two water molecules?
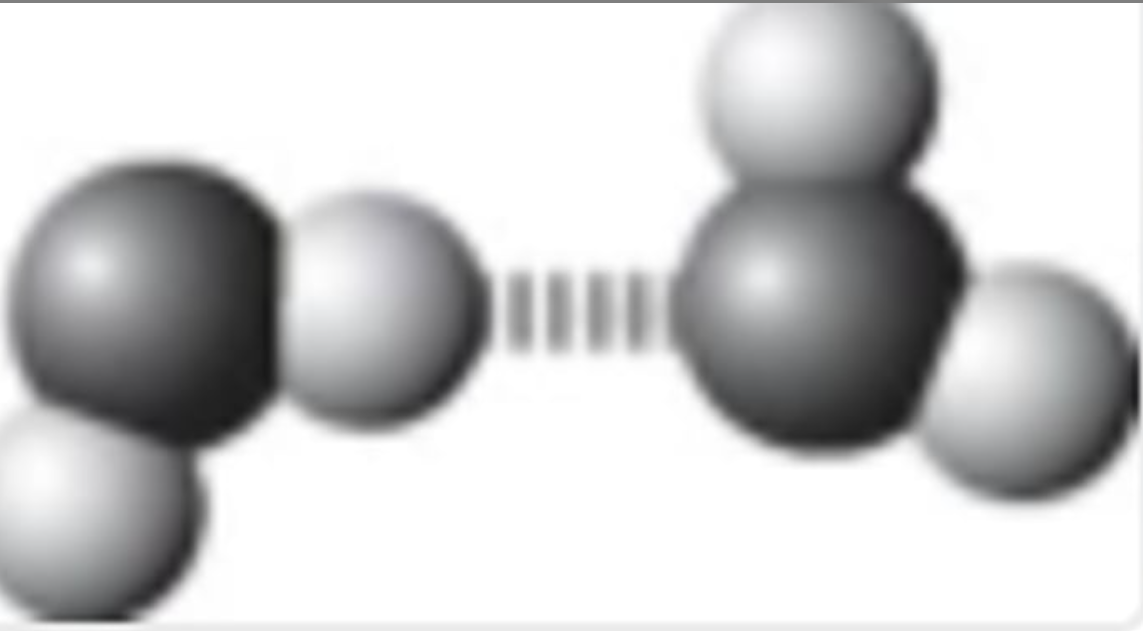
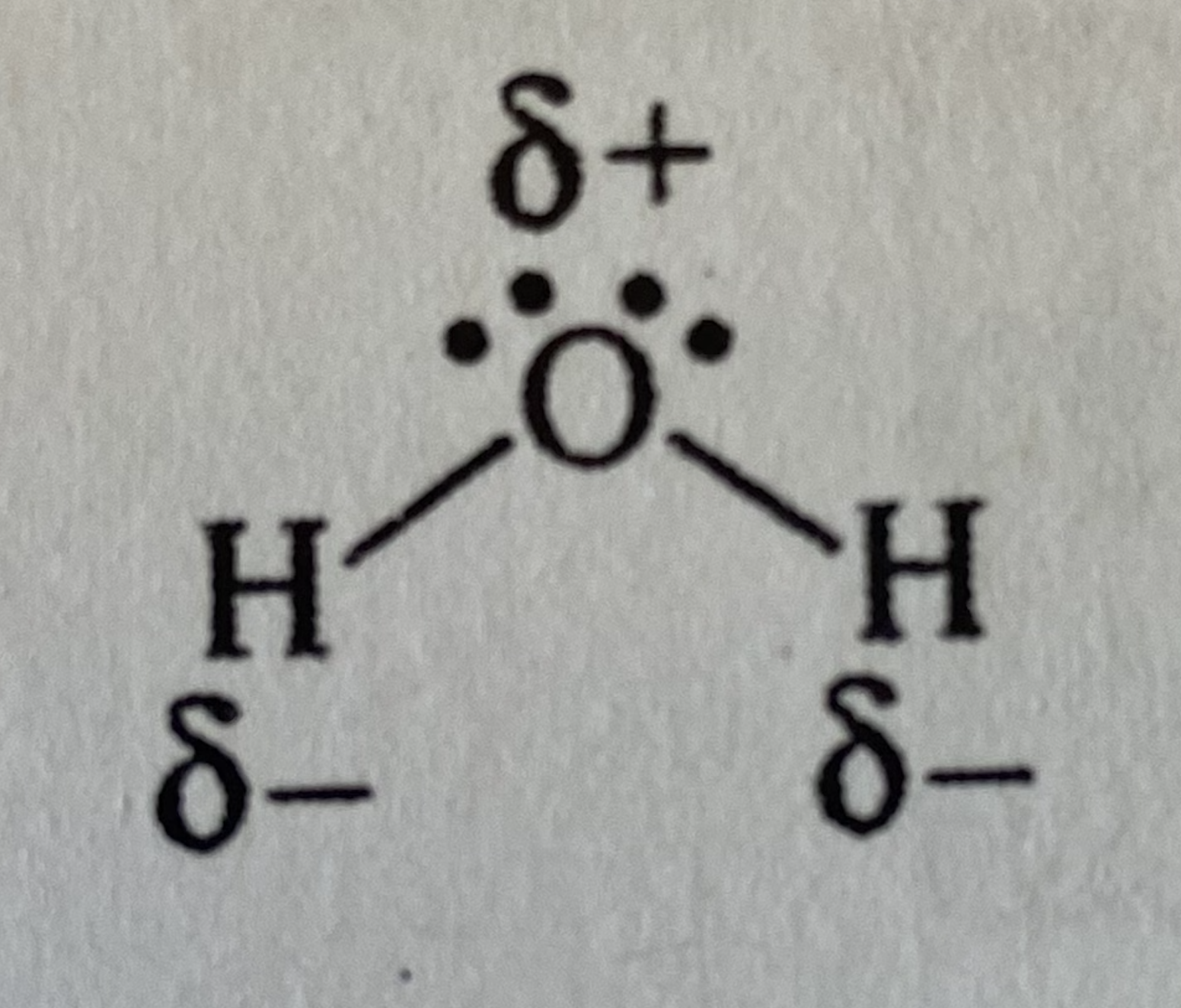
In Lewis structure of water, where do partial charges go and what are they?
positive partial charge on each H, negative partial charge on O
Which is not a consequence of hydrogen bonding in water?
-water has a high boiling point
-ice floats on water
-water has a high specific heat
-is colorless and odorless
is colorless and odorless
Water has one of the highest specific heats. This means that the temp. of a water sample____ with the input of a ____amount of energy.
-decreases greatly, small
-decreases only slightly, large
-increases only slightly, large
-increases greatly, small
increases only slightly, large
Which statement is not true?
