BI108 Carbohydrates & Lipids
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Macromolecules
molecules that make up living organisms
Functional groups:
groups of atoms with particular properties
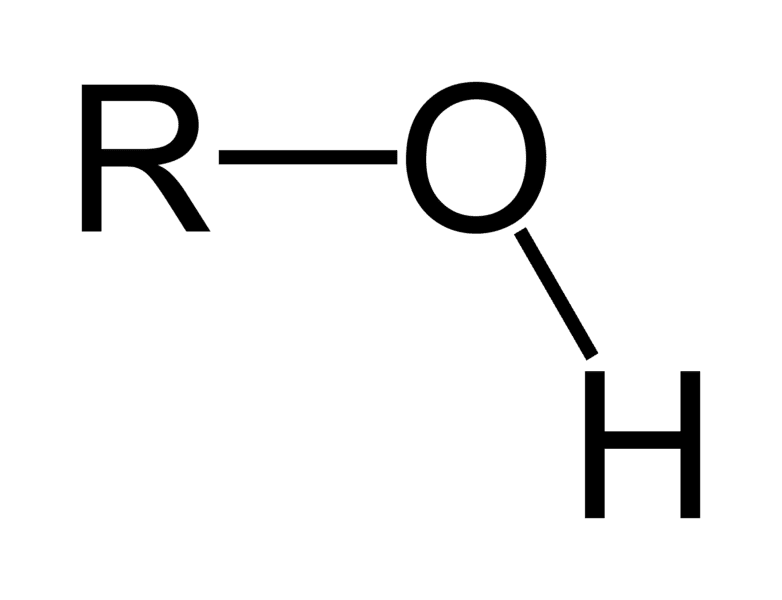
Hydroxyl group
polar
Methyl group
nonpolar
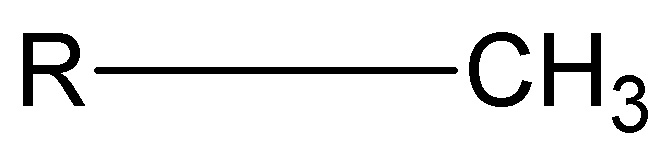
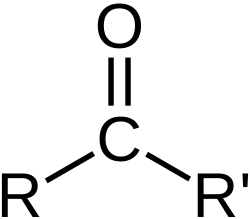
Carbonyl group
polar
Sulfhydryl group
polar

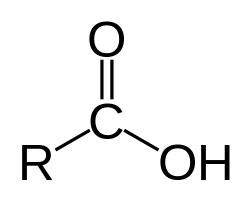
Carboxyl group
charged
ionizes to release H+
acidic
Amino group
charged
accepts H+ to form NH3+
basic
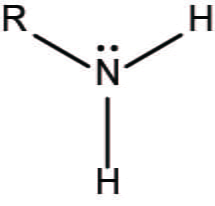
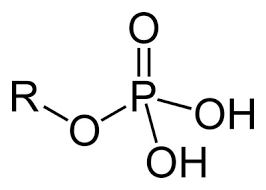
Phosphate group
charged
ionizes to release H++
acidic
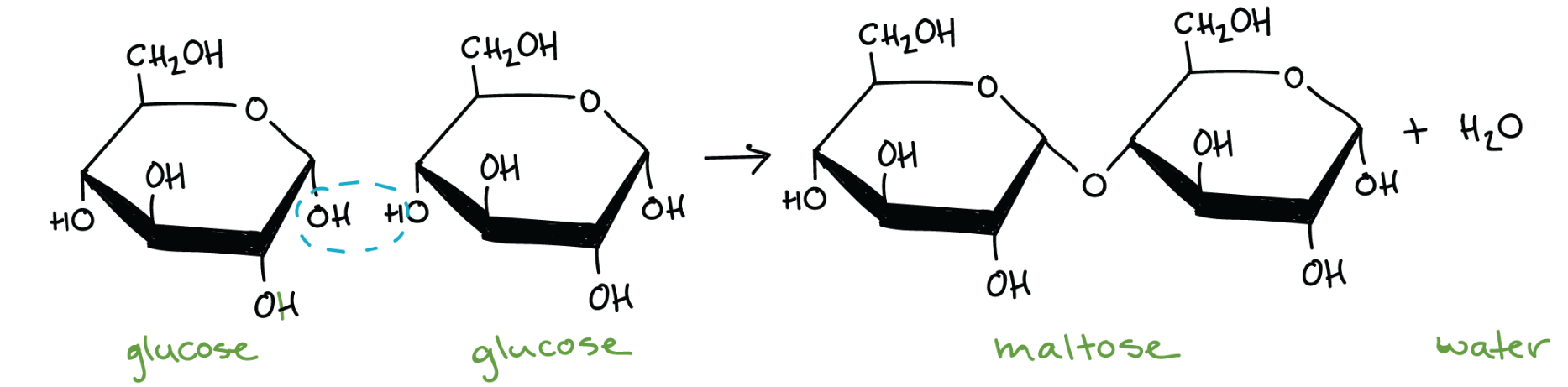
Condensation reaction (dehydration synthesis)
removing water and making a bond
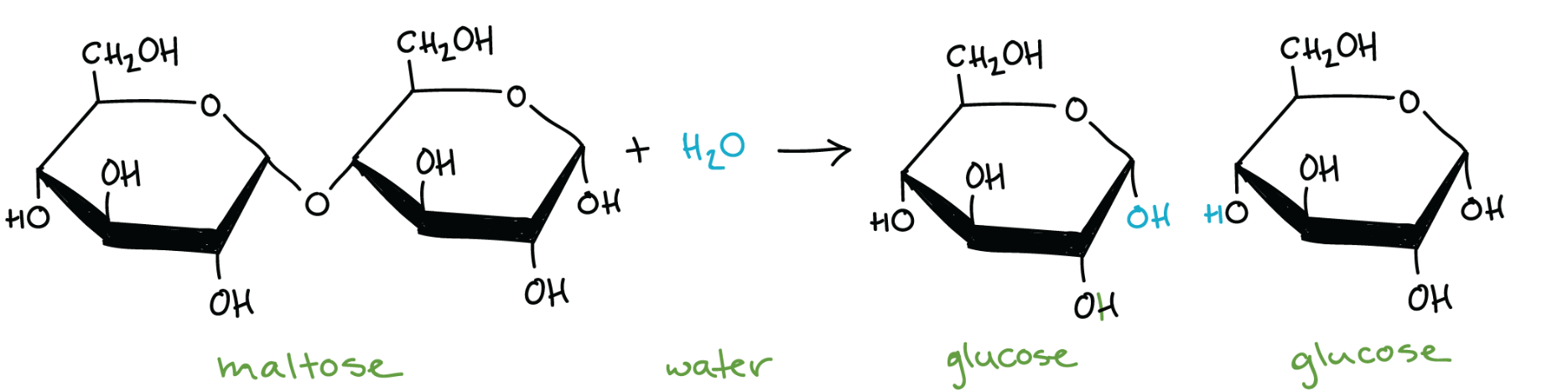
Hydrolysis reaction
inputting water and breaking a bond
Carbohydrate
source of stored energy within plants and animals
Monosaccharide
simple sugars; “one sugar”
Disaccharides
“two sugars” linked by covalent bonds
Oligosaccharides
medium-sized polymers; 3-20 monosaccharides
Polysaccharides
“many sugars”; hundreds or thousands of monosaccharides
Glycosidic linkages
a covalent bond that connects a carbohydrate molecule to another molecule; formed by condensation reactions (H2O is a product)
