PHYS (#6-10) 🚫MATH
1/112
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
113 Terms
Does light include radio waves? Why or why not?
YES. Visible light and radio waves are both forms of electromagnetic (EM)
radiation. They consist of varying electric and magnetic fields and travel at the speed of
light.
If the frequency of an electromagnetic wave increases, does the number of waves passing by us increase, decrease, or stay the same?
Number of waves passing by: Increases.
Reason: Frequency 𝜈 = waves per second (Hz). Higher 𝜈 means more waves pass a point per unit time.
If the frequency of an electromagnetic wave decreases, then the number of waves passing (λ) will: DECREASE
WHY: 𝜆𝜈 = 𝑐 , Since c (speed of light) is constant, if 𝜈 increases, λ must decrease.
Photon energy: Increases. Reason: 𝐸 ∝= 1/𝜆. Energy is inversely proportional to frequency, if λ decreases, E must increase
Compared to infrared (IR) radiation, ultraviolet (UV) radiation have-
Shorter • Wavelength
Higher • Energy per photon
Reason: From 𝐸 ∝= 1/𝜆, IF wavelength λ short, then photon energy high
Why are sound waves not included in the electromagnetic spectrum?
— Analogy: Just like water waves on a pond, sound waves do not belong on the electromagnetic spectrum
They are pressure (mechanical) waves traveling through a medium like air, not oscillations of electric and magnetic fields.
Red light < blue light (energy)
Red light < blue light (frequency)
Red light > blue light— 𝜆 wavelength
INVERSE: From 𝐸 ∝= 1/𝜆 and 𝜆𝜈 = 𝑐,
lower frequency = lower energy and longer wavelength.
With limited funds to build a large telescope, which would be the best choice, a refractor or a reflector? Why?
Best Choice: Reflector (mirror telescope).
1) Large refracting lenses are heavy. Need support at edges (SAGGING).
2) Lenses can’t be corrected for chromatic aberration.
3) Mirrors are easier and cheaper
Why put optical telescopes at the tops of mountains? Why put radio telescopes in deep valleys?
HINT: Optical telescopes: High mountaintops reduce the thickness of the atmosphere the light must pass through, minimizing blurring and absorption.
Radio telescopes: Radio waves are sensitive to human-made radio noise. Placing them in deep valleys helps shield them from this.
Why do single-dish radio telescopes have poor resolving power compared to optical telescopes of the same diameter?
• Key idea: Resolution depends on the ratio of aperture diameter (𝐷) to wavelength (𝜆).
Radio waves have much longer wavelengths than visible light. Result: Diffraction is greater for radio waves.
CAN be detected with an X-ray telescope but NOT be detected with an infrared telescope
—> Hot gas from exploding stars and regions near black holes.
These sources emit high-energy X-rays, not infrared radiation.
Small telescopes are often advertised as “200 power” or “magnifies 200 times.” How would these advertisements need to be changed to market those telescopes to astronomers?
• Key point: Astronomers do not rate telescopes by magnification.
What matters: Light-gathering power and resolution, both determined by the aperture
(size of the primary mirror or lens). High magnification is USELESS if image is too dim/lacks detail.
How many protons, neutrons, and electrons are in a neutral hydrogen atom? In a neutral helium atom? How many times more massive is the He atom compared to the H atom?
Hydrogen (neutral atom):
• Protons: 1
• Neutrons: 0 (most common isotope)
• Electrons: 1
So hydrogen is basically the simplest atom — just a proton with one electron around it.
Helium (neutral atom):
• Protons: 2
• Neutrons: 2 (most common isotope)
• Electrons: 2
So helium is more complex — it has a nucleus made of two protons plus two neutrons, with two electrons orbiting outside.
Mass comparison:
Hydrogen ≈ 1 atomic mass unit (amu)
• Helium ≈ 4 amu (2 protons + 2 neutrons)
• So helium > hydrogen. This is why helium is heavier and settles differently in stars compared to hydrogen.
How is an isotope different from an ion?
Ion:
• Atom that has lost or gained electrons
• Has a positive or negative charge
Isotope:
Hint: Ions differ by electrons, and isotopes differ by neutrons.
Atoms of the same element (same protons). Differ by the number of neutrons
• Ex: Oxygen always has 8 protons, but O-16, O-17, and O-18 have 8, 9, and 10
neutrons
A 3He (helium-3) nucleus contains two protons and one neutron. If the atom is neutral, how many electrons surround the nucleus? Is 3He an element, an ion, an isotope, and/or a molecule? Why?
