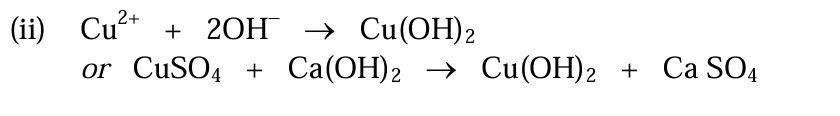d Block
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
Why can d-block elements form more than one type of ion
successive ionisation energies for the transition elements tend to increase gradually without large jumps in value
3d and 4s electron energy levels are very similar in energy
Energies of the d-orbitals are very similar
Define a transition metal
have either a partly filled 3d sub shell of form ions that have a partly filled 3d sub-shell
Why are most compounds that contain elements in high oxidation numbers referred to as complexes
If these ions were to form ionic compounds they would have a very high charge density
It would require too much energy to remove the 4,5,6,or 7 electrons to create the ion
Define ligand
A species possessing a lone pair of electrons which can form a co-oordinate bond to a central metal ion.
![<p>How can <span><span>[</span></span><strong><em>CoCl4]<sup>2- </sup></em></strong>be used as a test for water</p>](https://knowt-user-attachments.s3.amazonaws.com/9b7210b3-c0db-4e44-8c41-e47358834f6f.png)
How can [CoCl4]2- be used as a test for water
it is a blue compound that turns pink in contact with water. It forms [Co(H₂O)₆]²⁺
![<p>it is a blue compound that turns pink in contact with water. It forms <span><span>[Co(H₂O)₆]²⁺</span></span></p>](https://knowt-user-attachments.s3.amazonaws.com/ee2dc48a-88aa-4c40-8848-9b2ef4b62f4e.png)
Name the kind of reaction on the previous flashcard
ligand exchange
What complexes have a coordination number of 4
tetrahedral
What complexes have a coordination number of 6
octahedral
What name does an atom or group of atoms have if it only forms one coordinate bond
monodentate ligand
Name of molecules that can form two coordinate bonds
bidentate ligands
Name of molecules that can form several coordinate bonds
polydentate ligand
![<p>What happens if HCl is added to <span><span>[Cu(H₂O)₆]²⁺</span></span></p>](https://knowt-user-attachments.s3.amazonaws.com/e3284f1e-9b60-49bb-915d-362eb48de033.png)
What happens if HCl is added to [Cu(H₂O)₆]²⁺
[CuCl₄]²⁻ + H2O is made. This is an equilibrium reaction.
[Cu(H₂O)₆]²⁺(aq) + 4Cl⁻(aq) ⇌ [CuCl₄]²⁻(aq) + 6H₂O(l)
![<p><span>[CuCl₄]²⁻ + H2O is made. This is an equilibrium reaction.</span></p><p></p><p><span><span>[Cu(H₂O)₆]²⁺(aq) + 4Cl⁻(aq) ⇌ [CuCl₄]²⁻(aq) + 6H₂O(l)</span></span></p>](https://knowt-user-attachments.s3.amazonaws.com/924ab0e8-3f8f-4a47-a29e-a64f40a1ecf3.png)
Colour change of this equilibrium if HCl is added
[Cu(H₂O)₆]²⁺(aq) + 4Cl⁻(aq) ⇌ [CuCl₄]²⁻(aq) + 6H₂O(l)
Bright blue to yellow-green
Cr3+
green
Cr2O72-
orange
Fe2+
Pale green
Co2+
pink
CrO42-
yellow
MnO4-
purple
Fe3+
Red-brown
Cu2+
Pale blue
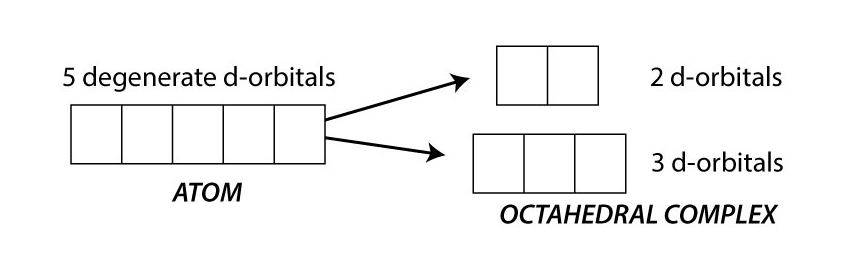
How are transition metals coloured
The presence of ligands around the central metal ion causes the d-orbitals to split into two energy levels.
