6. SWITCHING ART IN VIROLOGICALLY SUPPRESSED PWH
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
When can you consider switching ART in people who are well-suppressed?
Most studies have studied switches after at least 6 months of stability on their current regimen. E.g. probably not a good idea to switch too often.
AND YOU NEED TO REVIEW FULL ART HISTORY, including FAILED ART, CUMULATIVE RESISTANCE TESTS, AEs.
AND continue HBV treatment in HIV/HBV co-infection
AND check DDIs with ART you are considering and their drug list
AND check pregnancy status or potential

After switching PWH regimen, how long should you provide close follow up, including phone calls, if needed? And when should you re-check their viral load?
Follow up closely for 3 months
Check VL at 4 weeks
When switching a virologically suppressed PWH to another regimen, does past history of resistance/failed regimens matter? Or do mutations fade out over time?
It totally matters. You need to consider past resistance as current resistance.
When switching a virologically suppressed PWH to another regimen, are in-class switches or out-of-class switches riskier?
Generally out-of-class switches are much riskier, because you may not have all their resistance history. Deep sequencing can mitigate this.
Examples of switching strategies table
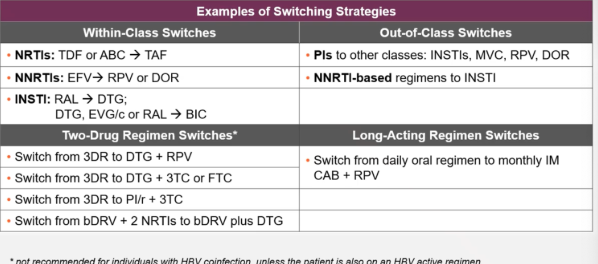
Potential switch options studied for suppressed PWH with no history of resistance. Table

If you have a legitimate reason to switch someone from a PI to another class (e.g. intolerability) while they are virologically suppressed, what is the risk?
Studies show that taking away a high resistance drug, specifically PIs, and adding a lower barrier resistance drug, specifically in patients with previous treatment failures, may precipitate a new treatment failure.
What happens if you accidentally DC a HBV drug in someone with HIV/HBV while switching regimens?
reactivation of HBV virus and hepatocellular damage
Do TAF and TDF both have good HBV suppression?
Yes. Either one is good for HBV suppression.
There has been many studies looking at switching TDF regimens to TAF regimens. Table. What were the main summarized results of these studies?
All showed non-inferior or superiority of TAF vs. TDF.
Increase in bone mineral density
Stability of eGFR and decrease in tubular proteinuria
Early weight gain after switch from TDF to TAF (0-9 months)

After switching ART in someone virologically supressed, how do you follow up?
Viral load 4-8 weeks after switch. Monitor closely their side effects for 3 months.
what type of test do you need before starting miraviroc?
Tropism assay to check whether HIV uses CCR5 or CXCR4 on CD4 cells to enter CD4 cells. Miraviroc only works on CCR5 virus.
What long acting regimen is a common SWITCH, but as of yet, not a recommended initial regimen?
Monthly IM CAB + RPV (Cabenuva)
(it is approved only for already-virologically suppressed PWH)
What are the components of Cabenuva?
CAB + RPV IM Monthly or QOM
Only after switching from their initial regimen to a month of ORAL CAB + RPV, and maintaining viral suppression on that
What are the FDA-approved long-acting ARVs approved for TREATMENT (not PREP)?
Trogarzo (IBA) (IV load, then IV q2 weeks)
Used as salvage therapy/MDR HIV, added on to other agents
Sunlenca (LEN) (SQ abdomen q 6 months (after oral initiation phase))
Used as salvage therapy/MDR HIV, added on to other agents
Cabenuva (CAB/RPV) IM gluteal q 4 or q8 weeks
Used as a good SWITCH option. Only approved for use once PWH has achieved viral suppression on their current regimen.
Switching from 3 drugs to 2 drugs in virologically supressed PWH
OPERA database analysis saw similar Viral suppression between 3 and 2 drug regimens.
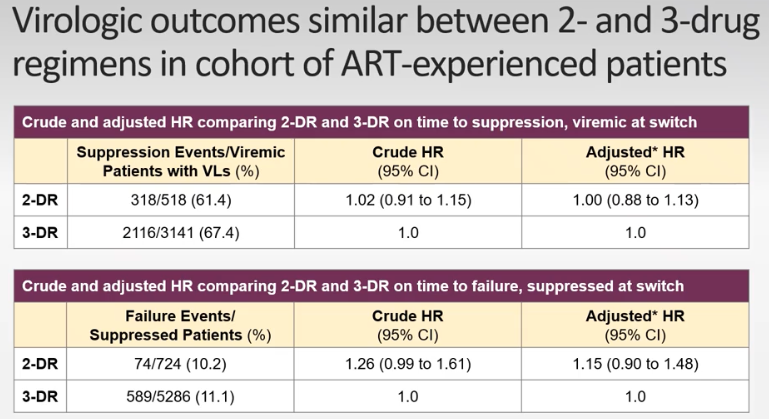
ARTISTRY 1 TRIAL results, switching to 2-drug regimen
switching heavily treatment experienced PWH w VS on a complex regimen to LEN + BIC vs remaining on complex regimen
The ARTISTRY-1 phase 2/3 trial (NCT05502341) found that switching to a once-daily, single-tablet regimen of bictegravir (75 mg) and lenacapavir (50 mg) successfully maintained high rates of HIV-1 suppression (<50 copies/mL) at 48 weeks, showing non-inferiority to complex, multi-tablet regimens. The combination was well-tolerated and improved treatment satisfaction.
(no brand name yet)