Alkene and Alkyne Reactions (Orgo 1)
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
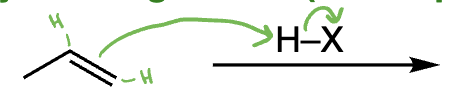
radical hydrohalogenation = radical hydrohalogenation (alkene + HBr + ROOR) | Anti-Markovnikov addition, radical mechanism, no rearrangements
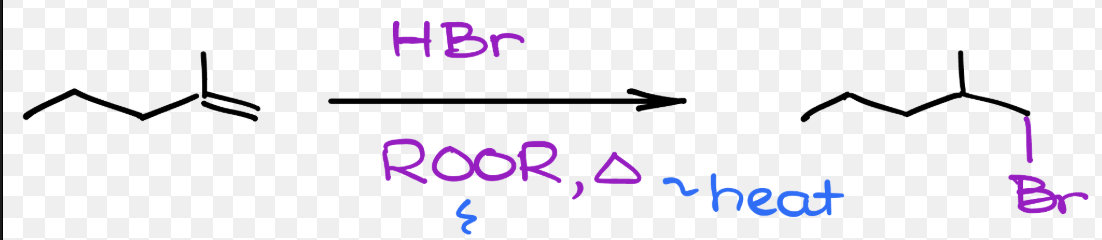
hydrohalogenation of alkenes = hydrohalogenation (alkene + HX) | Markovnikov addition, carbocation intermediate, rearrangements possible
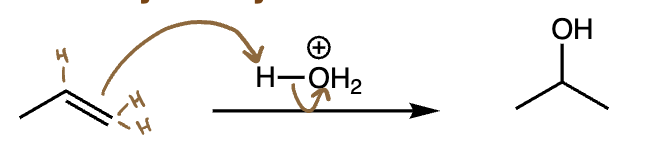
acid-catalyzed hydration = acid hydration (alkene + H2O + H2SO4) | Markovnikov addition of OH, carbocation intermediate, rearrangements possible
oxymercuration-demercuration = oxymercuration-demercuration (alkene + Hg(OAc)2/H2O then NaBH4) | Markovnikov alcohol formation, no carbocation rearrangements
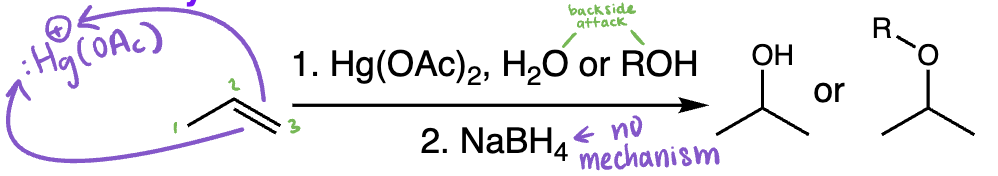
hydroboration-oxidation = hydroboration oxidation (alkene + BH3/THF then H2O2/NaOH) | Anti-Markovnikov alcohol, syn addition
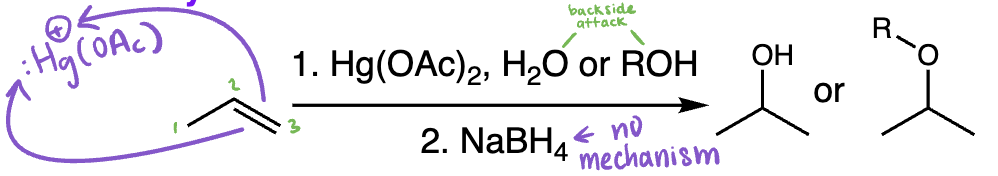
halogenation of alkenes = halogenation (alkene + Br2 or Cl2) | Anti addition via halonium ion, vicinal dihalide
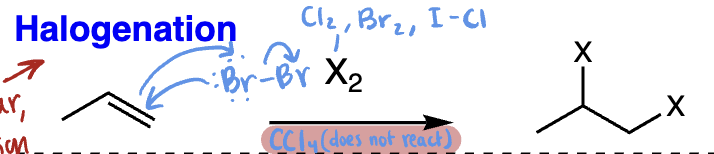
halohydrin formation = halohydrin formation (alkene + Br2/H2O or Cl2/H2O) | Anti addition, OH goes to more substituted carbon
