Unit 1: Chemistry and Structure of Life
1/105
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
science
guided by natural law
testable against the observable world
conclusion are tentative
falsifiable
scientific method
make observations
ask a question
formulate possible answers
make predictions
design and conduct experiment
*cycle of revising hypothesis, asking new questions, changing experiment
comparative experiment
look for differences between multiple groups without manipulating factors
controlled experiment
manipulate one or more factors being tested
dependent variable
response variable (y-axis)
independent variable
manipulated variable (x-axis)
control group
baseline group (usually absense of manipulated variable)
experimental group
manipulated variable
null hypothesis
no real difference between the groups
alternative hypothesis
there is a real difference in the group
statistical significance
determine if differences between groups are significant and unlikely due to chance alone (less than 0.05)
atoms
all matter is composed of atoms
electrons
on outer shell, negatively charged; the number and distribution of electrons dictates interactions in the nucleus
protons
within the nucleus, positively charged; number of protons determine the identity of the element
neutrons
within the nucleus, no charge
chemical reaction
change in the distribution of electrons between atoms
chemical bond
the attractive force that links atoms together
molecule
atoms linked together by bond
covalent bond
shared electrons in orbitals, strong/stable
electronegativity
the attractive force that an atomic nucleus exerts on electrons of another atom
polar covalent bond
unequal sharing of electrons due to differences in electronegativity of the atoms (partial charges); if electronegativity is equal then nonpolar, if electronegativity is different then polar
ionic bonds
highly electronegative atom pulls electron away from an atom with low electronegativity (bonds formed by positive and negative ion), strong
hydrogen bond
electrostatic attraction between slightly positive charged H and slightly negative charged O or N atom, weak
van der waals interactions
temporary dipole causes partial charges that attract, weak but can be substantial when summed
