Ch 16: Reactions of Aromatic Compounds
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
What is the characteristic reaction of benzene?
Electrophilic aromatic substitution
What occurs in an electrophilic aromatic substitution reaction?
A hydrogen atom of the ring is replaced bу an electrophile.
What are the two steps of the general mechanism for electrophilic aromatic substitution?
1. Addition of the electrophile to form a resonance-stabilized carbocation; 2. Deprotonation with a base
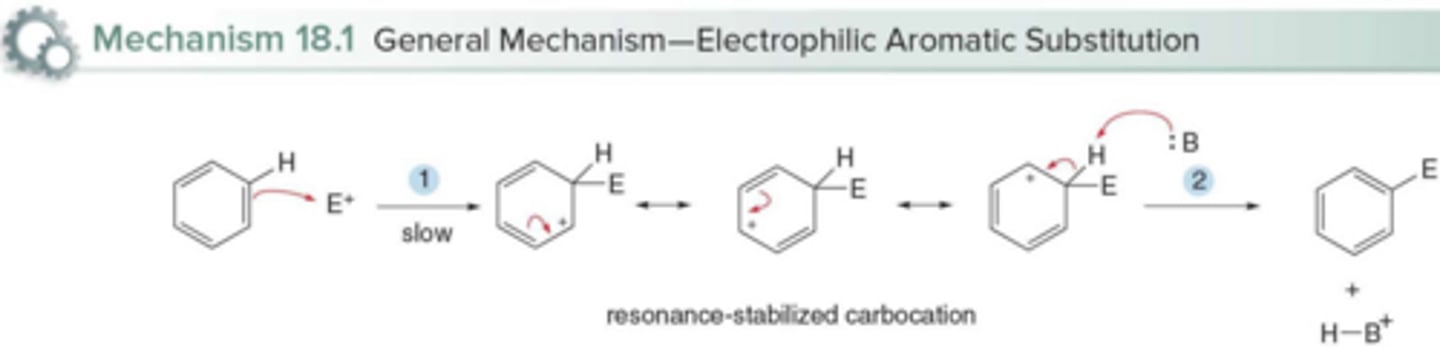
Which step in the electrophilic aromatic substitution mechanism is rate-determining?
The addition of the electrophile, because the aromaticity of the ring is lost
What is benzene halogenation?
Replacement of H by X (Cl or Bl)
What is benzene nitration?
Replacement of H by NO₂
What is benzene sulfonation?
replacement of H by SO₃H
What is Friedel-Crafts Alkylation?
Replacements of H by R (an alkyl group)
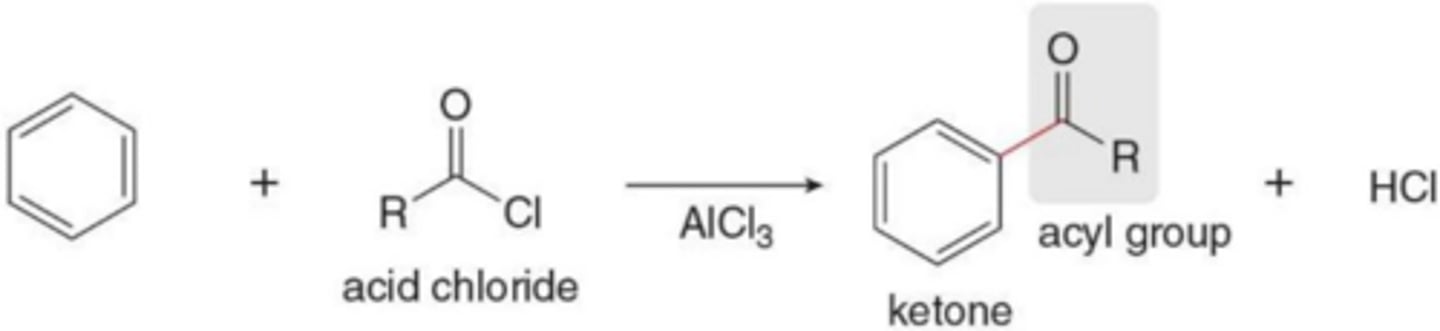
What is Friedel-Crafts Acylation?
Replacement of H by RCO (an acyl group)
What catalyst is required for benzene halogenation?
