physics: Temperature
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
The kinetic theory of gases lead to the equation: 1/2m<c2> = 3/2kT
(i)Explain the significance of the quantity 1/2m<c2> [2]
(ii) You say equation to suggest what is meant by absolute zero of temperature. [1]
(i)
it is the main kinetic energy
Of atoms or molecules or particles
(ii)
at absolute zero atoms have no kinetic energy

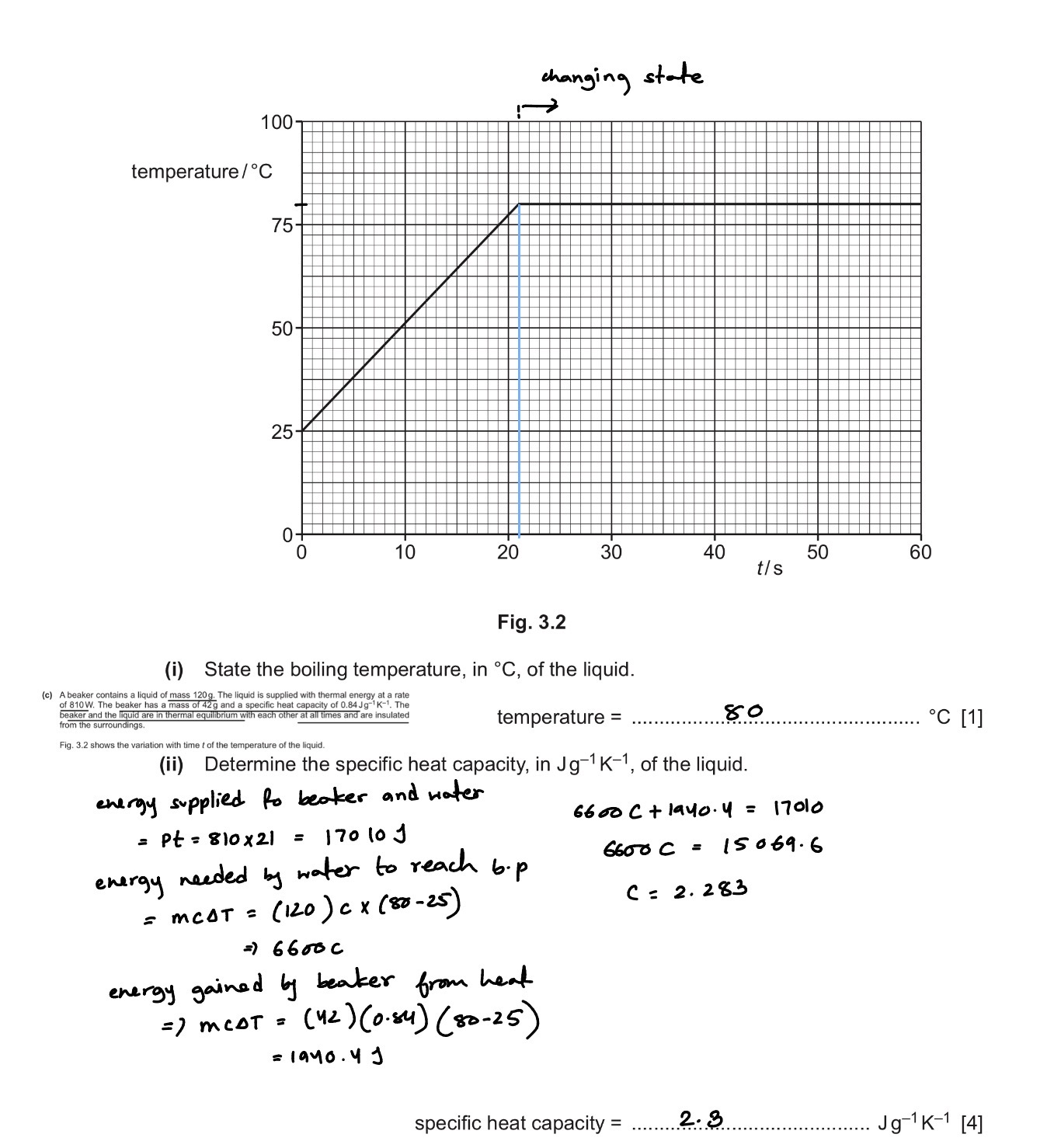
Suggest a suitable thermometer for the measurement of the initial temperature of gold (when melting)
thermocouple/resistance thermometer
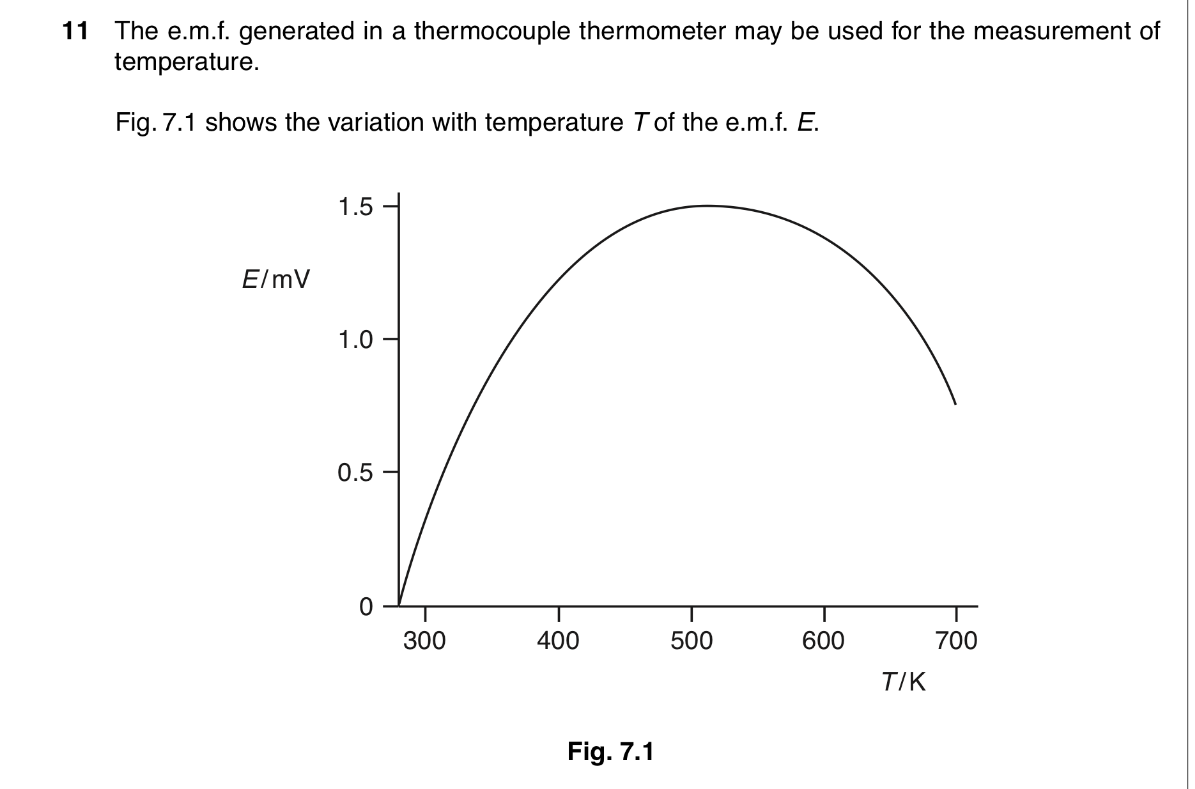
![<p>State and explain the feature of the figure which shows that the thermometer has a sensitivity that various temperature. [2]</p>](https://assets.knowt.com/user-attachments/8b2ee754-a397-489a-94f9-005eae551b32.jpg)
State and explain the feature of the figure which shows that the thermometer has a sensitivity that various temperature. [2]
The gradient of the graph is a measure of sensitivity
The gradient varies with temperature
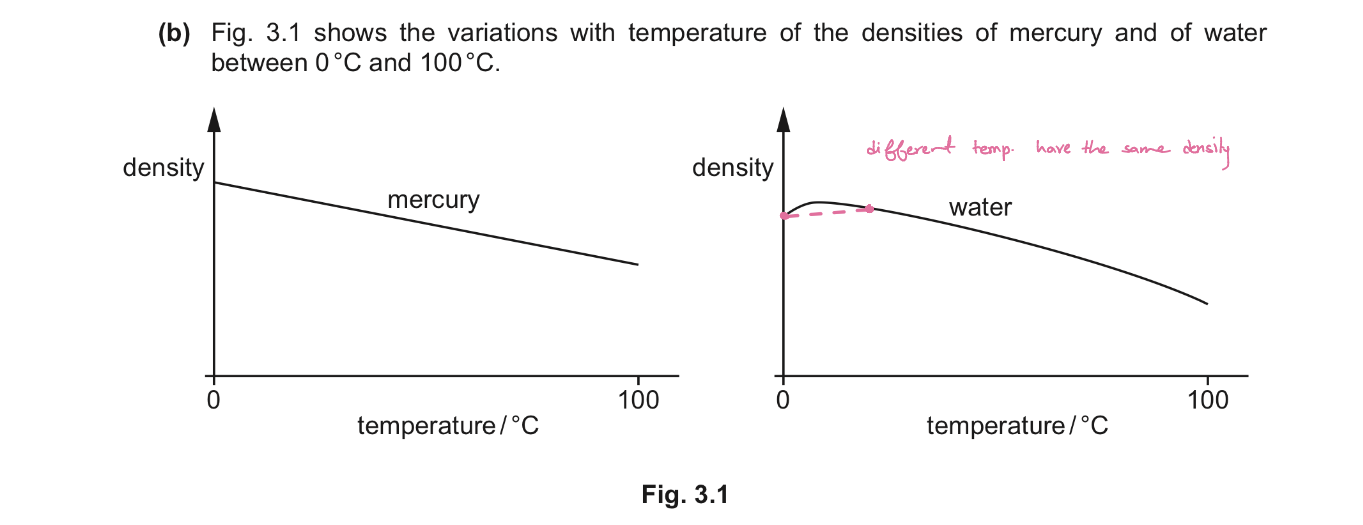
State the physical properties of materials that can be used for measuring temperature.
Density of liquid (mercury or alcohol)
EMF of a thermocouple
Resistance of a metal
The volume of a fixed mass of gas at a constant pressure.
A student states, quite wrongly, that temperature measures the amount of thermal energy (heat) in a body