syn dihydroxylation = syn dihydroxylation (alkene + OsO4 or cold dilute KMnO4) | Syn addition of two OH groups, vicinal diol
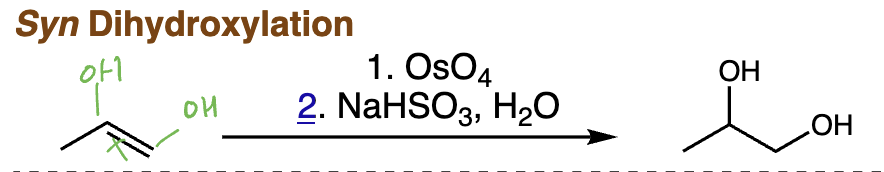
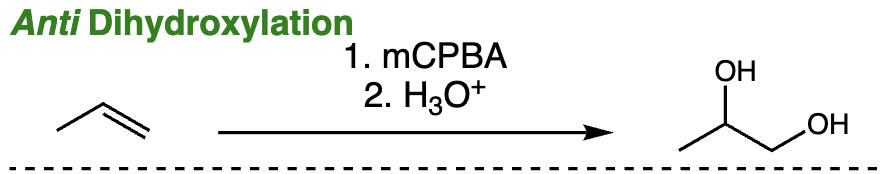
anti dihydroxylation = anti dihydroxylation (alkene + mCPBA then H3O+) | Epoxide intermediate then anti diol formation
epoxidation = epoxidation (alkene + peroxyacid e.g. mCPBA) | Syn addition, epoxide formation
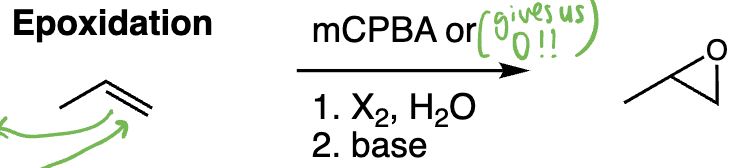
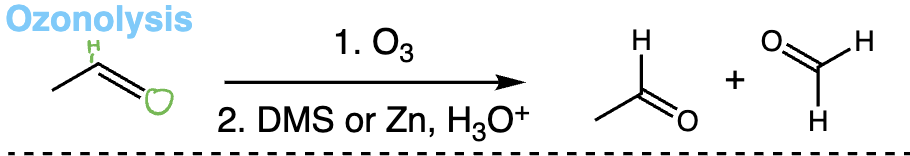
ozonolysis = ozonolysis (alkene + O3 then DMS or Zn/H2O) | Cleaves double bond to aldehydes/ketones
oxidative ozonolysis = oxidative ozonolysis (alkene + O3 then H2O2) | Cleaves double bond to ketones/carboxylic acids
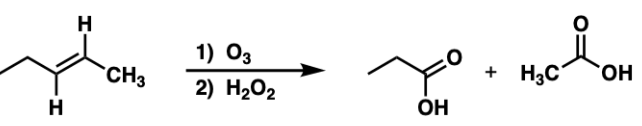
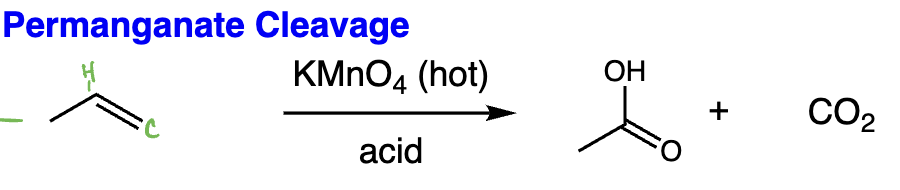
oxidative cleavage with KMnO4 = oxidative cleavage (alkene + hot KMnO4) | Cleaves double bond, strong oxidation to ketones/acids
complete hydrogenation = complete hydrogenation (alkyne + H2 + Pd/C, Pt, or Ni) | Reduces alkyne to alkane
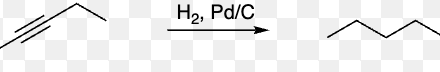
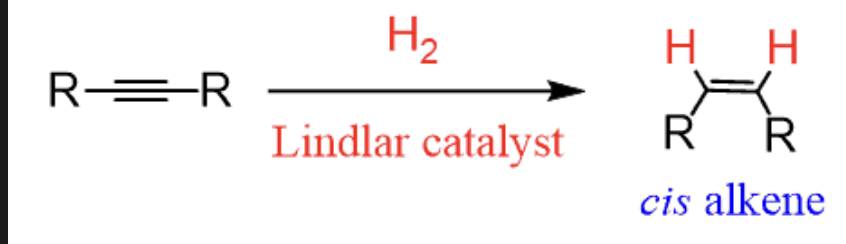
Lindlar reduction = Lindlar reduction (alkyne + H2 + Lindlar catalyst) | Syn addition, forms cis-alkene
