Alkene Addition Reactions
1/43
Earn XP
Description and Tags
This flashcard set covers key reactions, mechanisms, regiochemistry, and stereochemistry of alkene addition reactions.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Hydrohalogenation Reaction
Alkene + H-X (where X is Cl, Br, or I) → Alkyl halide
Hydrohalogenation Mechanism
Protonation → carbocation intermediate → nucleophilic attack by X⁻
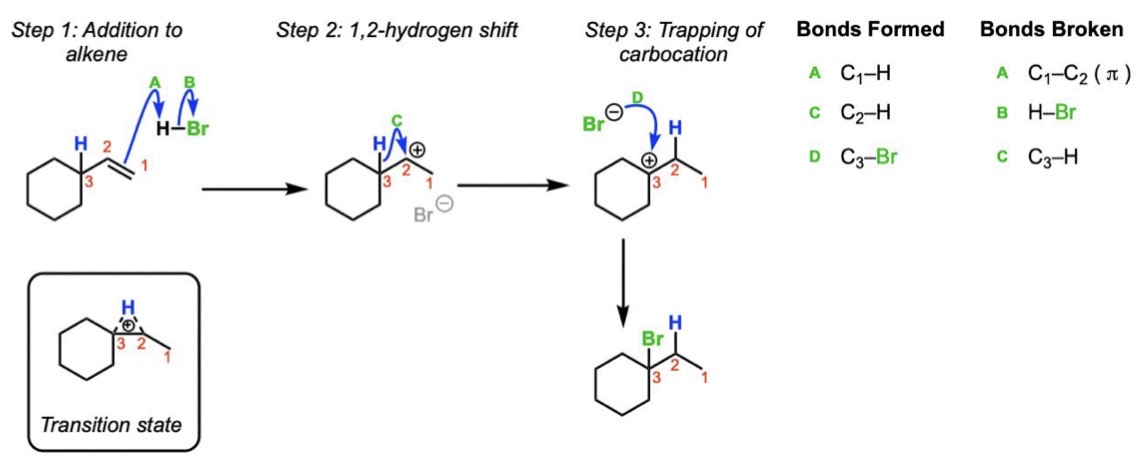
Hydrohalogenation Regiochemistry
Markovnikov (X goes to more substituted carbon)
Hydrohalogenation Stereochemistry
Not stereospecific
Hydrohalogenation (Br and peroxide present) Reaction
Alkene + HBr + ROOR → Alkyl bromide
Hydrohalogenation (Br and peroxide present) Mechanism
Radical chain mechanism (initiation, propagation, termination)
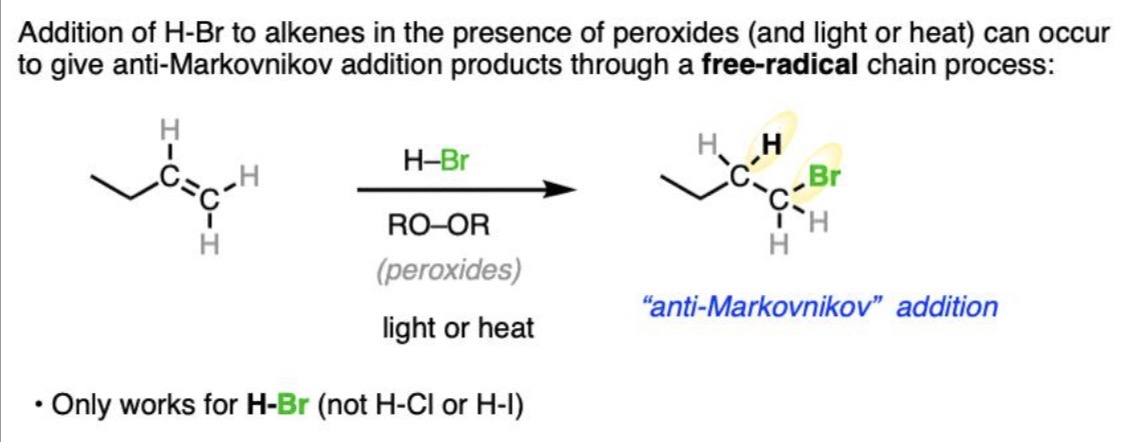