Electrons: Neutral atom → electrons = protons. ³He has 2 protons → so it has 2 electrons orbiting the nucleus
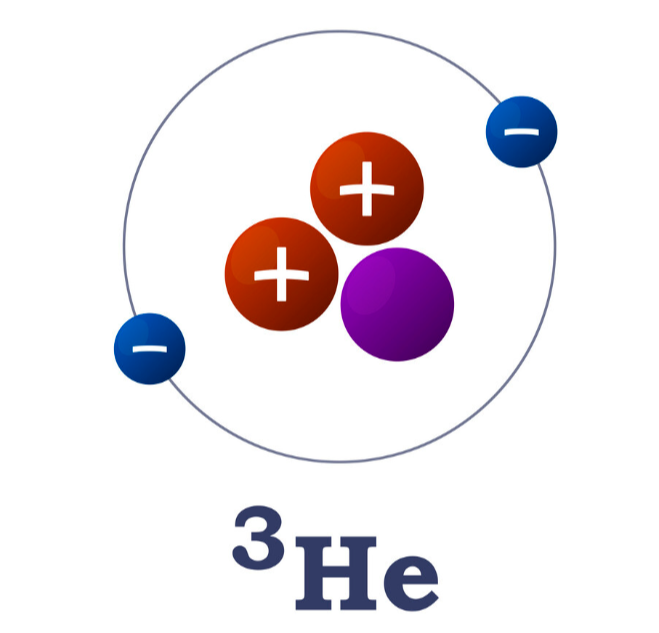
What is ³He?
Helium (defined by having 2 protons)
• Isotope: Yes → it has fewer neutrons (1)
• Ion: No → atom is neutral (no gain/loss of electrons)
• Molecule: No → it’s a single unbonded atom
So, ³He is a neutral isotope of helium with 2 electrons.
Atom excitement #1 Absorption of a photon:
• When photon energy = gap bet two electron energy levels
• The electron in the lower level absorbs the photon and jumps to the higher level
Atom excitement #2 Collision with another particle:
• Kinetic energy from the collision can be transferred to an electron
• The electron moves from a lower to an excited energy level
Why do different atoms have different lines in their spectra?- Electron energy levels differ:
Each element has a unique set of energy levels for its electrons.
Why do different atoms have different lines in their spectra?- Composition matters:
The number of protons, neutrons, and electrons in an atom determines these energy levels.
Why do different atoms have different lines in their spectra?- Transitions produce spectral lines:
• The differences between energy levels determine the energies of photons that can be absorbed/ emitted.
Why do hot stars appear bluer than cool stars? Particle motion:
HINT: Temperature of a star directly affects the color we see, with hotter stars looking bluer.
• In hot stars, atoms and electrons move faster
• Collisions between particles produce thermal radiation
Why do hot stars appear bluer than cool stars? Photon energy:
Hint: Wavelength and Energy INVERSE!!
• Hot stars emit more high-energy, short-wavelength photons → blue light
• Cooler stars emit more red, long-wavelength photons
Why do hot stars appear bluer than cool stars? Medium stars (like the Sun) emit roughly equal amounts of red, green, and blue. Appear white.
• Hottest stars emit mostly blue → appear bluish-white
• Cooler stars emit mostly red → appear reddish
Due to the relationship between an object's temperature and the wavelength of light it emits, aka blackbody radiation.
Celestial object A has a temperature of 60 K, and celestial object B has a temperature of 600 K.
Which object has the shorter wavelength of maximum intensity? Which objects has the least total energy emitted? 1/2
Wavelength of maximum intensity (Wien’s Law)
• Higher temperature → shorter λmax
• Object B (600 K) has a shorter wavelength peak than object A (60 K)
Celestial object A has T= 60 K, and object B has T=600 K.
Which has the shorter wavelength of maximum intensity? 2/2
Total energy emitted (Stefan-Boltzmann Law):
• Lower temperature → less energy per square meter
• Object A emits less energy per unit area than object B
What kind of spectrum does a neon sign produce? Describe this process. What colors are
associated with a true neon sign? Type of spectrum:
A neon sign produces an emission line spectrum
What kind of spectrum does a neon sign produce? What colors are associated with a true neon sign?
