4.23-4.28 Alkenes
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
General formula for alkenes
CnH2n
Why are alkenes unsaturated hydrocarbons
They contain one or more C=C double bonds - they have less than the max number of hydrogen atoms
What is the molecular and structural and displayed formula of ethene
C2H4
CH2CH2
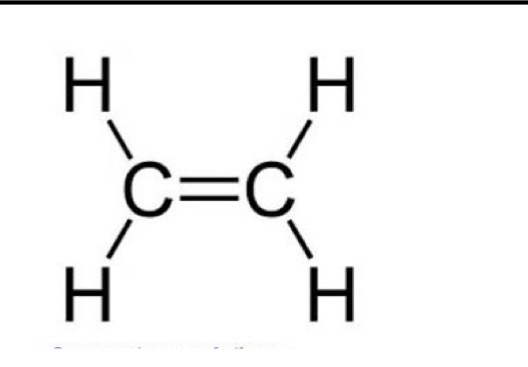
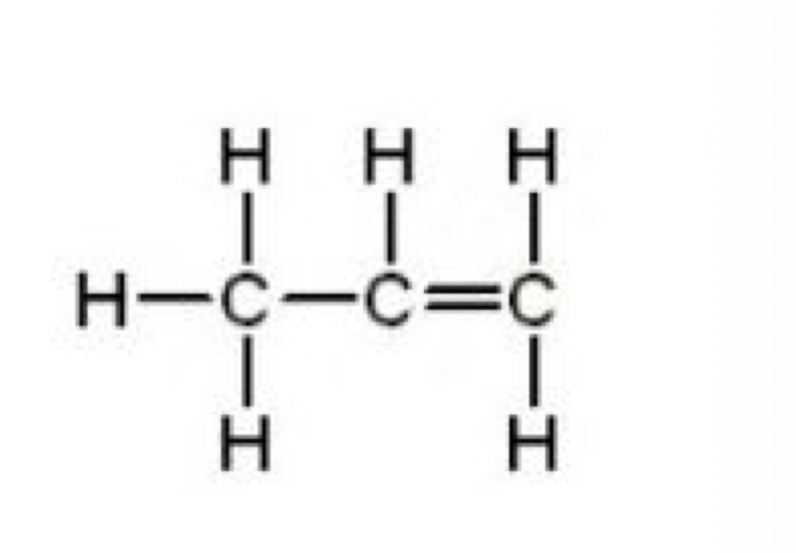
What is the molecular, structural and displayed formula for propene
C3H6
CH3CHCHCH3
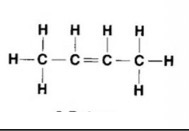
What is the molecular, structural and displayed formula for butene
C4C8
CH3CHCHCH3
What type reaction do alkenes undergo with a halogen?
Addition reaction in which atoms of a simple molecule add across the C=C double bond.
What is produced from bromine and alkene reacted?
A dibromoalkane
Draw the addition of bromine to ethene
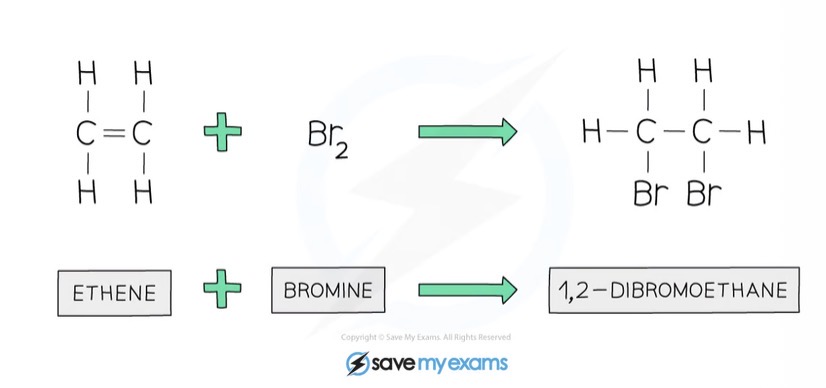
How to test for alkenes
Alkenes react with bromine water - turning from orange to colourless
Alkanes do not react - remains orange (because already fully saturated)