-In forming a solution, the solute dissolves other substances called solvents
-water is a solvent in a aqueous solution
-pure water does not conduct electricity
-electrolytes in water form a solution that conducts electricity
in forming a solution, the solute dissolves other substances called solvents
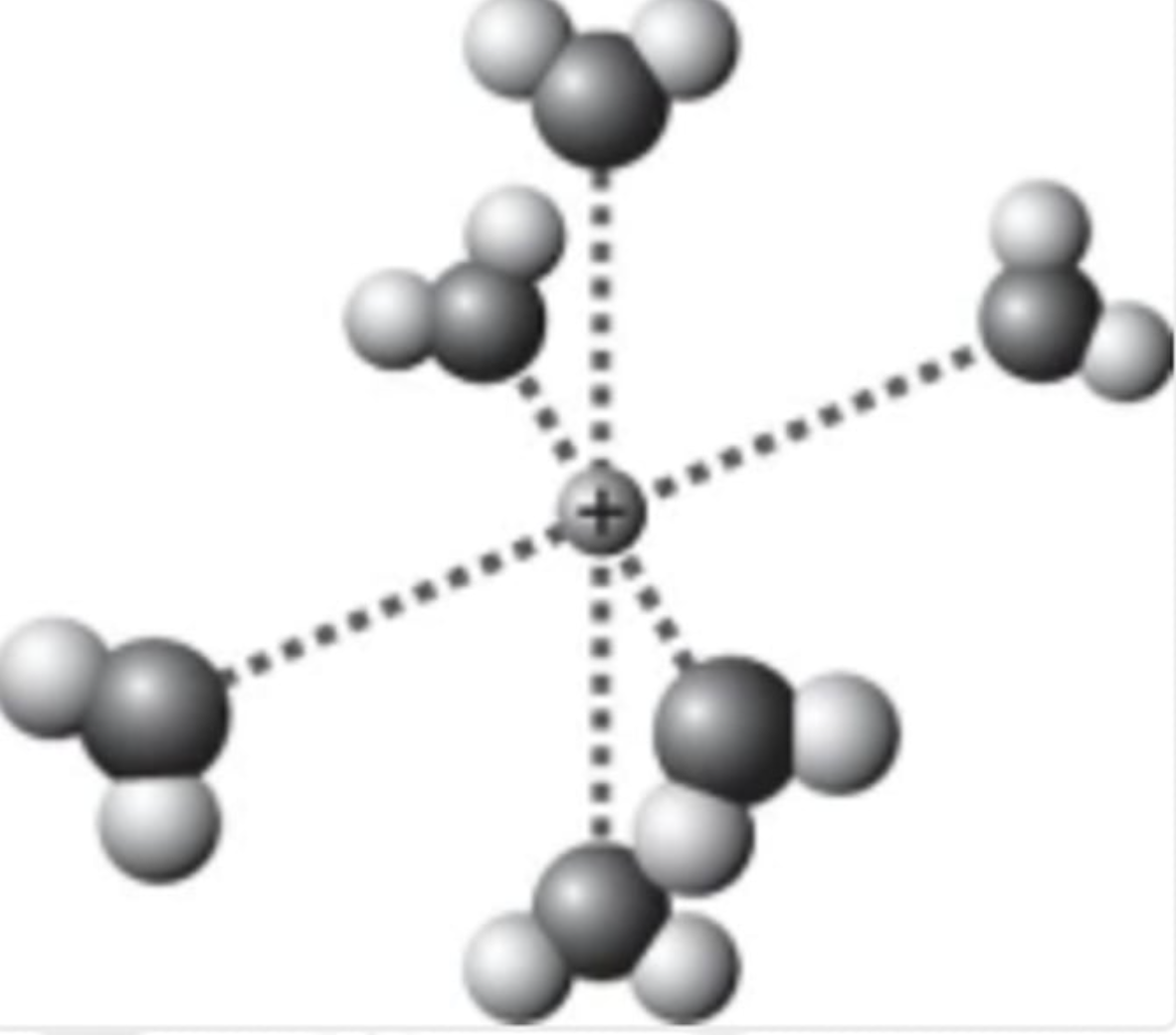
Which is the best representation showing a sodium cation in water?
Which corresponds to the composition of the ion typically formed by fluorine?
9, 10, 1-
Which corresponds to the composition of the ion typically formed by magnesium?
12, 10, 2+
An aqueous solution of KCl is tested for conductivity. What do you predict will happen?
-the bulb will not light up; KCl is a nonelectrolyte
-the bulb will not light up. KCl is in the molecular form when dissolved in water
-the bulb will shine dimly; KCl is only partially ionized
-the bulb will shine brightly; KCl is highly ionized
the bulb will shine brightly; KCl is highly ionized in aqueous solution
How well would a conductivity testing setup with a bulb work with sucrose? Why?
the bulb will not light up; sucrose is in the molecular form in aqueous solution.
When the _____ molecules of ethanol are added to water, the ethanol molecules...
-nonpolar; are attracted to the nonpolar water molecules
-polar; form hydrogen bonds with the polar water molecules
-polar; form covalent bonds with the polar water molecules
-nonpolar; are not attracted to the polar water molecules
polar; form hydrogen bonds with polar water molecules
Which compound should be most soluble in water?
