BioChem Exam 3 Study Guide
1/51
Earn XP
Description and Tags
*Know 10 steps of Glycolisis = enzymes responsible for each step, which steps use ATP , which steps make ATP, which steps use NAD+, how the steps are regulated.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Requirements for Functional Biological Membrane
1) SELECTIVE PERMEABILITY
-hydrophobic chains are impermeable to most molecules; but let small, nonpolar ones through
-membrane proteins regulate movement of ionic and polar substances
2) SELF-SEALING
-lipids must be able to fill in gaps after small disruptions in the membrane (need to be able to move around)
3) ASYMMETRY
-lipid composition of each leaflet can VARY
-differences are necessary for function
How does the membrane provide a SELECTIVE BARRIER?
By separating the cytoplasm from the environment
Why does the FLUIDITY of the membrane matter?
-Must stay flexible enough for normal function
-Allows lipids and many proteins to move laterally
-Helps the membrane self-seal after disruption
-Supports transport, signaling, and proper membrane proteins function
How does CHOLESTEROL regulate membrane fluidity?
-By BROADENING the Tm (melting temperature), it doesn’t actually change it
-Makes this transition from gel to liquid state less abrupt
Describe lipid movement between one leaflet to another + do they ever flip from one bilayer to another?
-Lipids RARELY flip from one leaflet to another
→ flip-flopping is energetically unfavorable, and usually requires energy and enzymes
→Because they rarely do this, the lipid bilayers are ASYMMETRIC
Why does asymmetry in the between the inner and outer leaflets matter?
-They dont have the same composition because the two leaflets do different jobs, so they can’t have the exact same composition
The outer leaflet: faces the extracellular space → helps with recognition, protection, cell-to-cell interaction (glycosphingolipids)
The inner leaflet: faces the cytoplasm → needs lipids that help with signaling and binding cytosolic proteins (PS and PIPs)
Describe what is meant by the fluid mosaic model:
-dynamic, flexible bilayer of lipids with proteins embedded in it
-phospholipids and many proteins can move laterally
-mosaic: patchwork of different components, mainly lipids, proteins, carbs
3 ways proteins can interact with the MEMBRANE
Integral, Peripheral, Lipid-Linked
1) Integral Membrane Proteins - aka TRANSMEMBRANE proteins embedded in the lipid bilayer, spanning it completely, acyl chains interact w/ hydrophobic amino acids
2) Peripheral Membrane Proteins - loosely/weakly attached to the membrane surface via interactions with lipid head groups or with integral proteins, DONT go into hydrophobic cores (non covalent, hydrogen bonds)
3)Lipid-Anchored Proteins- aka Acylated COVALENTLY attached lipid that inserts into the bilayer, while the protein itself stays on one side of the membrane
Two structural classes of Integral/Transmembrane Proteins
Alpha-helical, Beta-Barrel
Why are alpha helical and beta barrel the only real options of transmembrane proteins?
These are the only good options because the polar protein backbone must form internal hydrogen bonds to cross the hydrophobic membrane interior without exposing backbone groups
→ both structures can internally satisfy backbone hydrogen bonding
→ alpha helices: backbone hydrogen bonds within the helix
→ beta-barrel: beta-strands hydrogen bond to each other in a closed barrel
Difference between a PERIPHERAL membrane protein and a LIPID-LINKED:
Peripheral : attached NONCOVALENTLY (weakly) to the membrane surface, interacting w/ lipid head groups or other membrane proteins
Lipid Linked: attached to the membrane by a COVALENTLY attached lipid anchor, that lipid goes into the bilayer and holds the protein to the membrane
Passive vs. Active Transport
Passive Transport
-doesn’t use energy
-moves substances DOWN their concentration gradient
Active Transport
-requires ATP directly, or indirectly from another gradient
-moves substances AGAINST their concentration gradient
Passive Transport: Simple vs. Facilitated Diffusion
Simple Diffusion:
-Molecule move directly though the bilayer
-No transport protein is used!
-moves down the gradient, no ATP
(Small non polar like O2 and CO2)
Facilitated Diffusion:
-Requires a transport protein
-Still moves down the gradient and doesn’t require energy
(ions, polar molecules, larger solutes)
Passive Transport: Symport vs Antiport vs Uniports
UNIPORT
-One type of molecule is moved, so one solute + one direction
-Glucose transporter moving only glucose
SYMPORTER
-2 different types of molecules are moved, but in the SAME direction
-Ex. Na+ and glucose both moving into the cell
ANTIPORTER
-2 different types of molecules moved into the OPPOSITE directions
-one ion moves in while the other moves out
Active Transport: Where does the energy come from?