The lower energy level contains three d-orbitals and the higher energy level contains two d-orbitals
When visible light is passed through a solution of this ion, some of the energy is absorbed which promotes an electron to a higher 3d orbital.
When light of a particular colour is absorbed, its complementary colour is reflected.
Why is Cu+ not coloured
Copper(I) complexes have an electronic configuration with a full d sub-shell
Why is Sc3+ ions not coloured
Sc3+ ions have an empty d sub-shell, meaning that electrons cannot move from lower to higher orbitals.
Equation when hydroxide ions are added to Cr3+
Cr3+(aq) + 3OH-(aq) ⇒ Cr(OH)3(s)
Observations for this reaction
Cr3+(aq) + 3OH-(aq) ⇒ Cr(OH)3(s)
green solution to grey green precipitate
Observations when OH- is added to Fe2+
Pale green solution to Dark green precipitate
Observations when OH- is added to Fe3+
Yellow solution to Red-brown precipitate
Observations when OH- is added to Cu2+
Pale blue solution to Pale blue precipitate
Observations when excess OH- is added to Fe2+
no further change
Observations when excess OH- is added to Cr3+
grey green precipitate dissolves giving a deep green solution
Equation when excess OH- is added to Cr3+
Cr(OH)3(s)+3OH−(aq)→[Cr(OH)6]3−(aq)
Observations when excess OH- is added to Fe3+
No further change
Observations when excess OH- is added to Cu2+
No further change
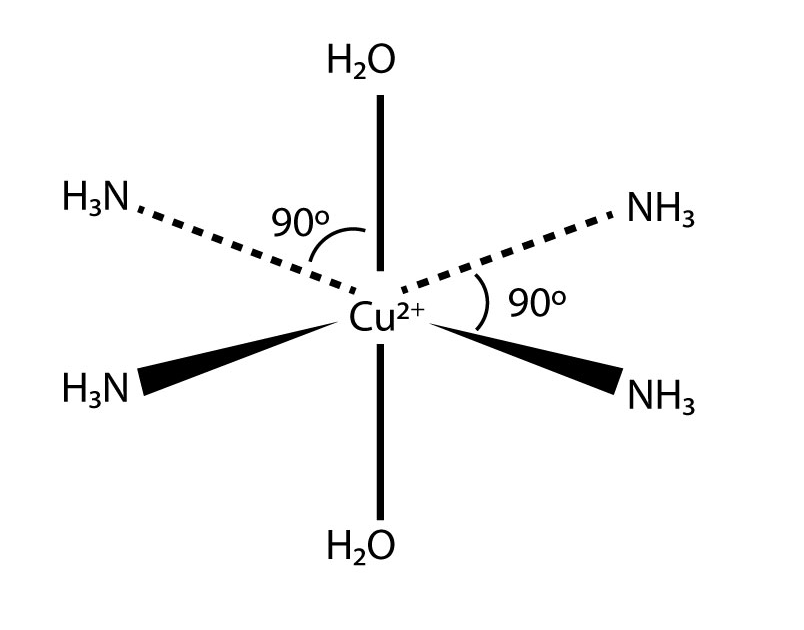
What happens if ammonia solution is added to [Cu(H₂O)₆]²⁺
[Cu(NH3)4(H2O)2]2+ + H2O
Shape of [Cu(NH3)4(H2O)2]2+.
Heterogenous
catalysts are in a different physical state to the reactants.
Homogenous
catalysts are in the same physical state as the reactants.
Define catalyst
substances that increase the rate of a chemical reaction by providing an alternative pathway with a lower activation energy
[Fe(H2O)6]2+
Pale green
[Fe(H2O)6]3+
Yellow
Catalyst for production of nitric acid
Platinum heterogenous
Catalyst for hydrogenation
Nickel heterogenous
Catalyst for polymerisation of Ethene
TiCl3 or TiCl4 - Ziegler-Natta catalysts
Catalyst for contact process
Vanadium oxide V2O5 heterogenous
Catalsyst for haber process
Fe heterogenous catalyst
Explain why both iron and copper are classed as transition elements, whilst zinc is not.
Iron and copper have partially filled d-orbitals in their ions, zinc does not