Nature of covalent and dative covalent bonds (3.1.3.2) + macromolecular structures
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
What is a covalent bond?
Covalent bonds occur between two non-metals.
Electrostatic attraction between the nuclei of two atoms and the bonding electrons of their outer shells.
What are the structures covalently bonded atoms can take?
Covalently bonded atoms can either have simple molecular structures (Cl2, CO2, O2) or macromolecular structures (diamond, silicon dioxide, graphite)
How can you represent covalent bonds?
Dot and cross diagram
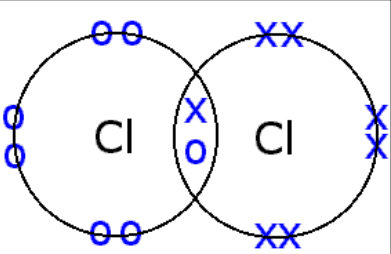
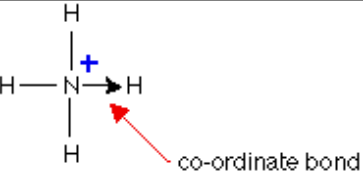
What are dative/coordinate bonds and how are they indicated?
Dative bonds - when the electrons in a shared pair are supplied from a single atom.
It is indicated using an arrow from the lone electron pair
What are the physical properties of simple molecular compounds?
Melting and boiling points
They have low MP and BP as they have intermolecular forces that require low amounts of energy to break
Conductivity
Very poor conductors as their structure contains no charged particles
What are the physical properties of a macromolecular compounds?
Melting and boiling points
They have many strong covalent bonds that require a lot of energy to break so they have high MP and BP
Conductivity
Most don’t conduct electricity except for graphite
Hardness
They are hard due to their rigid structure. Except graphite whose layers can slide over each other due to the weak intermolecular forces between them