1) ATP HYDROLYSIS
-Primary active transport uses this
-Directly uses the energy released when ATP is broken into ADP and Pi
-moves solute against the gradient
2) ION GRADIENT
-Secondary active transport uses this
-Doesn’t use ATP directly, instead uses energy stored in a gradient that was created earlier by ATP powered pumps
ex. SGLT1 uses the Na+ gradient to bring glucose into the cell against its gradient
→ both are used to move molecules against their gradients low→ high concentration
What is METABOLISM?
-ALL the chemical reactions in a cell
-It is compartmentalized → maintains separate pools of metabolites, allows simultaneous opposing pathways, facilitated controlled behavior of enzymes
-2 main categories
1) Catabolism: degradative, breaking down macromolecules
-produces small molecules
-generates energy → produces ATP, NADH, FADH2
2) Anabolism: biosynthetic, building larger molecules
-requires energy input
-uses ATP and reducing power like NADPH
3 types of METABOLISM
LINEAR, CYCLIC, SPIRAL
Linear
-series of sequential reactions
-product of one step becomes substrate for the next
-Ex. Glycolysis
Cyclic
-starting intermediate is regenerated
-intermediates are replenished after each turn
-Ex. TCA cycle
Spiral
-same reactions repeat over and over
-common in polymerization or breakdown reactions
-Ex. Beta-oxidation
Purpose of COMPARTEMENTALIZATION
-Makes metabolism more efficient, controlled, and specific
-maintains separate pools of metabolites
-allows simultaneous opposing pathways
-creates high local concentrations of metabolites
-improves regulation and efficiency
Ways that a Single Pathway can be organized → 3 ways
Loosely, Tightly, Membrane Associated
Loosely Associated
-enzymes are separate
-intermediates diffuse from one enzyme to the next
Tightly associated/ Multienzyme Complex
-enzymes are physically associated
-intermediates can be channeled directly between enzymes
Membrane Associated
-enzymes are organized on or in a membrane
-helps to localize the pathway and improve coordination
FUTILE CYCLE - why are they so AVOIDED?
-When 2 opposing metabolic pathways run at the same time
-Converting a molecule back and forth and consuming ATP with little net output
It’s avoided because they waste energy, often as heat, making metabolism inefficient and can interfere with proper pathway control
Why are ATP and Acetyl-CoA considered high energy molecules?
Their hydrolysis is strongly favorable, so cells can use them to drive other reactions
Both molecules can couple favorable reactions to unfavorable ones and transfer useful groups in metabolism (exergonic to endergonic)
1) ATP
-phosphoanhydride bond cleavage relieves charge repulsion and forms more stable, resonance stabilized products
2) Acetyl-CoA
-thioester bond is less resonance stabilized, and hydrolyses to a more stable carboxylate
Major Nutrient Sources for MAMMALS
Carbohydrates, Fats (lipids), Proteins
-Mammals also need smaller amounts of vitamins, minerals, and water
1) Carbs → quick energy, glucose
2) Fats → long-term energy storage and membrane components
3) Proteins → amino acids for building proteins can be used for energy
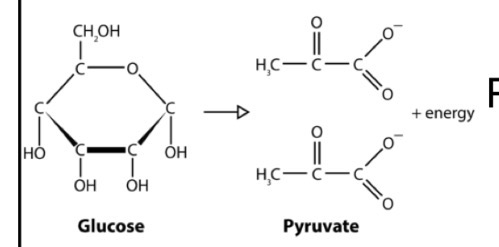
What is GLYCOLYSIS?
A CATABOLIC pathway that releases energy that is stored in carbohydrates to perform work
→ Glucose is transformed into 2 Pyruvates under ANAEROBIC conditions!