Alpha (ɑ) glycosidic linkages
Bent, branched
Relatively easy to break
Glycogen
storage of glucose in animals
Starch
storage of glucose in plants
Beta (β) glycosidic linkages
Linear
Hydrogen bonding makes it relatively hard to break
Cellulose
very stable, good for structural components, found in plants
Chitin
structural support and protection that forms the exoskeleton of arthropods (insects, crustaceans), the cell walls of fungi, and various hard structures in invertebrates
Glycan
polymers of simple sugars bonded with glycosidic linkages
Hydrocarbon chains
chains that consist of hydrogen and carbon atoms
Fats
important in energy processing and storage
Glycerol
has three hydroxyl groups, serving as the backbone of triglycerides
Fatty acid
nonpolar hydrocarbon with a polar carboxyl group (-COOH)
Saturated fatty acid
when all C-C bonds are single bonds; long straight chain
Unsaturated fatty acid
contains C=C bonds that are double bonds; introduces bends in the structure
Ester bond
when carboxyls interact with hydroxyls of glycerol
Steroids
a lipid containing carbon atoms arranged in four “fused rings”
Phospholipids
phosphate-containing compound, glycerol, 2 fatty acid chains
hydrophilic head
hydrophobic tail
Amphipathic molecules
molecules that have hydrophilic and hydrophobic regions; opposing chemical properties