Chem- Exam 2
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
titration
a quantititative method to determine an acidic or basic solution’s concentration using the neurtralization reaction (acid + base —> salt + water)
buffer solution
a solution that is able to resist large changes in pH when an acid or base is added
our ____ is a buffer system keeping a pH ~ ____
blood; 7.4
buffer consists of either
a weak acid and its conjugate base (strong)
or
weak base and its conjugate acid (strong)
choose the pair that will make a buffer
A.HCl and NaCl
B.H3PO4 and Na2HPO4
C.H2SO4 and Na2SO4
D.H2CO3 and NaHCO3
D.
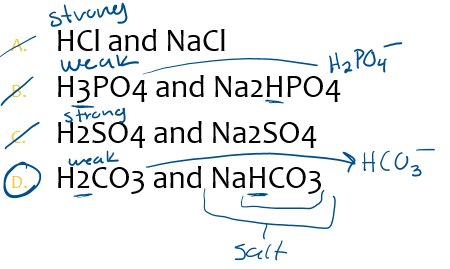
buffer capacity
measure of the ability of the buffer to resist pH changes
Ka
acid dissociated constant (like Keq but for specific reaction of a weak acid in water)
blood pH~7.4 is maintains partly by a _____ buffer system
carbonic acid-bicarbonate
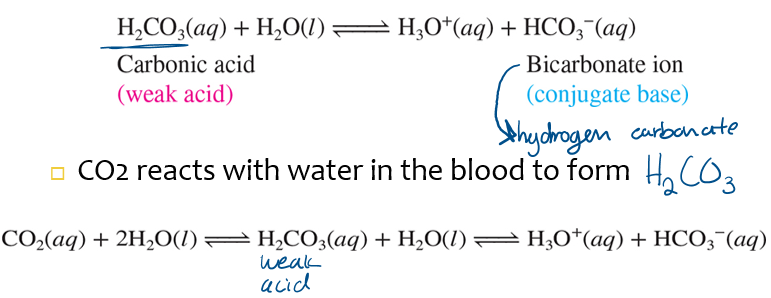
high CO2 shifts the equilibrium to the _____; blood becomes ____
right’ acidic (H3O+)
low CO2 shifts the equilibrium to the _____; blood becomes ____
left; basic
Oxidation Reduction (redox) reaction
reaction in which electrons are transferred from one species to another; OIL RIG
Which is reduced?
Fe(s) + Co2+(aq) à Fe2+(aq) + Co(s)
A.Fe(s)
B.Co2+(aq)
C.neither
B. Co2+(aq)
oxidizing agent
is reduced, gains electrons, causes oxidation
reducing agent
is oxidized, loses electrons, causes reduction
What is the oxidizing agent in the following reaction?
4 Fe + 3O2 -->2 Fe2O3
A.Fe
B.O2
C.Fe2O3
D.Fe3+
E.O2-
B. O2
How many electrons are transferred in the following reaction?