How it works:
• Electric current excites neon atoms → electrons jump to higher energy levels
• Electrons then fall back to lower energy levels → photons are emitted at specific wavelengths
What kind of spectrum does a neon sign produce?
HINT: The glowing red-orange color of a neon sign comes directly from the emission lines of excited neon atoms.
• Emission lines of neon atoms produce reds and oranges
• Signs of other colors are usually made with different gases or compounds, not
neon
The emission spectra from a star shows a hydrogen spectrum that is shifted toward the blue end of the electromagnetic spectrum. Where is the star moving. Towards Earth or nah?
HINT: Whenever spectral lines shift to the blue, the object is approaching us; a redshift = moving away.
Interpretation (Doppler effect): Blue shift → wavelengths get shorter. Shorter wavelengths mean the source is moving toward the observer. Conclusion: The star is moving toward Earth
Which kind of spectrum is produced by a white household incandescent lightbulb.
HINT: An incandescent bulb does not produce dark or bright lines; it emits a full, continuous range of colors.
Continuous spectrum. Reason: Produced by blackbody (thermal) radiation from the hot filament
• All visible wavelengths are emitted → smooth rainbow-like spectrum
Why can’t we view deeper into the Sun than the photosphere? /2
• Its gas has low density and low photons.
• The photosphere is therefore the thin layer from which most solar photons can
reach space. /2
Sun burns are caused by ultraviolet (UV) light with wavelengths shorter than ~300 nm. By Wien’s law, that wavelength corresponds to a temperature of about 10,000 K, like the mid-chromosphere.
The chromosphere is much less dense and about 1000 times fainter than the photosphere. Sun burn are from the Photosphere, which emits the largest share of solar radiation reaching Earth.
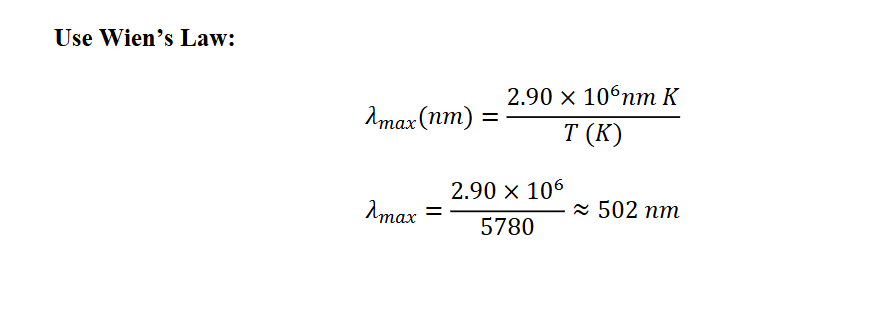
The average temperature of the photosphere is 5780 K. What color is the maximum intensity
of a 5780 K blackbody? Is this the color we normally associate with the photosphere?
This wavelength corresponds to green light. BUT the blackbody spectrum is broad w/significant intensity across red, blue, and other wavelengths.
- The combined effect of all wavelengths is perceived by the human eye as nearly white, not green.
Which atmospheric layer is associated with the Sun’s continuous spectrum? AKA the absorption/emission spectrum? /1
1) Continuous spectrum: Dense photosphere, emits thermal radiation.
2) Absorption spectrum: When light pass through cooler, low-density gases in (upper) photosphere. Produces dark absorption lines.
Which atmospheric layer is associated with the Sun’s continuous spectrum? /2
• Emission spectrum: Produced in low-density chromosphere and corona, where excited atoms emit light at specific wavelengths. /2
• Differences- Granules: Small convection cells just below the photosphere; visible as bright and dark patches.
• Super granules: Much larger convection cells, deeper in the Sun’s interior; linked to patterns in spicules in the chromosphere.
Likeness: Granules and super granules are BOTH the result of convection inside the Sun.
What is the evidence that the corona has a very high temperature?
HINT: The spectrum of the corona shows highly ionized atoms. To strip multiple electrons from atoms, very energetic collisions are required.
• Such high-energy collisions mean atoms and electrons are moving at very high speeds.
• High particle speeds correspond to a very high temperature in the corona.
Why does hydrogen (in the Sun’s atmosphere) have weak spectral lines, WHEN calcium (not abundant) has very strong spectral lines?
HINT: At the Sun’s photosphere temperature, most hydrogen electrons are low energy. Hydrogen mainly absorbs at UV wavelengths (Lyman series), not in the visible range.