Hydrohalogenation (Br and peroxide present) Regiochemistry
Anti-Markovnikov (Br to less substituted carbon)
Hydrohalogenation (Br and peroxide present) Stereochemistry
Not stereospecific
Acid-Catalyzed Hydration Reaction
Alkene + H₂O (H⁺) → Alcohol
Acid-Catalyzed Hydration Mechanism
Hydration reaction: Carbocation intermediate → nucleophilic attack from H₂O → deprotonation
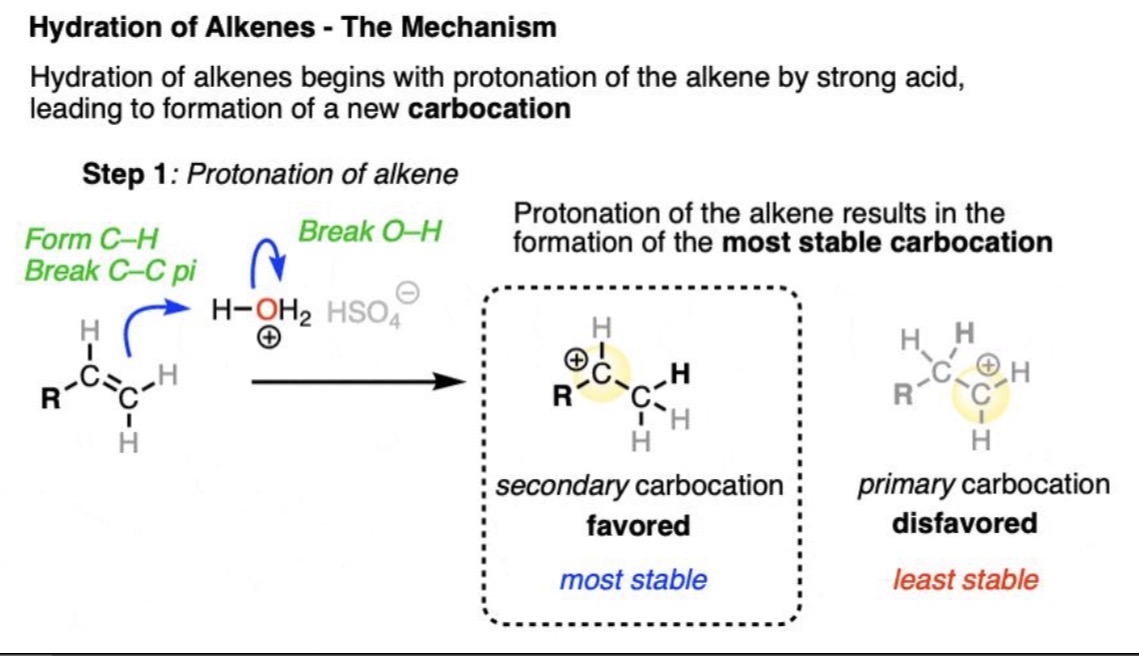
Acid-Catalyzed Hydration Regiochemistry
Markovnikov (X goes to more substituted carbon)
Acid-Catalyzed Hydration Stereochemistry
Not stereospecific
Oxymercuration–Demercuration Reaction
1. Alkene + Hg(OAc)₂, H₂O
2. NaBH₄ → Alcohol
Oxymercuration–Demercuration Mechanism
Hydration reaction without carbocation intermediate (mercurinium ion)
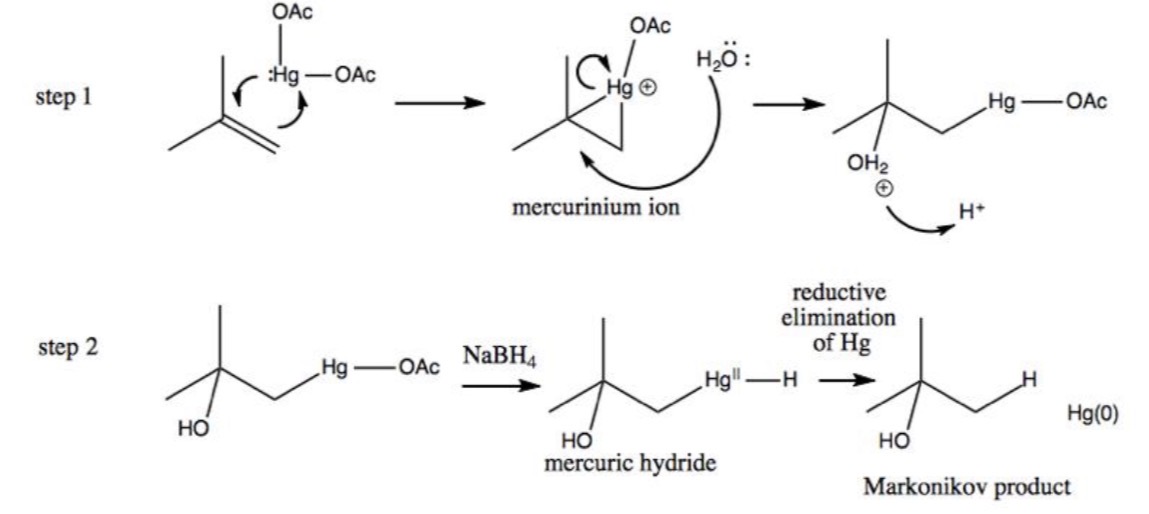
Oxymercuration–Demercuration Regiochemistry