USES: Glucose, ATP, ADP + Pi, and NAD+
PRODUCES: Pyruvate, ATP, NADH
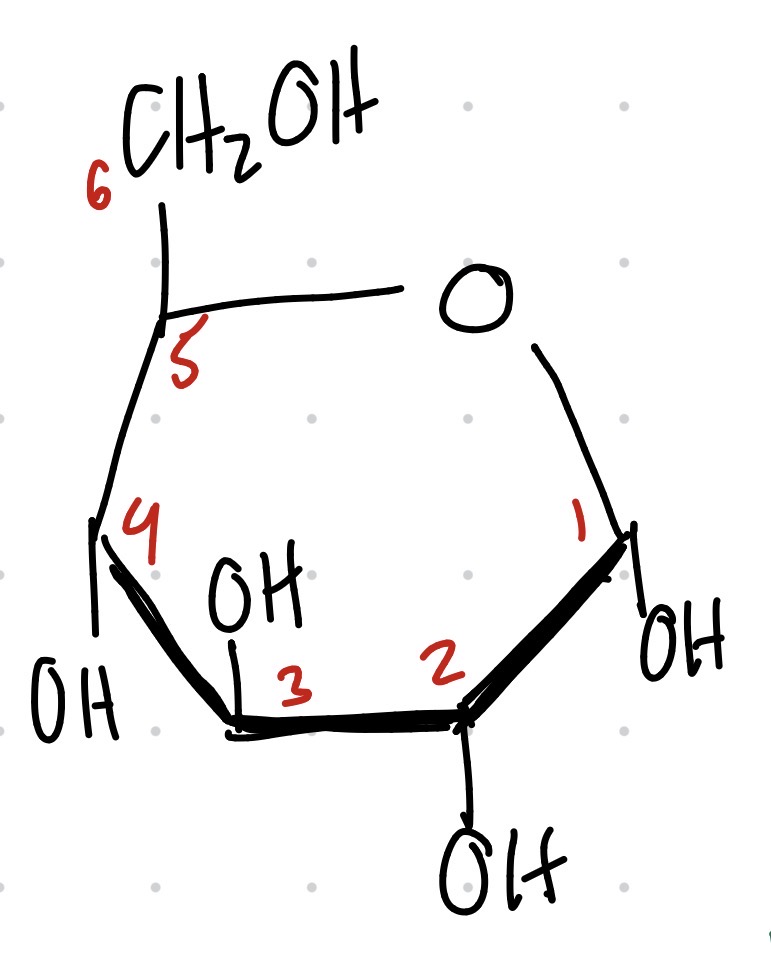
Structure of Gluose
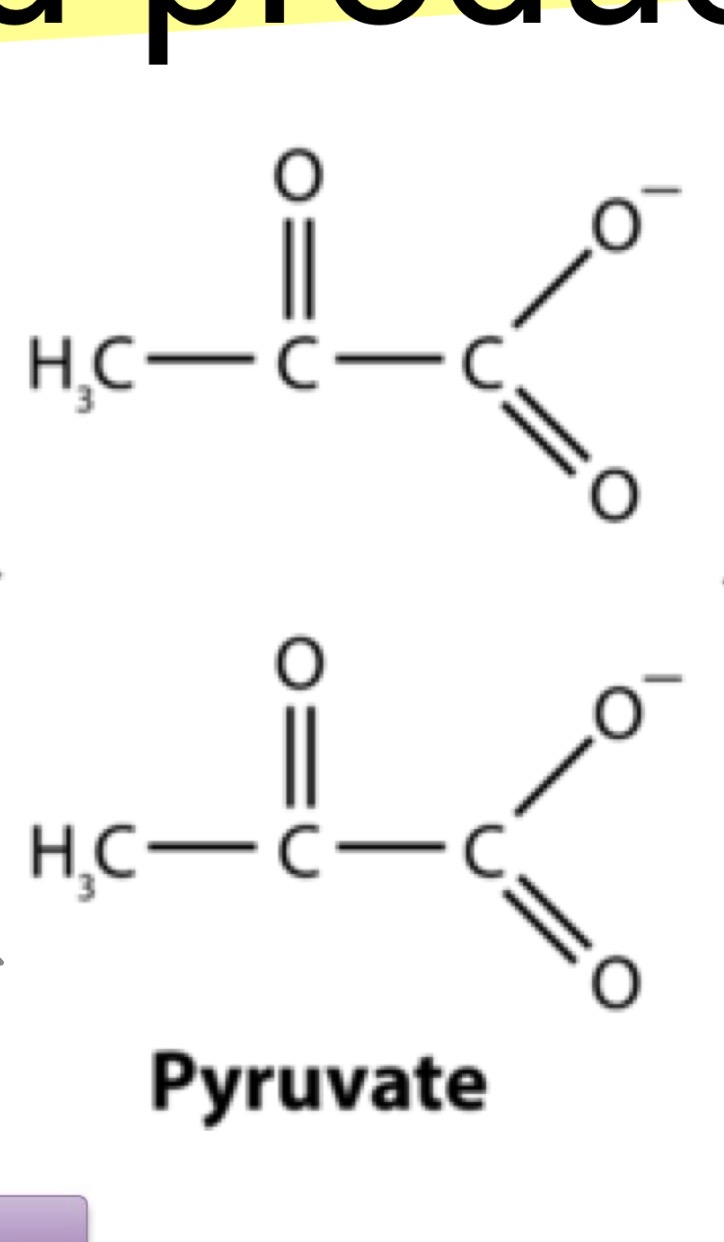
Structure of Pyruvate
Glycolysis Overall Equation:
Glucose + 2 ADP + 2 NAD+ +2 Pi → 2 Pyruvate + 2 ATP + 2 NADH + 2H+ + 2 H20
The 3 Fates of PYRUVATE in the cell
1) Aerobic - pyruvate is converted to Acetyl-CoA for the TCA cycle
2) Anaerobic - converted to lactate (animal muscles) or ethanol (yeast)
3) Anaerobic - reoxidize NADH to NAD+ which allows glycolysis to continue (helps the cycle start again)
Overview of Glycolysis
10 steps, linear
Some steps are TIGHTLY regulated
Each round
-Splits glucose into 2 pyruvates
-2 ATP are produced each
-2 NAD+ are reduced to NADH
2 MAIN STAGES = investment (energy is put in ) and dividend (energy production)
Investment Phase
Steps 1-5
Energy is put in to prime reactions in order to maximize energy production in stage 2
2 ATPs are invested per glucose molecule
Glucose is activated via phosphorylation making Glucose-6-phosphate (G6P)
At the end of this phase, glucose is converted into 2 molecules of Glyceraldehyde-3-phosphate (G3P)
Dividend Phase (Is repeated 2x)
Steps 6-10
Each G3P is converted into Pyruvate
By the end → 4 ATP + 2 NADH are produced
Since 2 ATP went in, net gain of 2 ATP
(*Glycolysis has modest energy return, it changes as pyruvate is further catabolized)
What’s so important about NAD+ and FAD, why do cells need them?
Their OXIDATION generates A LOT of ENERGY
-they ACCEPT H2 (2 electrons and 2 H+) from an organic substrate that is being oxidized (electron acceptors)
How do you know if OXIDATION occurred?
If a molecule
-LOST a H+ from carbon
-Gains an OXYGEN
-Has more C-O bonds
*Coupled with reduction!
How do you know if REDUCTION occurred?
If a molecule
-GAINED a H+ from on carbon
-Has FEWER C-O bonds
*Coupled with oxidation!
GLYCOGEN - why is it such an important STORAGE molecule in mammals?
It’s a highly branched polymer of glucose
Stores tons of free glucose in a compact form