Calcium has many atoms with electrons (in energy levels) that absorb at visible wavelengths, making its lines APPEARS strong. Hydrogen’s UV Lyman lines are SUPER strong, but invisible from the ground ://
Energy can be transported by convection, conduction, and radiation. Which of these is (or are) associated with the interior of the Sun?
• Radiation: Deep layer
• Convection: Outer layer
• Conduction: Not significant
How can solar flares affect Earth? HINT: Solar flares occur when magnetic field lines reconnect, releasing huge amounts of energy. /1
X-rays & UV radiation: Increase ionization, interferes w/ radio communication.
Charged particles: Trapped by Earth’s magnetic field → aurorae (northern & southern lights). /1
How can solar flares affect Earth? HINT: Solar flares occur when magnetic field lines reconnect, releasing huge amounts of
energy. /2
▪ Can distort the magnetic field, disrupting navigation systems.
▪ Cause surges in power lines on Earth.
▪ Can damage satellites in orbit /2
How can astronomers detect neutrinos from the Sun? Neutrinos travel straight from the Sun’s core through space (and Earth) at nearly the speed of light. Occasionally, neutrinos trigger nuclear reactions in certain atoms: /1
• Davis experiment: Neutrinos passing through a large tank of cleaning
fluid convert a few chlorine atoms into radioactive argon, which can be
detected. /1
How can astronomers detect neutrinos from the Sun? Neutrinos travel straight from the Sun’s core through space (and Earth) at nearly the speed of light. Occasionally, neutrinos trigger nuclear reactions in certain atoms: /2
• Modern detectors: Measure neutrino interactions with deuterium (heavy
hydrogen) to detect and study them. /2
Why are Earth-based parallax measurements limited to the nearest stars? /1
• Parallax measures the apparent shift in a star’s position due to Earth’s motion around the Sun.
• Earth’s orbital diameter (2 AU) is TINY compared to distances between stars. Even the nearest stars show only a tiny angular shift (fractions of an arcsecond) /1
Why are Earth-based parallax measurements limited to the nearest stars? /2
1) For distant stars, the parallax angle becomes too small to measure accurately with Earth- based instruments.
SO Earth-based parallax measurements are limited to only the nearest stars. If Earth’s orbit were larger, the parallax angles would be larger and measurable for more distant stars /2
Star A and Star B have measured stellar parallax of 1.0 arcsecond and 0.75 arcsecond,
respectively. Which star is closer? How do we know?
The larger the parallax angle, the closer the star is to Earth.
If a star’s apparent magnitude is equal to its absolute magnitude, what must be the star’s distance?
+When a star’s apparent magnitude (m) equals its absolute magnitude (M), it means there is no dimming due to distance.
M is the apparent magnitude a star would have if it were 10 parsecs away. Therefore, if m = M, the star’s distance is 10 parsecs
What does luminosity measure that is different from what absolute visual magnitude measures?
Hint: Luminosity measures the total energy output per second from a star across all wavelengths (the star’s total power). It represents the total energy emission
• Mv measures the brightness only in the visible part of the spectrum (light detectable by the human eye). Absolute Magnitude represents only the visible light output
We observe strong FeH (iron hydride) lines but weak CH4 (methane) lines in the spectra of a celestial object. What spectral class would we assign to the object based on line strength?
Hint: The spectrum shows strong FeH (iron hydride) lines and weak CH₄ (methane) lines.
• Indicates a warm atmosphere compared to cooler methane-rich objects.
• Shows L-type spectral class objects. So, the object is classified as an L dwarf, which is a type of brown dwarf.
How can we be sure that supergiants really are larger than main-sequence stars? /1
• Supergiants, etc can have the same surface temperature (and therefore the same color). BUT, supergiants are way more luminous than main-sequence stars of the same Temp. /1
How can we be sure that supergiants really are larger than main-sequence stars? /2
• Luminosity depends on both temperature and surface area (𝐿 = 4𝜋𝑅2𝜎𝑇4), their much higher luminosity must come from a much larger radius. /2
A star is known to be much larger than a main-sequence star and is blue in color. What kind of star is it?
• A blue color indicates a very high surface temperature.
• Being much larger than a main-sequence star means it is in a giant or supergiant stage.
• Therefore, the star is a blue supergiant (a very hot, massive, and luminous evolved star).