Markovnikov (X goes to more substituted carbon)
Oxymercuration–Demercuration Stereochemistry
Anti addition (two substituents added to opposite sides/faces of double/triple bond)
Hydroboration–Oxidation Reaction
1. Alkene + BH₃•THF
2. H₂O₂, OH⁻ → Alcohol
Hydroboration–Oxidation Mechanism
Concerted (no intermediate carbocation)
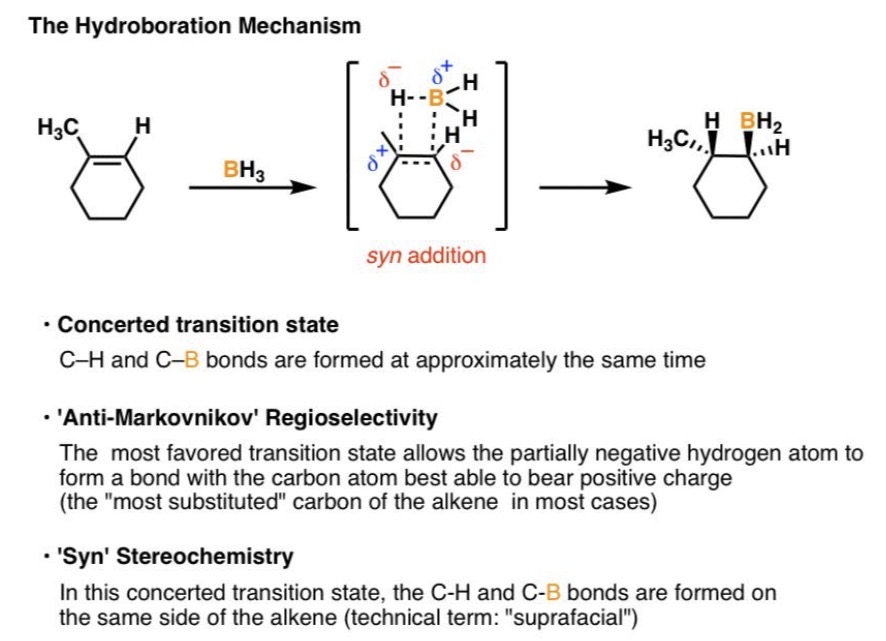
Hydroboration–Oxidation Regiochemistry
Anti-Markovnikov (X to less substituted carbon)
Hydroboration–Oxidation Stereochemistry
Syn addition (two substituents added to same sides/faces of double/triple bond)
Catalytic Hydrogenation Reaction
Alkene + H₂ and heterogenous catalyst (Pd, Pt, or Ni) → Alkane — reduction reaction
Catalytic Hydrogenation Mechanism
Surface reaction on metal catalyst
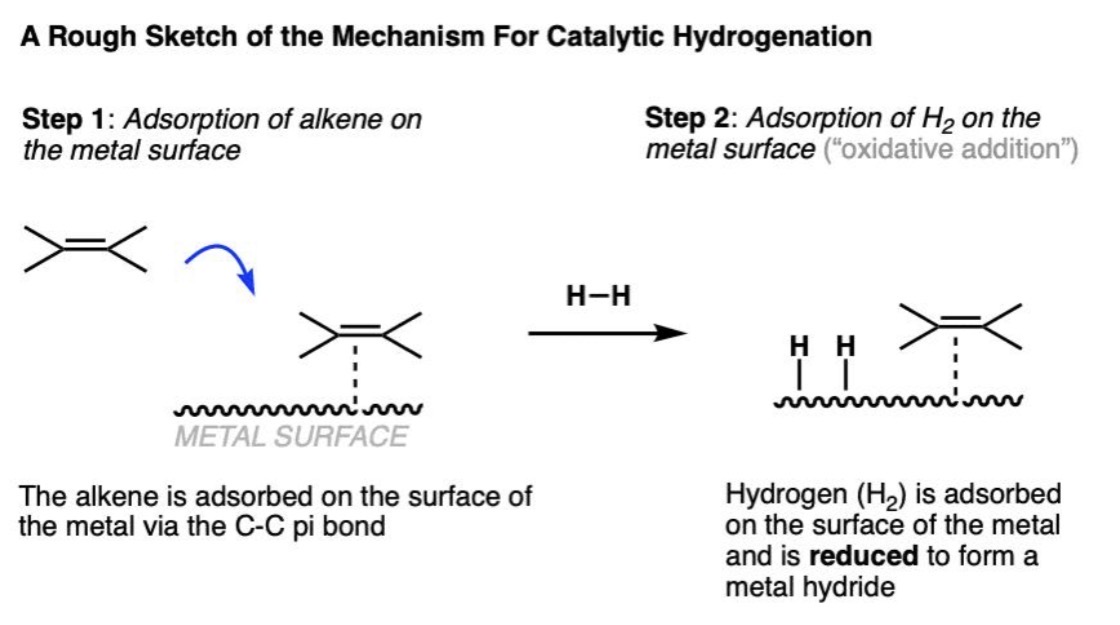
Catalytic Hydrogenation Regiochemistry