4 Fe + 3O2 -->2 Fe2O3
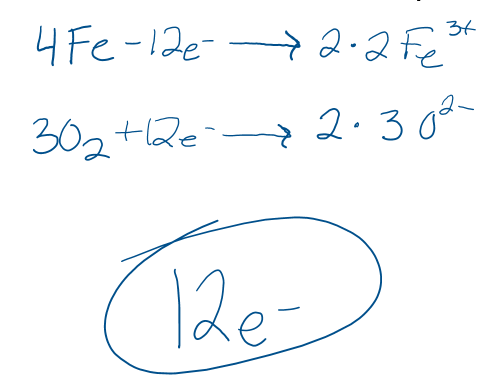
organic molecules contain _____
carbon, hydrogen, may also have O,S,N,P, or halogens
saturated hydrocarbon
hydrocarbon has only C—C bonds that are single (no double bonds), and max amount of hydrogen filling every C
unsaturated hydrocarbons
hydrocarbons with at least 1 C--C double bond or a C—C triple bond
alcohol
contain the hydroxyl group (OH-) [R—OH]
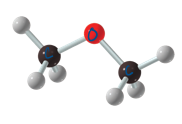
ether
contais oxygen between 2 carbon groups
thiol
contains a sulfhydryl group (R—SH)
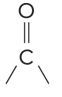
aldehydes
have a cobonyl group, must have at least 1 hydrogen bonded to the C of the carbonyl
carbonyl gorup
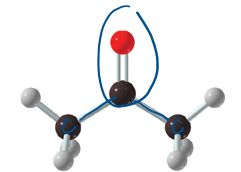
ketones
carbonyl group, only carbon that are bonded to carbonyl C
amines
based off of ammonia, if you replace at least 1 H with an R-group (C-group)

alkanes
saturated hydrocarbons (109.5o bond angles)
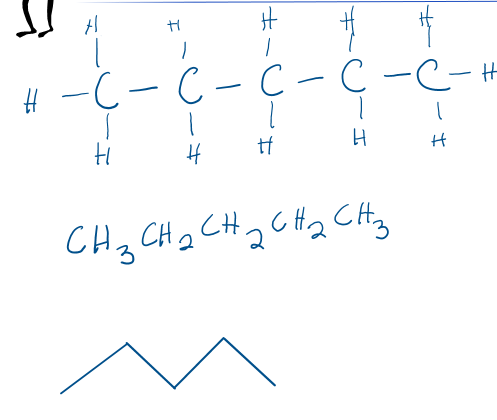
Name the straight chain alkane C5H12
A.Butane
B.Pentane
C.Octane
D.Nonane
B. Pentane
Draw C5H12
constitutional isomers
same molecular formulas (C4H10) but different structural formulas (how carbons bond)
substituents: halogens and akyl groups
F- fluoro
Cl- chloro
Br- bromo
I- iodo
—CH3 methyl
—CH2CH3 ethyl
—CH2CH2CH3 propyl
—CH2CH2CH2CH3 butyl
—CH2CH2CH2CH2CH3 pentyl
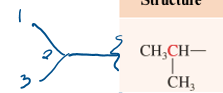
isoproyl
3 carbon substituent attached to 2nd carbon
sec-butyl
“second”, 4 carbon substituent attached to 2nd carbon
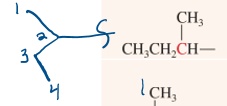
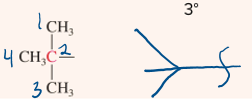
t-butyl or tert-butyl
4 carbon substituent that froms a T or chicken foot shape
hydrocarbons are… (polarity, solubility in water and organic solvents)
nonpolar, insoluble in water, soluble in organic solvents
since there are only ____ forces bewteen hydrocarbons, alkanes tend to have relativley ____ MP and BP
dispersion forces, low
branched alkanes have a ____ MP and BP than the straight chain isomer
lower
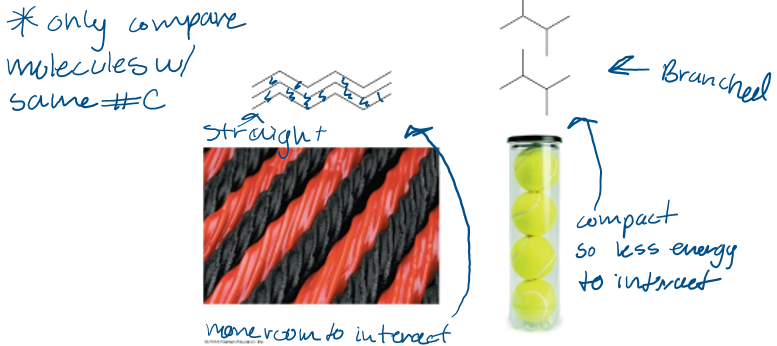
Which has the highest mp?