A star seems to have no measurable stellar parallax. Can we still find the distance to the star? If
so, how? If not, why not? /1
Yes, we can still estimate the distance even if the star’s parallax is too small to measure. Using spectroscopic parallax (not a true parallax). Determines Spectral type + luminosity class to find Mv /1
A star seems to have no measurable stellar parallax. Can we still find the distance to the star? If
so, how? If not, why not? /2
• By comparing its apparent magnitude (m) with the absolute magnitude, we can
calculate its distance using the distance modulus formula: 𝑑 = 10(𝑚𝑣−𝑀𝑣+5/5 )

How do the masses of stars along the main sequence illustrate the mass–luminosity relation?
Binary star systems allow astronomers to determine stellar masses accurately./1
Stars at the top of the main sequence are massive and very luminous, while those at the bottom are low-mass and faint. Shows the mass–luminosity relation → luminosity increases rapidly with mass./1
How do the masses of stars along the main sequence illustrate the mass–luminosity relation?
Binary star systems allow astronomers to determine stellar masses accurately./2
• The approximate relation is given by, 𝐿∝𝑀^3.5. Thus, a small increase in mass results in a large increase in luminosity, clearly
illustrated along the main sequence of the H–R diagram/2
If we view only the brightest stars in the night sky, what type of star are we likely to be observing? Why?/1
Upper main-sequence stars (hot and massive), Giants, Supergiants. Their brightness is due to their intrinsic luminosity./1
If we view only the brightest stars in the night sky because they’re NEARBY, what type are we seeing? Why?/2
Rigil Kentaurus (Alpha Centauri) – a nearby main-sequence star (~1.3 pc away)
Sirius – a relatively nearby main-sequence star (~2.6 pc away)
ISM contains gas:
• Absorption lines: Detected light via distant stars, lines reveal interstellar material at lower density + temperature than the star’s surface.
• 21-cm radiation: Emitted by neutral hydrogen (H I), shows vast clouds of interstellar gas.
• Molecular emission spectra: Some clouds show emission lines from molecules (like CO and H₂), indicating molecular gas regions.
ISM contains dust:
• Extinction: Starlight dimmed as it passes the interstellar medium, caused by dust absorbing/ scattering light.
Stars appear red cuz blue light is scattered by dust
• Reflection nebulas: Dust scatters the blue light. Therefore nebula BLUE.
What is the difference between H I and H II?
• H I: Neutral Hydrogen. H II: Ionized Hydrogen:
How is the blue color of a reflection nebula related to the blue color of the daytime sky? /1
• Both are caused by the scattering of light by small particles.
• Short-wavelength (blue) light is scattered more efficiently than longer wavelengths (red or yellow), which is known as Rayleigh scattering. /1
Whether it's interstellar dust or air molecules, smaller particles scatter blue light more strongly, creating the familiar blue glow
In the Daytime Sky: Air molecules in Earth's atmosphere scatter sunlight, making the
sky appear blue. /2
In a Reflection Nebula: Dust grains in the interstellar medium scatter the blue light from
nearby or embedded stars. /2
Why some distant stars appear redder than their spectral types suggest:
• Interstellar reddening occurs when starlight passes through interstellar dust.
• Small dust grains scatter blue light more efficiently than red light.
As a result, less blue light reaches us, making the star appear redder than it really is. The spectral type is determined by the absorption lines, not by the star’s color.
Why is the ISM transparent at near-infrared and radio but opaque in visual wavelengths?/1
Hint: Dust particles and Hydrogen absorption
1) Dust particle size: The dust grains scatter, making it opaque in visual wavelengths.
2) Hydrogen (70% of ISM) absorbs light at visible (Balmer) wavelengths. INCREASE opacity. /1
Why is the ISM transparent at near-infrared and radio but opaque in visual wavelengths?/2
3) Longer wavelengths (infrared and radio) are too large to be scattered by dust grains.
4) Wrong photons. Therefore, the ISM is transparent at these wavelengths./2
Name the four components of the ISM in order of hottest to coldest.
1) Coronal gas – Hottest component
2) Intercloud medium
H I clouds. —> Molecular clouds – Coldest component
How can the H I clouds and the intercloud medium have similar pressures when their temperatures are so different?
1) Gas pressure depends on both temperature and density. The hot intercloud medium has a low density, while the cool H I clouds have a higher density.
These differences balance out, allowing both regions to have similar pressures despite
their temperature difference
How does the interstellar medium interact with stars?