water
all life occurs in water, in order for water molecules to disassociate, the H bonds must be broken
biological consequences of hydrogen bonding between water molecules
liquid water exists at a broad range of temperatures
internal temperatures are able to remain stable because of the large amount of water in living tissues
ice floats (less dense because hydrogen bonding pushes away each other)
aqueous solution
the environment for biological reactions, bonds aren’t broken/formed but instead distributed through solvent
hydrophilic
dissolve easily in water, polar or charged ionic
hydrophobic
don’t dissolve easily in water, nonpolar
acid
pH is high, release H+ ions, H+ concentration high
bases
low pH, accepts H+, low H+ concentration
neutral
pH value = 7, H+ concentration = 10-7
macromolecules
polymers of smaller molecules called monomers joined together by covalent bonds
lipid
NOT covalently bonded: ester bond, non polar hydrocarbons (lots of hydrogen and carbons), hydrophoic
triglycerides
glycerol + 3 fatty acid molecules, condensation reaction to form ester bonds
phospholipids
glycerol + 2 fatty acids + phosphate containing compound (chlorine and phosphate), hydrophilic head, hydrophobic tail, amphipathic
saturated
no double bonds in the tail, usually solid at room temperature
unsaturated
double bonds within the fatty acid tail which causes bends or “kinks” in the chain, usually liquid at room temperature
amphipathic
a molecules with opposing chemical properties (polar and nonpolar sections)
carbohydrates
CmH2nOn, source of stored energy, used to transport stored energy, carbon skeleton for many other molecules, extracellular structures
monosaccharides
simple sugar (hexoses: 6 carbon or pentoses: 5 carbon)
disaccharides
two simple sugars linked by glycosidic bond (alpha 1,2)
oligosaccharides
3 to 20 monosaccharides, bind together in condensation reaction forming glycosidic linkages
polysaccharides
hundreds or thousands of monosaccharides
cellulose
starch
glycogen
cellulose
very stable, good for structural components (plants), linear
starch
storage of glucose in plants, branched
glycogen
storage of glucose in animals, highly branced