A.Pentane
B.2-methylbutane
C.2,2-dimethylpropane
D.Cannot be determined
A. Pentane
cycloalkanes
carbon rings with 2 fewer H than straight-chain alkanes with same number of C; name using prefix of cyclo-
steroisomers
molecules with the same structural form but different arrangment in space (of substituents)
cis- stereoisomer
same side of ring
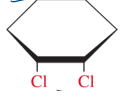
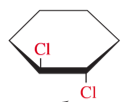
tans- steroisomers
opposite side of ring
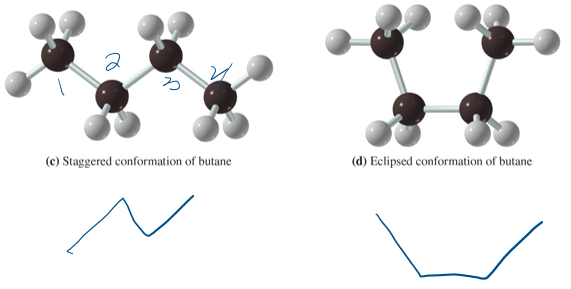
conformations of alkines
structure that can be converted to one anotehr by rotaion around a bond (same molecules)
combustion
hydrocarbon + O2 —> CO2 + H2O
release a lot of energy
balancing combustion reactions
1.Write the equation for the reactions
2.Balance elements that appear only once in the reactants and products
3.Balance the remaining elements
4.Make sure all coefficients are whole numbers
halogenation
reaction in which a H is replaced with a halogen
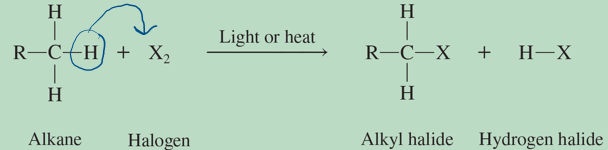
substitution reaction
reations where one group replaces another
elkenes
include double bonds between C (120o bond angles)
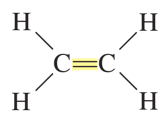
alkyne
contains triple bonds between C (180o bond angles)
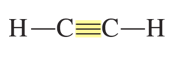
strongest IMF force between alkene and alkyne molecules are _____ so MP and BP are ____ and tend to ____ as MM increases
dispersion forces; low; increase
Which has the highest mp?
A.Ethene
B.Propene
C.1-butene
D.1-pentene
D.1-pentene
geometric isomers of alkenes
cis- same side of double bond; trans- opposite sides of double bond
addition reactions
reaction in which 2 molecules add together
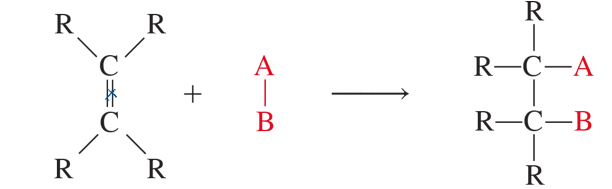
hydrogenation
addition of H2
halogenation
addition of a halogen
hydration
addition of a H2O; H add to 1 C of a double bond, OH added to the other double bond
hydrohalogenation
addition of a halogen and H (HX) coumpound (ex. HBr, HCl); breaks the double bonds and add 1 H to each C
markovnikov’s rule: in assymmetrical alkenes the major product is formed when ___________
the H adds to C double bond with more H’s
aromatic hydrocarbons
hydrocarbons that are more chemically stable than their formula would predict
molecules based off of benzene
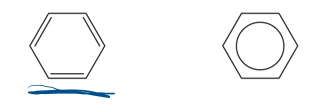
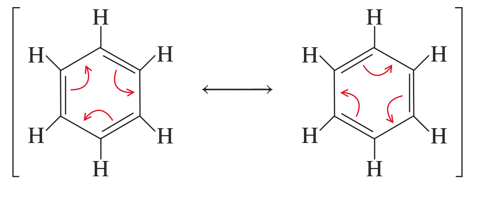
benzene
its 6 C atoms share 6 extra electrons, very stable
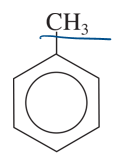
toluene
another name for methylbenzene
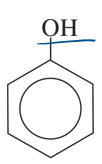
phenol
benzene with OH group
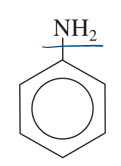
aniline
benzene with NH2
benzene is unusually ____ (stability); doesn’t undergo _____ reaction like alkenes, but can undergo _____ reactions like alkanes
unusually stable; no addition reaction; yes substitutions
heterocyclic aromatic compounds
have an atom other than C in the ring
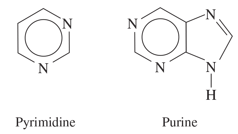
classification of alcohols
classified based on number of C bonded to the C bonded to the —OH group
primary (1o)- 1 C attached to C bonded to OH
secondary (2o)- 2 C attached to C bonded to OH
tertiary (3o)- 3 C attached to C bonded to OH
Which is true for alcohols?