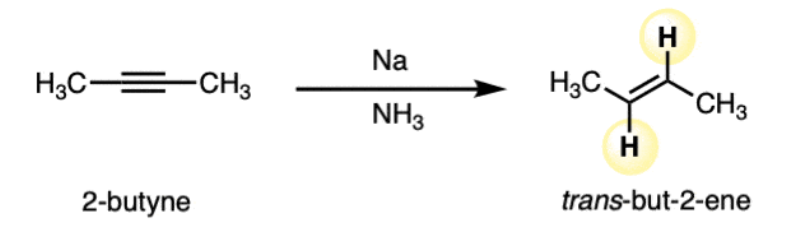
dissolving metal reduction = dissolving metal reduction (alkyne + Na or Li in NH3) | Anti addition, forms trans-alkene
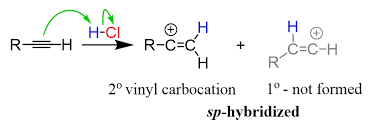
hydrohalogenation of alkynes = hydrohalogenation (alkyne + HX) | Markovnikov addition, forms vinyl halide then geminal dihalide with excess HX
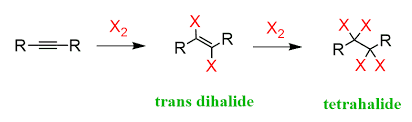
halogenation of alkynes = halogenation (alkyne + X2) | Anti addition, forms dihaloalkene then tetrahalide with excess
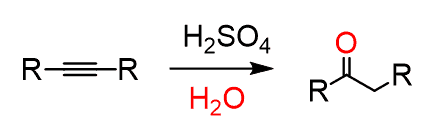
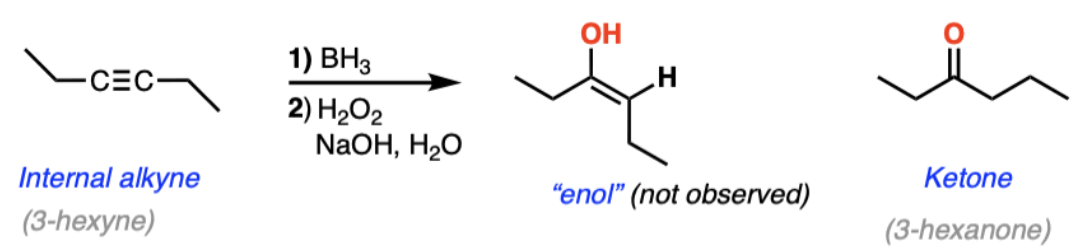
acid-catalyzed hydration of alkynes = acid hydration (alkyne + H2O + H2SO4 + HgSO4) | Markovnikov addition, enol intermediate tautomerizes to ketone
hydroboration-oxidation of alkynes = hydroboration oxidation (alkyne + R2BH then H2O2/NaOH) | Anti-Markovnikov hydration, forms aldehyde (terminal) or ketone (internal)
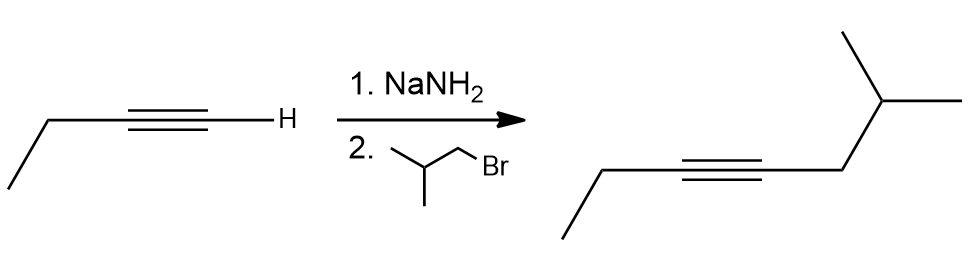
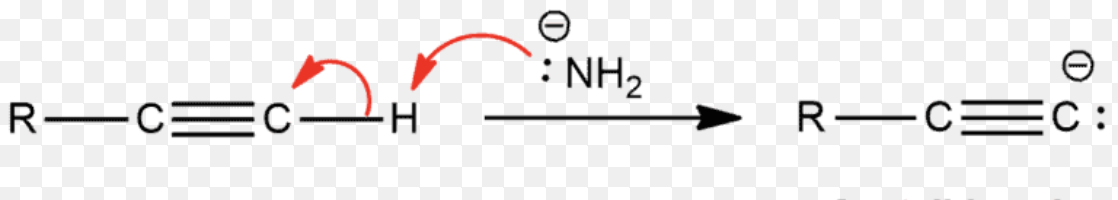
alkyne deprotonation = alkyne deprotonation (terminal alkyne + NaNH2) | Forms acetylide anion, strong base required
acetylide alkylation = acetylide alkylation (acetylide + primary alkyl halide) | SN2 reaction, chain extension