Gas-Star-Gas Cycle: The ISM and stars are connected in a continuous cycle.
• Contribution from Stars:
Hot stars and supernovas expel gas and dust into the ISM. Cool stars also contribute gas and dust
The gas and dust in the ISM collect into giant molecular clouds,
where new stars can form.
Star Formation
The second process (or objects) that remove material from the ISM.
White dwarfs matters, neutron stars, or black holes is REMOVE!!! from the ISM.
Gas and dust from the ISM are used to form new stars.
The second source that contribute material to the ISM.
1) Supernovas: Explosions of massive stars eject gas and dust into the ISM.
2) Stellar winds: Outflows from stars continuously add material to the ISM.
THREE ways thermal energy can be transported.
1) Convection – Transfer of heat by the MOVEMENT of hot material, which rises while cooler material sinks.
2) Conduction – Transfer of kinetic energy through COLLISIONS between neighboring particles.
Radiation – Transfer of energy by PHOTONS, emitted and absorbed as a result of random particle collisions (thermal emission).

Larger telescope → smaller 𝜃→ better resolution
Smaller 𝜃→ sharper detail.
Blue light bends more than red light.
▪ Stars appear with rainbow halos.
▪ Image gets blurred
Chromatic Aberration’s Problem: Different colors bend differently
Nature also causes interference: Some observations impossible near full Moon. Bright Moonlight washes out faint objects.
Avoiding cities: Light pollution = sky brightening from artificial lights. Scattered city lights make faint stars invisible.
Mountain tops improve “seeing”.
Smooth airflow reduces turbulence. Stars appear sharper.
Mountain Top— Thin, dry air improves transparency.
High altitude means less atmosphere to block light. Dry air is critical for infrared astronomy
Challenges of building on mountains.
Expensive and logistically difficult. Worth it for clearer, sharper images.
New mirror designs: Thin "floppy" mirrors → lighter, cool faster.
Segmented mirrors (hexagons) → easier to construct.
Active optics improve images: Computers adjust mirror shape in real time.
Compensates for bending and distortion.
Mounts have evolved: Old equatorial mounts mimicked Earth's tilt.
New alt-azimuth mounts are simpler, controlled by computers.
Atoms are mostly empty space /1
Mass is concentrated in the nucleus; electrons very low mass. /1
▪ Loss of electrons → positive ion.
▪ Gain of electrons → negative ion
Ions form when atoms gain or lose electrons.
▪ High temperatures in stars → violent collisions → fewer chemical bonds.
▪ Cool stars → TiO
Atoms can form molecules by sharing or exchanging electrons.
Atoms absorb photons with the right energy._
• Energy must match the gap between levels.
• Wrong energy → photon not absorbed.
• Electrons fall back down within a tiny fraction of a second.
• Extra energy is released as a photon of a specific wavelength.
> Excited atoms are unstable.
Each element absorbs and emits a unique set of wavelengths → atomic fingerprint.
> Each atom has unique energy levels.

Continuous Spectrum are produced by hot, dense objects (like a filament or lava).
• Shows all wavelengths of light → smooth rainbow-like spectrum.
• No lines → purely continuous.
Examples: lightbulb filament, hot lava, star photosphere. Wien's Law to find T.
Photosphere: visible “surface”.
Chromosphere: thin, pinkish glow during eclipses.
Corona: outer layer, very hot, source of solar wind.
The Sun’s atmosphere has three main layers. (PCC)
Photosphere: dense enough to emit light.
Chromosphere: low-density, ionized gas.
Corona: extremely hot, very low density
The Sun is mostly hydrogen and helium.
92.6% hydrogen atoms, 7.3% helium atoms
Trace elements: carbon, nitrogen, oxygen, iron

Umbra = dark central core.
Penumbra = lighter outer region.
Structure of sunspots
Maunder Minimum (1645-1715).
Very few sunspots. Coincided with "Little Ice Age".
Maunder Minimum (1645-1715) - Climate Link
Europe and N. America experienced colder winters. Shows solar activity affects Earth's climate.
Maunder Minimum (1645-1715)- Other low-activity periods recorded.
Similar events noted in historical records. Suggest long-term solar cycles influence climate

Hydrogen Fusion: Proton–Proton Chain Step 1
Two protons fuse → deuterium, positron, neutrino. Positron annihilates with electron → gamma rays