nucleic acid
polymers specialized for the storage, transmission, and expression of genetic information
DNA
deoxyribonucleic acid, ATGC, double stranded helix with antiparallel strands; A-T: 2 hydrogen bonds, G-C: 3 hydrogen bonds
RNA
ribonucleic acid, AUGC, single stranded, can have base pairings in some reasons
nucleotides
monomers of nucleic acid, linked by phosphodiester bonds as two phosphate groups are removed, added to 3’ end, grow in 5’-3’ direction
pentose sugar
phosphate group
nitrogen containing base
pentose sugar
ribose for RNA [OH], deoxyribose for DNA [H]
proteins
function directly related to 3D shapes (structure), the shape is determined by the sequence and chemical properties of the monomers
amino acids
monomers of protein
includes: alpha carbon, carboxyl group, amino group, R group (R group varies)
amino acids bond together in a condensation reaction that forms peptide linkages
can only add monomers onto the C terminus (end) and not the N terminus (beginning)
amino acids with electrically charged hydrophilic side chains
hydrophilic, polar molecules, can form ionic bonds with amino acids of opposite charge (charges in the R group)
amino acids with polar but uncharged side chains
hydrophilic, polar molecules, can form hydrogen bonds (polar functional groups and polar covalent bonds)
amino acids with nonpolar hydrophobic side chains
hydrophobic, nonpolar molecules, cluster together in aqueous environments (CH bonds, nonpolar covalent bonds)
special cases
cysteine, glycine, proline
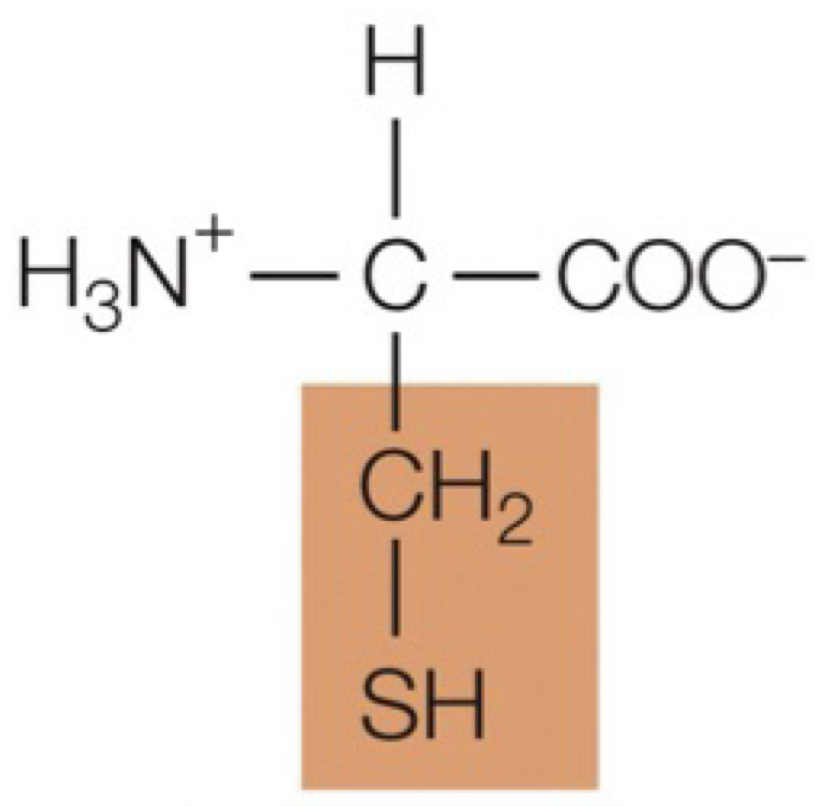
cysteine
forms disulfide bridges
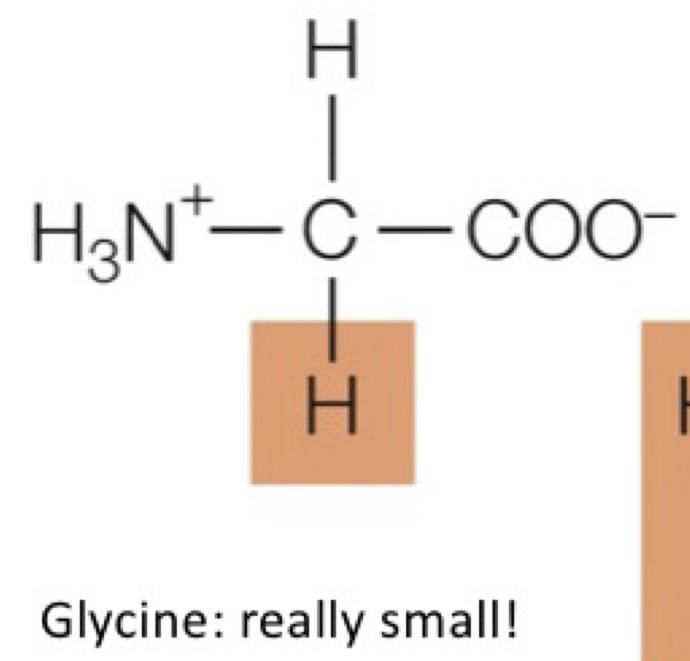
glycine
really small
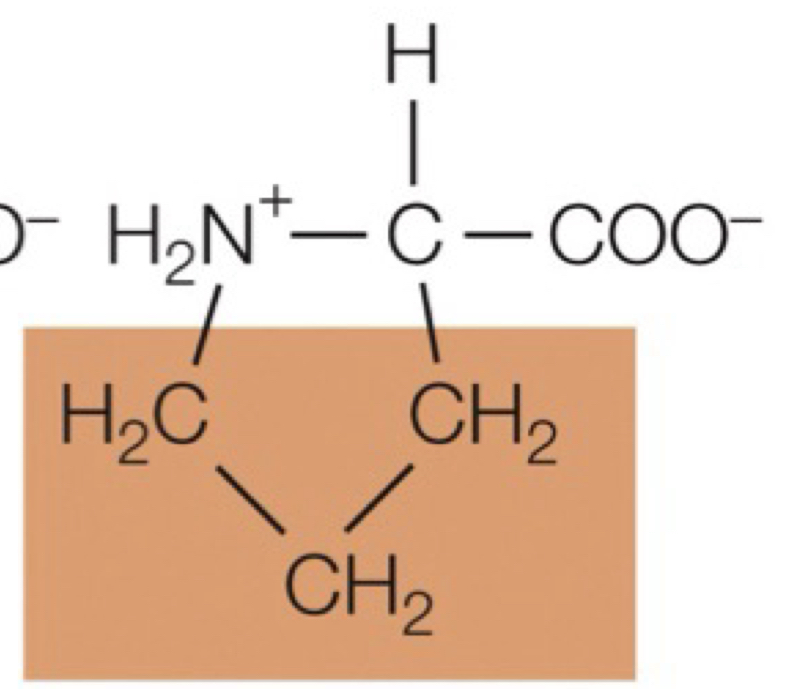
proline
really bulky
condensation reaction
when two or more monomers join to form a polymer with the loss of water molecules
primary structure
polypeptide: one strong of amino acids
secondary structure
alpha helices and beta pleated sheets, hydrogen bonds form between the chains within the amino and carboxyl groups (within the backbone NOT the r group)
tertiary structure
bending and folding results in a macromolecule with specific 3D shapes (between R groups), disulfide bridges, hydrogen bond, van der waals interactions, ionic interactions
