chem pack 1 passage 8
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
Passage 8 (Questions 43–46)
Recent work suggests that nitrogen can be used to store and supply energy in a manner similar to that of gasoline and ethanol. In the atmosphere, nitrogen is diatomic and contains a triple bond. Theory suggests that, under sufficient pressure, nitrogen can be converted into a solid form with the structure shown in Figure 1; it resembles the structure of phosphorus.
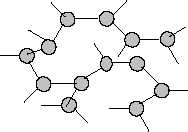
Figure 1 | Structure of a solid form of nitrogen |
Theories and experiments dealing with elements subjected to high pressures reveal a trend in which extreme pressures applied to an elemental solid cause it to assume the structure and bonding characteristics of a heavier element in the same column of the periodic table.
At normal atmospheric pressure, solid nitrogen will be metastable and return to the gaseous form, releasing energy as it returns. It is expected to have an energy density (joules•cm–3) equal to or greater than hydrocarbon fuels and greater than methanol (CH3OH) and other alcohols.
In the absence of an energy barrier, the solid form of nitrogen is predicted to be stable above 65 GPa pressure (1 atm = 1 × 105 Pa), with the diatomic form stable below that pressure. However, to overcome energy barriers, additional pressure must be applied to convert the gaseous form into the solid form.
The structure and bonding of diamond, which is formed from graphite at extreme pressures, should be similar to that of elemental:
A
aluminum and gallium.
B
silicon and germanium.
C
phosphorus and arsenic.
D
sulfur and selenium.
Solution: The correct answer is B.
The passage states that at extreme pressures an elemental solid assumes the structure and bonding characteristics of a heavier element in the same column of the periodic table. The structure and bonding of diamond, which is a form of carbon, would therefore be most like other elements in the same group of the periodic table as carbon. This group contains silicon and germanium. Thus, B is the best answer.
What bonding accounts for the expected increase in energy density of solid nitrogen as compared to methanol?
A
Solid nitrogen contains covalent and ionic bonds; methanol contains only weak ionic bonds.
B
Solid nitrogen contains covalent and ionic bonds; methanol has covalent bonds within each molecule and weak van der Waal’s interactions between molecules.
C
Solid nitrogen contains only covalent bonds; methanol contains only weak ionic bonds.
D
Solid nitrogen contains only covalent bonds; methanol has covalent bonds within each molecule and weak intermolecular interactions.
Solution: The correct answer is D.
Solid nitrogen contains only covalent bonds. Methanol contains only covalent bonds within each molecule but is capable of intermolecular hydrogen bonding because of the –OH group. Thus, D is the best answer.
Hydrogen bonding is strong, relative to other intermolecular forces such as London dispersion, etc.
But it is still quite weak compared to a typical covalent bond.
It's all relative to your reference frame.
A covalent bond is a chemical bond formed when two atoms share one or more pairs of electrons in order to achieve a stable electron configuratio
What prediction can be made about the entropy change during the proposed synthesis of solid nitrogen?
A
∆S is positive, entropy decreases.
B
∆S is positive, entropy increases.
C
∆S is negative, entropy decreases.
D
∆S is negative, entropy increases.
Solution: The correct answer is C.
In the synthesis of the more ordered solid nitrogen, the entropy of the system decreases and therefore ΔS is negative. Thus, C is the best answer.
Would deviations from the ideal gas law be observed for gaseous nitrogen at 180 GPa and room temperature?
A
No, because both temperature and pressure must increase before such deviations are observed
B
No, because nitrogen molecules are symmetrical and do not interact with each other
C
Yes, because at this pressure, molecular volumes and intermolecular forces become significant
D
Yes, because at room temperature, molecular volumes and intermolecular forces become significant
Solution: The correct answer is C.
According to the passage, the solid form of nitrogen is predicted to be stable above 65 GPa. At 180 GPa, deviations from the ideal gas law would be observed for gaseous nitrogen. The ideal gas law assumes that molecular volumes and intermolecular attractive forces are negligible. At a pressure at which solid nitrogen is predicted to be stable, neither of these assumptions would be valid. Thus, C is the best answer.
such high pressures allow for formation of solid nitrogen which are an indication of significant intermolecular reactions, thus a deviation of the ideal gas law
The explanation isn't the greatest because high pressures or low temps force molecules close together and cause deviations from the ideal gas law even when nitrogen isn't solid.
The gas particles themselves occupy a negligible volume compared to the total volume of the container.
There are no intermolecular forces (attraction or repulsion) between gas particles.
Collisions are perfectly elastic
The ideal gas law relies on two primary assumptions: gas particles occupy negligible volume and there are no intermolecular forces between them. However, at extreme pressures like 180 GPa, these assumptions fail:
Molecular Volume: High pressure forces molecules so close together that the actual space they occupy becomes a significant fraction of the total volume.
Intermolecular Forces: At such proximity, attractive and repulsive forces between molecules are no longer negligible and significantly impact the gas behavior.
Nitrogen specifically is predicted to transition into a solid phase above 65 GPa, further indicating that its behavior at 180 GPa deviates drastically from a simple ideal gas.