A Lewis acid catalyst, such as FeCl₃ or FeBr₃
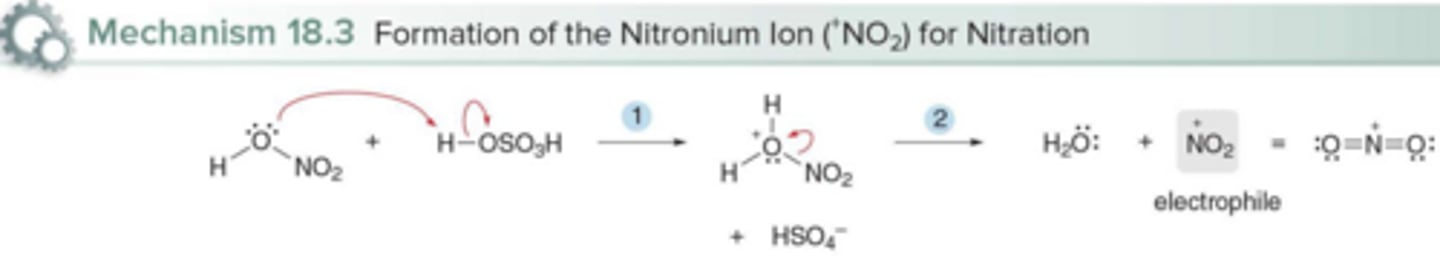
What is the electrophile in benzene nitration? How is it formed?
The nitronium ion (NO₂⁺); Protonation of HNO3 followed by dehydration
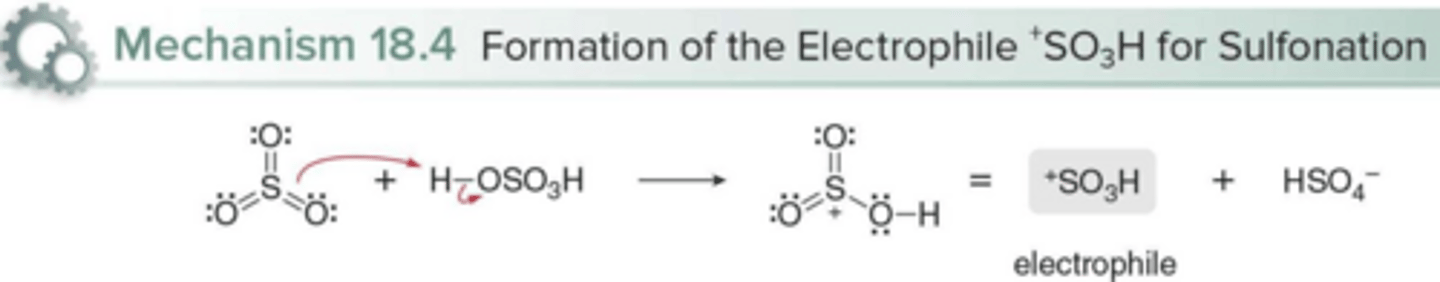
What is the electrophile in benzene sulfonation? How is it formed?
SO₃H⁺; Protonation of sulfur trioxide (SO₃)
What is the primary purpose of Friedel-Crafts reactions?
To form new carbon-carbon bonds
What reagents are used for Friedel-Crafts alkylation?
An alkyl halide and a Lewis acid
What reagents are used for Friedel-Crafts acylation?
An acid chloride and AlCl₃
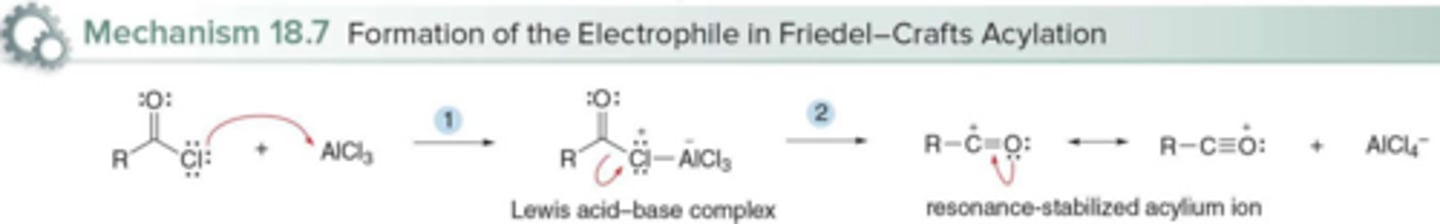
What is the electrophile formed during Friedel-Crafts acylation?
