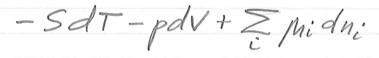Stat Thermo Exam 1
1/123
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
124 Terms
in a microcanonical ensemble BLANK stays constant across system copies
Energy, Volume, Number of particles
In a microcanonical ensemble, system copies M are not necessarily in the same
state
In a canonical ensemble BLANK stays constant across system copies, but BLANK is exchanged
Volume and Number of particles, Energy
When energy is exchanged in a canonical ensemble, this is called
fluctuation
In a grand canonical ensemble BLANK is constant across copies of the system, but BLANK is exchanged
Volume, Energy and Number of particles/mass
The statistical average of a thermodynamic dependent ensemble variable is determined by
the sum of the variable across all copies, divided by the total number of copies M
Equal apriori probability states that
all states with the same energy are equally likely, and in a closed system with fixed E, V, and N, all states are equally likely
Entropy grows with
degeneracy
S(E,V,N) =
klnΩ(E,V,N)
In thermodynamic equations, k is
the boltzmann constant
k is equivalent to
R/NA
The degeneracy of M systems ΩM is equal to
ΩM
E (particle in a box) =
(h²/8mL²)(nx²+ny²+nz²), where L² = V3/2
dE =
TdS - pdV + μdN
dS =
(1/T)dE + (p/T)dV - (μ/T)dN
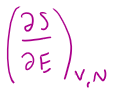
=
1/T
in determining E from S, E =
(3/2)NkT
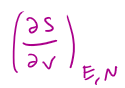
= BLANK, where BLANK is equal to BLANK
P/T, where P/T is equal to Nk(1/V)
Cv=
=(3/2)Nk
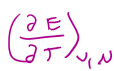
ni
the number of members with energy Ei
{ni}
set of occupation numbers
for ni, when i gets large all higher ni
are equal to zero
Weith (W) =
N!/(n0!n1!n2!…), where N is total number of partipating particles, and ni is the number of particles participating in that specific state
the total degeneracy of a system, ΩM =
the sum of all W
dlnW =
0
Q (canonical partition function) =
Σie-βEi
Pi =
ni/M = e-βEi/Q
Average E per copy can also be determined by the
product of Ei and Pi
Average energy E =
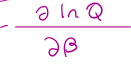
S (average entropy) =
kβE + klnQ
β =
1/kT
Pi =
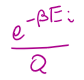
A(T,V,N) =
-kTlnQ = E - TS
in terms of average energies, Cv =
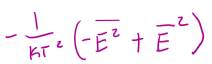
the degeneracy of a rigid rotor is
(2J+1)
for a harmonic approximation, the molecular partition function (degeneracy dependent) q =
1/(1-e-βε)
Ξ (grand canonical partition function) =
ΣN e-γNQ(N), where Q is the canonical partition function, and γ = -μ/kT
in terms of the grand canonical partition function, E =
where γ = -μ/kT
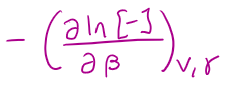
in terms of the grand canonical partition function, N =

entropy S of a grand canonical ensemble =

μ =
= -kTγ
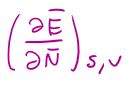
grand canonical ideal gas law
pV = kTlnΞ
λ (generalized activity) =
eμ/kT
in terms of activity, Ξ =
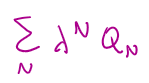
average N in terms of grand canonical partition =
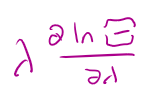
the probability of finding a particle in any state with N particles =

how different a copy N is to average N is equal to
σN/N (where σN is the square root of the root mean square)
σN2 =
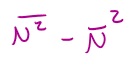
κT (isothermal compressibiity) does not change with
increasing number of systems
κT is NOT well behaved
near a phase transition
for an ideal gas, κT =
1/p
For 2 gaseous independent sub-systems we assume that VAB is
far less in magnitude to HA+HB
wavefunctions are BLANK while energies are BLANK
multiplicative, additive
Thermodynamic functions are
additive
lnΞ =
λq
qT (where T indicates translation energy only) =
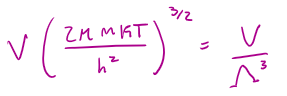
Λ (in units of length) =
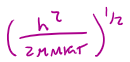
Λ is called the
characteristic thermal wavelength
if q » N and QN = qN/N!, then
V/Λ³ » N or NΛ3 « 1
ΔS for isothermal expansion =
Nkln(V2/V1)
in isothermal expansion, as the volume drops, e-levels available for translation
become closer together until they smooth to no longer be quantized
in an adiabatic process, the gas law appears as
pVγ = constant where γ = Cp/CV
γ is called
the adiabatic index
for monoatomic gases in an adiabatic process, ΔS =
0 (using the derivates with respect to helmholtz A)
What is the relationship between partition functions (canonical Q) in an adiabatic process
Q2 = Q1
what is the classical value of the adiabatic index for monoatomic gases
5/3
linear degrees of freedom
3n - 5
non-linear degrees of freedom
3n - 6
q sums over BLANK not BLANK
states, levels
Q for polyatomic gases appears as
qN/N! = (qTN/N!)qRNqVNqelN
Cp - CV =
R (the gas constant)
In a potential well the lowest possible value of ET is BLANK, ER is BLANK, Evib is BLANK, Eel is BLANK, and lowest possible energy E is BLANK
0, 0, 1/2ћω, -De, -D0
in a potential well De =
D0 + 1/2ћω
D0 is the
necessary energy to dissociate a molecule
De is the
depth of the potential energy well
rotational energies εi =
BJ(J+1), where B = ћ2/2I
qrot =
OR T/ΘR
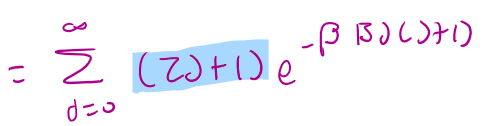
ΘR =
B/K
ΘR is called
the characteristic rotational temperature
when is qrot = T/ΘR true?
when T » ΘR
εvib =
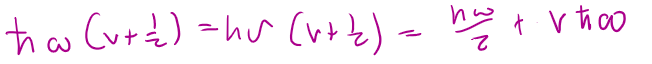
qvib =
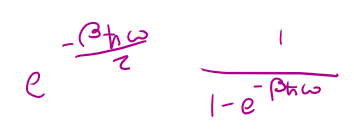
Avib =
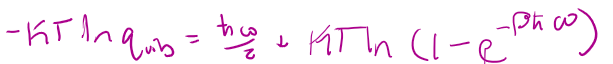
Θvib =
ћω/k
Θvib is called the
characteristic vibrational temperature
if T » Θvib, qvib =
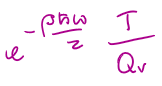
if T » Θvib,
Θvib/T = ћω/Tk « 1
near T = 0 K,
Cv ∝ T3
ε as T → 0 =
ћω/2
ε as T → ∞ =
ћω/2 +kT
dS =
dqrev/T
Extensive thermodynamic properties
E, V, n, S, ni
Intensive thermodynamic properties
p, T, μi
in terms of E, T =
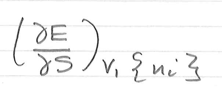
in terms of E, -p =
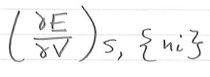
in terms of E, μi =
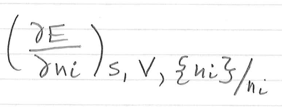
H =
E + pV
dH =
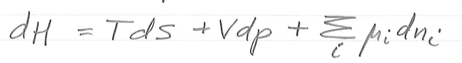
A =
E - TS
dA =