It is branched so enzymes can remove glucose units rapidly
Good for short term, fast access energy
It’s stored so muscle can quickly make ATP for contraction
What is GLYCOGENOLYSIS?
BREAKDOWN of glycogen
-Removes glucose units from stored glycogen → produces glucose-1-phosphate
-Does this by CLEAVING alpha-1,4 glycosidic bonds
-When enzyme reaches branch point, a debranching enzyme has to help remove the alpha-1,6 linkage
→ this glucose comes off as a free glucose
Under what conditions does GLYCOGENOLYSIS occur?
When the body needs glucose or quick energy
Happens during fasting or when the insulin”glucagon ration is LOW (In the liver)
Muscle: happens during exercise especially intense fight or flight situations → make ATP quickly
→ epinephrine/adrenaline stimulates this through cAMP and phosphorylation cascade
What does GLYCOGENOLYSIS have to do with the reducing and non reducing ends of a glucose polymer?
Works at the NONREDUCING ends of glycogen
*Nonreducing ends are missing a free anomeric carbon, so glucose can be added and removed here
→ removes glucose residues one at a time from those ends and stops 4 resides before an alpha-1,6 branch point
→Branched form of glycogen allows many enzymes to be working at once, giving many nonreducing ends
How does insulin, glucagon, epinephrine, and cortisol regulate glycogen metabolism ?
They all regulate by deciding if the body should store or release glucose
Insulin → stores glucose
-released when blood glucose is high, like after a meal = “fed” state
-promotes glycogen synthesis, inhibits glycogen breakdown
Glucagon → release glucose from liver glycogen
-released during fasting, when blood glucose is low
-turns on glycogen phosphorylase
Epinephrine → rapid glucose breakdown, especially in stress/exercise
-promotes glycogen breakdown by signaling phosphorylase kinase
-muscles need ATP quickly for contraction
Cortisol → supports a long-term glucose raising state
-long-term stress hormone
-does not act fast
-increases gluconeogenesis
-maintains glucose avaliability
What is GLUCONEOGENESIS?