An acylium ion (R-C⁺=:O:)
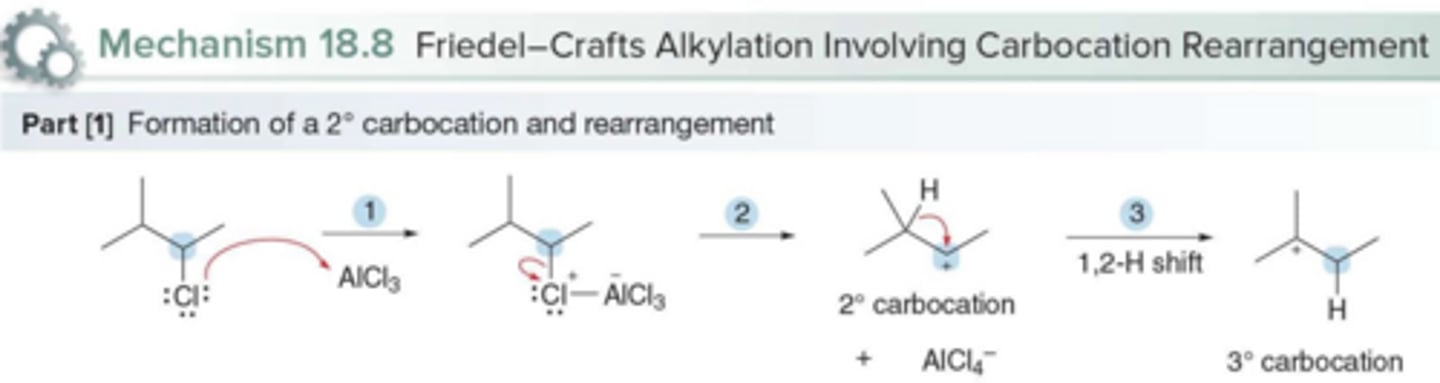
Why do vinyl and aryl halides not react in Friedel-Crafts alkylation?
The carbocation formed from these compounds is too unstable
When can rearrangements occur in Friedel-Crafts alkylation reactions?
when primary and secondary alkyl halides are used as starting materials to make the carbocation formed more stable
Besides alkyl halides, what other compounds can be used as starting materials for Friedel-Crafts alkylation?
Any compound that readily forms a carbocation, such as alkenes and alcohols
What is the product of Friedel-Crafts acylation?
A ketone
In the mechanism of Friedel-Crafts alkylation with 1° alkyl chlorides, what serves as the electrophile?
a lewis-acid baswe complex formed from the reaction of the lewis acid AlCl₃ with the alkyl chloride
In the mechanism of Friedel-Crafts alkylation with 2° and 3° alkyl chlorides, what serves as the electrophile?
The 2° or 3° carbocation
What two factors determine whether a substituent is electron-donating or electron-withdrawing?
Inductive effects and resonance effects.
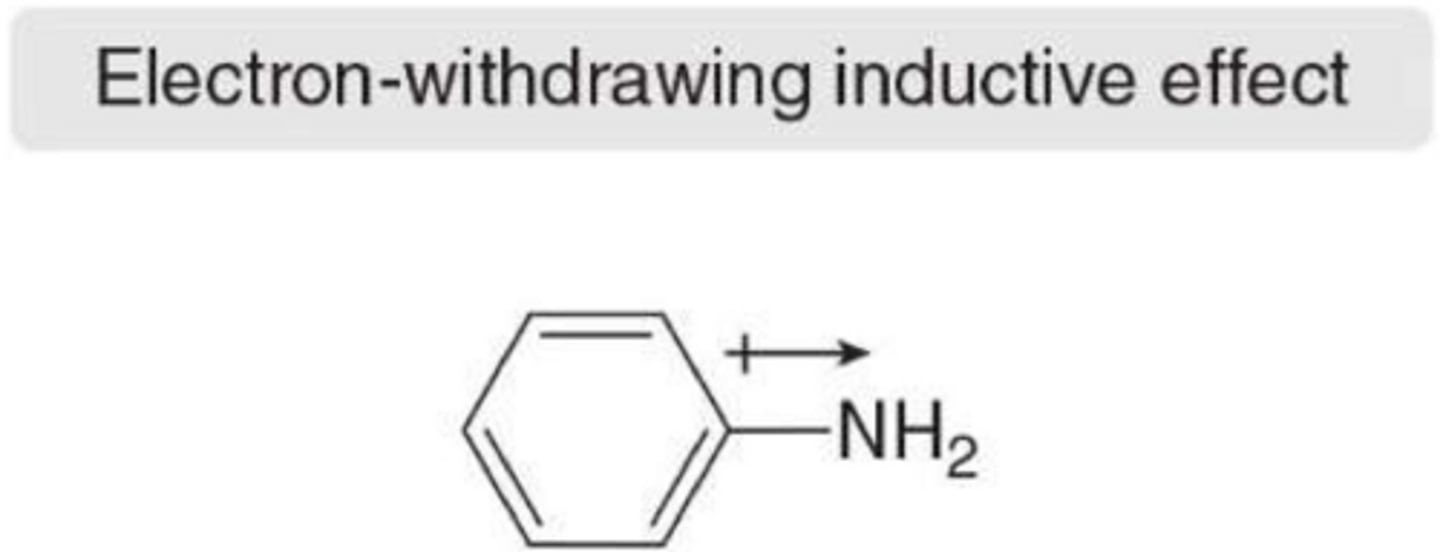
What causes inductive effects in a ring?