quaternary structure
results from the interaction of subunits by hydrophobic interactions, van der waals, ionic attractions, and hydrogen bonds; each subunit has its own unique tertiary structure
cell membrane
separates interior of the cell from its environment, van der waals and hydrophobic interactions maintain lipid bilayer, separation of two aqueous environments
fluid mosaic model
many different components can move freely
membrane contains proteins
integral protein, transmembrane protein, peripheral protein, anchored
integral protein
embedded in the lipid bilayer, exposed hydrophilic and hydrophobic regions
transmembrane protein
integral protein that spans through the membrane hydrophilic and hydrophobic domain
peripheral protein
not embedded, but interact with phospholipid heads, polar or charged regions
anchored protein
covalently attached to lipids, which intserts into the bilayer
selective permeability
some substances can pass through, but some can’t
diffusion
the process of random movement toward equilibrium, the net movement from regions of greater concentration to regions of lesser concentration, net movement is directional till equilibrium is reached
osmosis
the diffusion of water across membrane; water will enter the thing that has a higher concentration and will leave those with lower concentration
hypertonic
isotonic
hypotonic
hypertonic
higher solute concentration
isotonic
equal solute concentration
hypotonic
lower solute concentration
passive transport
no outside energy required (diffusion)
simple diffusion
facilitated diffusion
simple diffusion
small, nonpolar, uncharged, hydrophobic molecules can pass through the membrane (rate of movement vary)
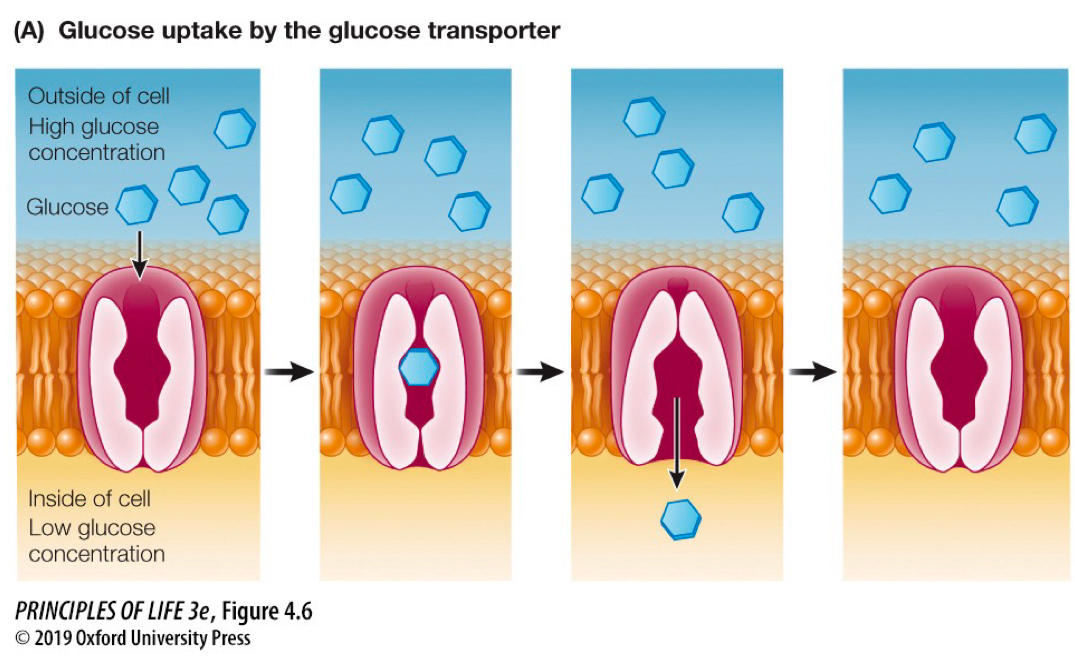
facilitated diffusion
the passive movement of polar/larger molecules through integral protein
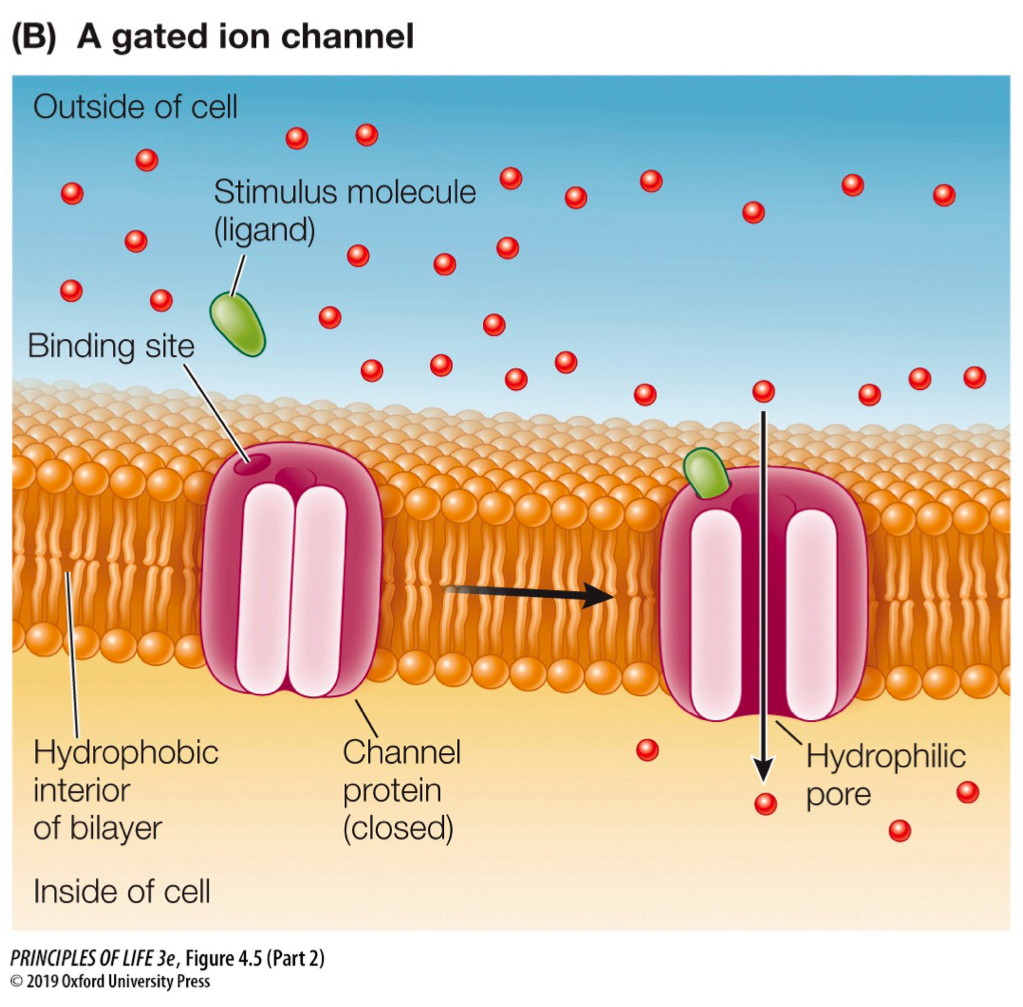
channel protein
carrier protein
channel protein
integral transmembrane protein that form a channel
ex) ion channels: most are gated, opened by stimulated to change shape by chemical signal (ligand) or an electrical charge difference (voltage-gated)
carrier protein
integral transmembrane proteins that bind some substances and speed their diffusion through the bilayer