A.They are nonpolar
B.They are polar
B.They are polar
solubility _____ as the length of the nonpolar carbon chain increases in an alcohol
decreases
Which is the strongest IMF present between alcohol molecules?
A.Dispersion
B.Dipole-dipole
C.H-bonds
D.Ionic
C.H-bonds
Alcohols will have _____ mp/bp than alkanes.
A.lower
B.the same
C.higher
C. higher
method for making alcohols
hydration (add H2O to a double bond), or reduction of aldehydes and ketones (add H2 and reduce number of C-O bonds)
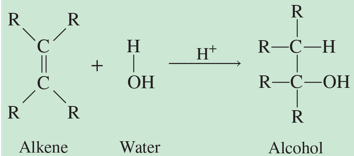
oxidation reactions
increase number of C-O bonds, decrease number of H;
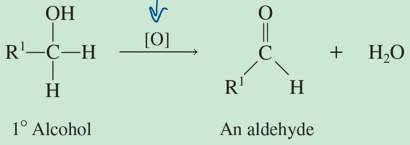
oxidation reaction: if there isn’t a H off the C attached to OH group to lose, the reaction doesn’t proceed
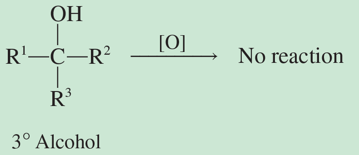
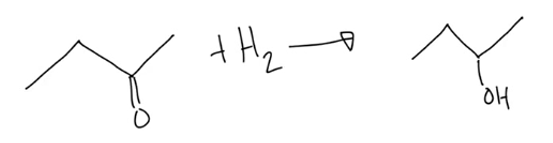
What type of reaction is below?
A.Combustion
B.Reduction
C.Oxidation
D.Hydration
B. reduction
phenol
hydroxul group on benzene, very stable
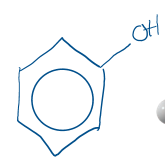
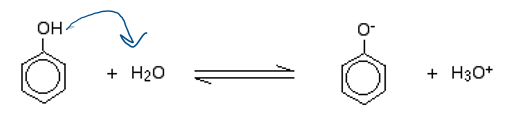
phenols as weak acids
slighlty soluble in water, nonpolar ring and OH balance, can dissociate in water (donate H)
ether
R—O—R (R represents C group)
Which is the strongest IMF present between ether molecules?
A.Dispersion
B.Dipole-dipole
C.H-bonds
D.Ionic
b. dipole-dipole
Which statement is true?
A.Ethers of low MW should be soluble in water
B.Ethers of high MW should be soluble in water
C.Ethers should not be soluble in water
A.Ethers of low MW should be soluble in water
thiols
contain the sulfhydrul group (—SH)