electronegativity of the atoms in the substituent and the polarizability of the substituent group
What is the primary cause of an electron-withdrawing inductive effect?
The presence of atoms more electronegative than carbon, such as N, O, and X.
What inductive effects do atoms more electronegative than a carbon induce? Why?
electron-widrawing as they pull electron density away from carbon
What inductive effects do polarizable alkyl substituent groups induce?
electron donating as they donate electron density to the ring
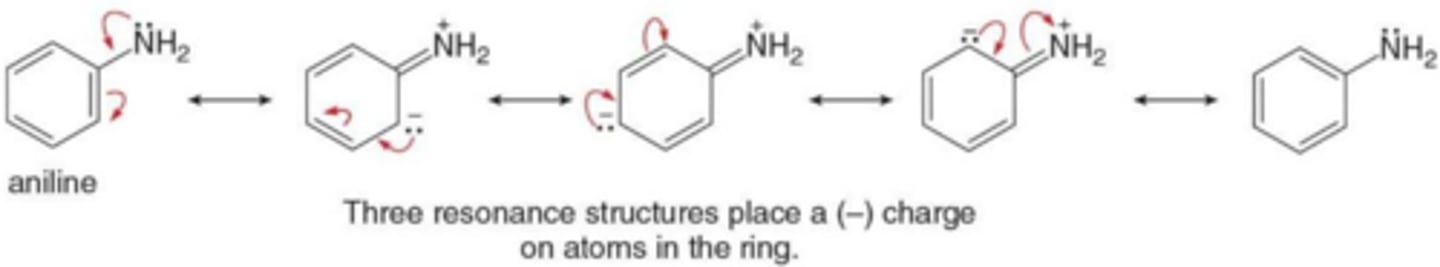
When is a resonance effect electron donating?
When the resonance structures place a negative charge on carbons of the benzene ring.
When are electron donating resonace effects observed?
When atom Z having a lone pair of electrons is bonded directly to a benzene ring
When is a resonance effect electron withdrawing?
When the resonance structures place a positive charge on carbons of the benzene ring.
When are electron withdrawing resonace effects observed?
in substituted benzenes having the general structurre Benzne-Y=Z where Z is more electronegative than Y.
Are neutral O and N atoms EDGs or EWGs? Why?
EDG. Resonance effects Dominate
How do electron-donating groups (EDGs) affect the rate of electrophilic aromatic substitution?
They activate the ring, making the reaction faster by lowering the energy of the transition state
How do electron-withdrawing groups (EWGs) affect the rate of electrophilic aromatic substitution?
They deactivate the ring, making the reaction slower than benzene by increasing the the energy of the transition state
What are the three general types of substituents based on their directing effects?
Ortho/para activator directors, ortho/para deactivators, and meta directors.
What is the general structure of ortho, para directors?
-R or -Z:
When are ortho, para directors activators?
When Z= N or O
Which specific group of substituents are ortho/para directors and only deactivators?
The halogens (F, Cl, Br, I).
All meta directors are...
deactivators
What is the relationship between carbocation stability and reaction rate in electrophilic aromatic substitution?
Increased carbocation stability lowers the transition state energy, resulting in a faster reaction rate.
Why do Friedel-Crafts reactions fail with meta-directing substituents?
The substituent strongly deactivates the ring, making it unable to undergo the reaction.
Why do Friedel-Crafts reactions fail with strong activators like -NH2?
The product bears a positive charge adjacent to the ring, which strongly deactivates it.