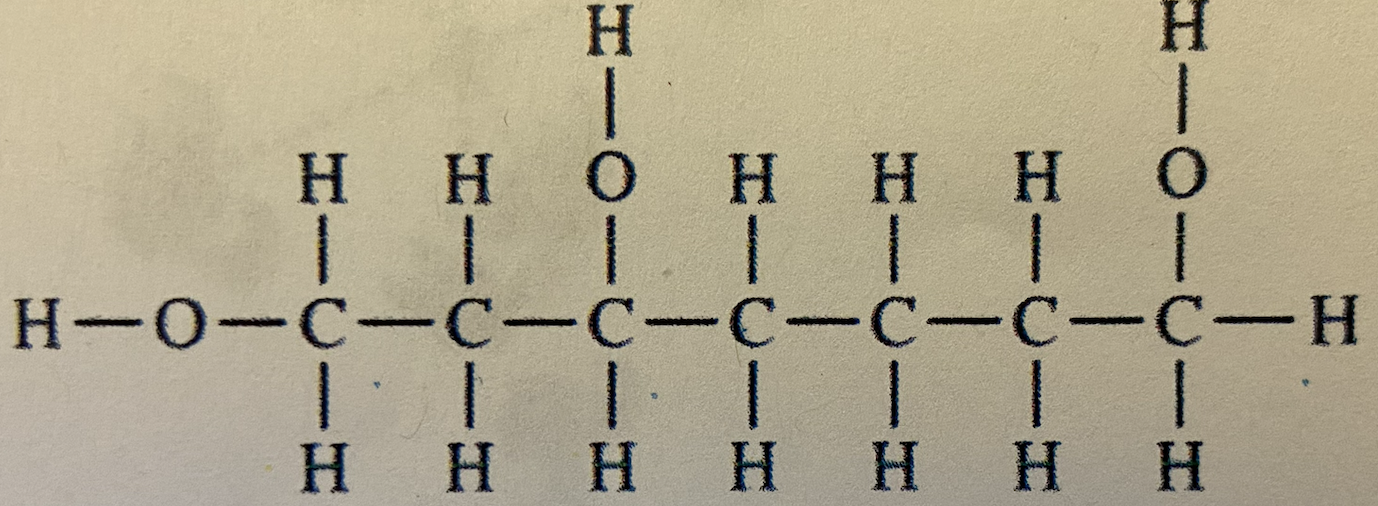
When table sugar dissolves in water, what type(s) of attraction between water and sugar molecules occur?
-hydrogen bonding
-polar-polar interactions
-covalent bonding
hydrogen bonding and polar-polar interactions
The attractions between anions and cations in a crystal are known collectively as
-covalent bonds
-polar covalent bonds
-hydrogen bonds
-ionic bonds
ionic bonds
Which is not a form of chlorine used to disinfect water?
-chlorine gas
-chloroform
-sodium hypochlorite
-calcium hypochlorite
chloroform
The steps in the typical purification process for municipal drinking water are
-filtration, distillation, chlorination, and filtration
-filtration, chlorination, ozonation, and flocculation
-filtration, flocculation, filtration, and chlorination
-flocculation, filtration, UV irradiation, and filtration
filtration, flocculation, filtration, and chlorination
Major disadvantages of using ozone to disinfect water?
-more expensive, leaves odor in water
-causes trihalomethane formation, leaves odor
-causes trihalomethane formation, more expensive
-does not provide long-term protection, and is more expensive
does not provide long-term protection against waterborne viruses because it decomposes quickly and it is more expensive than chlorination
A disadvantage of ozonation over chlorination is
the higher cost of ozonation and ozonation does not protect the water after the initial process is complete
The main reason that water supplies are chlorinated is
-to produce gels that remove solids from water
-to kill disease-causing organisms in water
-to soften the water
-to precipitate lead salts from the water as insoluble lead chloride
to kill disease-causing organisms in water
Which form of water disinfection continues to provide protection after the water leaves the plant?
-chlorination
-ozonation
-UV radiation
-filtration
chlorination
Potable water is water that
is fit for human consumption
Illustration of structure of a soap molecule; which region would dissolve in a nonpolar substance like grease? (4 regions)
region 1
Which ions, if present in drinking water, would pose a significant health risk?
-Pb2+
-Hg2+
-Cd2+
-Ca2+
all except Ca2+ (I, II, & III
What is reverse osmosis used for?
-to purify water by ion exchange
-to remove dissolved salts from seawater
-for the ozonation of water
-to add fluorine to municipal water
to remove dissolved salts from sea water (potentially A is correct too)
The drawing shows a simple way of purifying salt water. What is the process called?
distillation
What type of water may be considered pure?
distilled water
What is biological oxygen demand? (BOD)
-a measure of the amount of dissolved oxygen microorganisms use up as they decompose organic wastes found in water
-a measure of the amount of dissolved oxygen necessary to purify wastewater
-a measure of the phosphate ion concentration of water
-a measure of the nitrate concentration of water
a measure of the amount of dissolved oxygen microorganisms use up as they decompose organic wastes found in water
Why is water a liquid in room temperature instead of gas?
Water has hydrogen bonds between the molecules
pH=
-log[H+]
pOH=
-log[OH-]
14=
pH + pOH
10^-14=
[H+]x[OH-]