The synthesis of NEW GLUCOSE
-Provides a source of blood glucose (that is not glucogen) to prevent hypoglycemia
WHERE does GLUCONEOGENSIS occur in the cell?
It is split across compartments
Part occurs in the MITOCHONDRIA
Reversal steps occur in the CYTOSOL
In the ER, the G6P to glucose conversion occurs
Why dont cells run glycolysis in REVERSE to make glucose instead of GLYCONEOGENESIS?
-Because 3 steps are strongly EXERGONIC and essentially irreversible under cellular conditions (catalyzed by hexokinase/glucokinase, PFK-1, and pyruvate kinase)
-They have a large negative free energy value, so running them backward would be thermodynamically UNFAVORABLE
How does GLUCONEOGENSIS BYPASS the pyruvate kinase step (PFK-1) of glycolysis?
The irreversible step is : fructose-6-phosphate → fructose-1,6-bisphosphate by PFK-1
Gluconeogenesis BYPASSES it with fructose-1,6-bisphosphatASE which converts fructose-1,6-bisphosphate back into fructose-6-phosphate by HYDROLYSIS, not by making ATP.
How does GLUCONEOGENSIS BYPASS the HEXOKINASE/GLUCOKINASE step of glycolysis?
This step is — glucose → glucose-6-phosphate by hexokinase or glucokinase
It can bypass it with glucose-6-phosphatase which converts glucose-6-phosphate to free glucose by hydrolysis
How is GLUCONEOGENISIS regulated?
-Reguluated RECIPROCALLY with GLYCOLYSIS so the cell does not break down and remake glucose at the same time
-the biggest hormonal signal is GLUCAGON which promotes GLUCONEOGENSIS during fasting by increasing cAMP/PKA signaling
-Cortisol also promotes it more slowly by supporting enzyme expression and the fasting response
*High glucagon = make glucose
*High insulin = stop making glucose
What does fructose-2,6-bisphosphatase do?
It is the phosphatase domain of the bifunctional enzyme (PFK2/FBPase-2)
→ decreases glycolysis, increases gluconeogenesis
-It breaks down fructose 2,6 bis to fructose 6 phosphate which decreases the fructose 2.6 bis levels
-These low levels then inhibit PFK1 and activates fructose 2,6 bisphosphatase so glycolysis decreases and GLUCONEOGENSIS increases
What is PFK2/FBPase-2?
It is a bi-functional enzyme that both makes and breaks fructose-2,6-bisphosphate
What does fructose-2,6-bisphosphate do?
Its a regulatory molecule that activates PFK1 (promotes glycolysis) and inhibits F16 bisphosphatase that INHIBITS gluconeogenesis
What is the CORI CYCLE?
-Describes how glycolysis and gluconeogenesis interact
-Occurs during exercise, when aerobic metabolism in muscles cannot keep up with energy needs
-The cycle between the muscle and liver that links the two
-Glucose to pyruvate, then pyruvate to lactate to regenerate NAD+ so glycolysis can keep making ATP
*Lactate travels back to liver where it is converted back into pyruvate, used for gluconeogenesis to make new glucose, which is then sent back to muscle
Why is the CORI CYCLE important?
Because it lets the muscle keep producing ATP anaerobically, regenerates NAD+ for glycolysis, helps prevent lactate buildup, and maintains glucose supply.
What are the 3 PURPOSES of the Pentose Phosphate Pathway?
1) Generate NADPH
-generates almost all the NADPH used in reductive biosynthesis
-fatty acid and steroid synthesis require NADPH
2) Generate ribose-5-phosphate (R5P)
-used for biosynthesis of nucleotides, nucleic acids, and enzyme cofactors
3) Convert Pentoses → Hexoses and Trioses
-5C sugars in the diet converted to 6C and 3C sugars used in glycolysis and gluconeogenesis
What are the products produced from the PPP in the Oxidative vs NonOxidative phases?
OXIDATIVE =
-Oxidizes glucose-6-phosphate → NADPH and ribulose-5-phosphate (and CO2)
NON OXIDATIVE =
-Converts Pentose Phosphates → ribose-5-phosphate or glycolytic intermediates
Why do G6PD mutations matter biologically (PPP)?
G6PD catalyzes the first, rate-limiting step of the oxidative PPP, so mutations that reduce G6PD activity reduce NADPH production
This matters most in RBCs because the RBCs lack mitochondria, so the PPP is their only significant source of NADPH → without means they cant keep glutathione reduces, so they become vulnerable to oxidative damage (hemolysis)