active transport
moves substances against a concentration and/or electrical gradient so it requires energy (energy source is ATP)
movement of molecules
passive transport, active transport
sodium-potassium pumps
moves 2 difference molecules against their concentration gradient using ATP
pushes 2 ions inside (K+)
brings in 3 ions outside (Na+)
vesicle
the plasma membrane folds around the material or separates from internal membrane and pinches off (vesicle is phospholipid layer so forges with membrane)
used to transport large molecules across the membrane
exocytosis
material in vesicle is expelled from a cell
ex) waste products, secreted proteinsm, digestive enzymes, neurotransmitter
endocytosis
brings materials into a eukaryotic cell
ex) large particles, fluids or dissolved substances, specific large molecule
prokaryotic cells (bacteria and archaea)
plasma membrane and most of them have cell walls , no membrane-bound nucleus or other organelles
eukaryotic (plants, animals)
membrane-bound nucleus and other organelles, some have cell walls
cell wall (extracellular support)
provides structural support and protection; plants, archaea, bacteria, fungi, some protists
extracellular matrix
holds cells in tissue, contributes to the physical properties of cartilage, skin, etc., filter materials, orients cell movements; animal cells
cytoskeleton
internally supports and maintains cell shape, holds organelles in position, moves organelles if needed, interacts with extracellular structures to hold cells in place, made from different protein and acts like a cell in place
organelles
allow an internal separation of functions within a single cell
cytoplasm
the material within the cell (except the nucleus), nucleotide region contains DNA
nucleus
largest organelle, contains DNA, site of DNA replication, site of gene expression, surrounded by nuclear membranes
endomembrane system
network of interconnected membranes in the cytoplasm, vesicles shuttle substances between the various components
includes: nuclear envelope, endoplasmic reticulum (smooth, rough), golgi apparatus
nuclear envelope
surrounds nucleus, pores control the movement of molecules across the envelope