Not applicable
Catalytic Hydrogenation Stereochemistry
Syn addition (two substituents added to same sides/faces of double/triple bond)
Halogenation Reaction
Alkene + X₂ (where X is Cl or Br)₂ → Vicinal dihalide
Halogenation Mechanism
Halonium ion intermediate
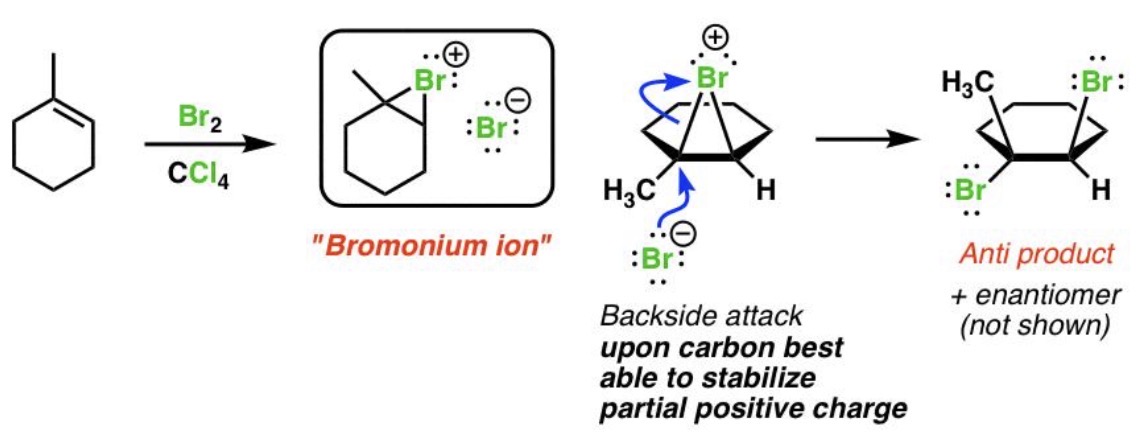
Halogenation Regiochemistry
Not applicable
Halogenation Stereochemistry
Anti addition (two substituents added to opposite sides/faces of double/triple bond)
Halohydrin Formation Reaction
Alkene + X₂ (where X is Cl or Br)₂ + H₂O → Halohydrin
Halohydrin Formation Mechanism
Halonium ion → water attack
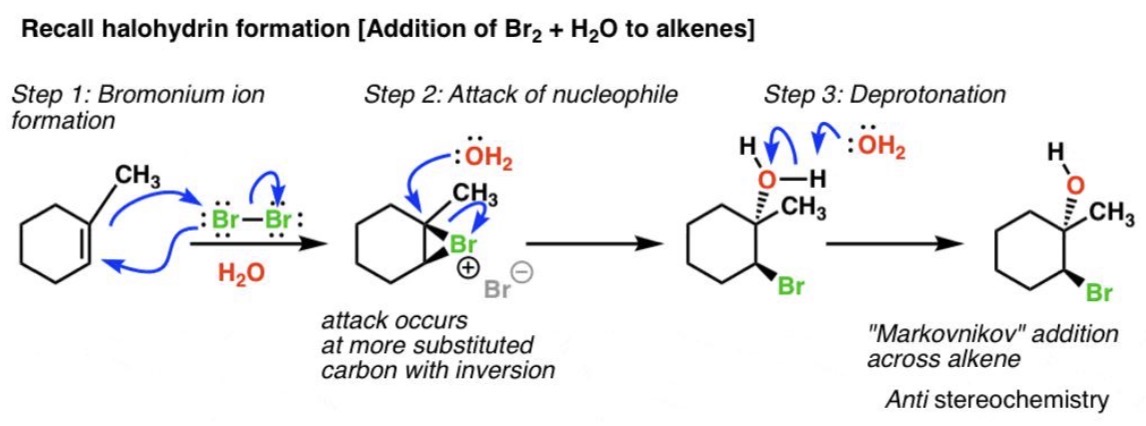
Halohydrin Formation Regiochemistry
OH to more substituted carbon (Markovnikov-like)
Halohydrin Formation Stereochemistry
Anti addition (two substituents added to opposite sides/faces of double/triple bond)
Anti Dihydroxylation Reaction
Alkene → epoxide (peracid) → acid-catalyzed opening → trans diol
Anti Dihydroxylation Mechanism
Epoxide intermediate → backside attack
Anti Dihydroxylation Regiochemistry
Attack at more substituted carbon (acidic conditions)
Anti Dihydroxylation Stereochemistry
Anti addition
Syn Dihydroxylation Reaction
Alkene + OsO₄ (or cold KMnO₄) → cis diol
Syn Dihydroxylation Mechanism
Cyclic intermediate
Syn Dihydroxylation Regiochemistry
Not applicable
Syn Dihydroxylation Stereochemistry
Syn addition
Oxidative Cleavage Reaction
Alkene + O₃ → carbonyls (aldehydes/ketones) (or KMnO₄ → ketones/carboxylic acids)
Oxidative Cleavage Mechanism
Ozonide intermediate → cleavage
Oxidative Cleavage Regiochemistry
Double bond is broken
Oxidative Cleavage Stereochemistry
Not applicable