State and explain observations that show why this statement is incorrect.
Two objects of the same material and at the same temperature, but with different masses, contain different amounts of thermal energy. (Because the larger mass has more total molecules).
During a change of state, such as melting or , thermal energy is absorbed by the substance, but there is no change in its temperature. This proves they are not measuring the same thing. (Since it is absorbing heat but its temperature is not going up, they cannot be measuring the exact same thing!)
Thermal energy (heat) naturally transfers from a region of higher temperature to a region of lower temperature, regardless of the total thermal energy stored in either body.
(Imagine you are sitting by a cozy campfire. A tiny, teeny-tiny spark flies out of the fire and lands on your jacket.That tiny spark is incredibly hot—maybe 1000∘C! Its temperature is way higher than your body temperature.Because it is hotter than you, the heat flows from the spark to you.But does that tiny spark have more total heat energy than your whole body? No way! You are much bigger.This proves that temperature only tells us which way the heat will flow (from the hotter thing to the cooler thing), not who has the most total heat energy.)
State what may be deducted from the difference in temperatures of two objects. [1]
Direction or rate of transfer of thermal energy
If different not in thermal equilibrium or energy is transferred
A thermometer and an electric heater are inserted into holes in an aluminum block, the heater is still switched on, and the readings of the thermometer of the block are taken at regular time intervals when the block reaches a constant temperature the heater is switched off and then further temperature readings are taken
Suggest why the rate of rise of temperature of the block decreases to zero.
as the blocks temperature rises, the rate of heat loss to the surroundings also increases
Eventually, the rate of heat loss equals to the rate of heat input from the heater
Hence there is zero net energy gained by the block, so the temperature stops rising
(I wrote that, block reaches equilibrium with heater and no net transfer of heat. This is wrong because, Heater is an electrical device that keeps getting extremely hot so instead of reaching thermal equilibrium with the block, the rate of heat loss equals the rate of heat input by the heater, and as the energy coming in equals to the energy coming out temperature does not rise.)
Given that a water has reached its boiling point and it’s been continuously heated by a heater. Why is the internal energy increasing.
water as it turns from liquid to vapor
Potential energy of molecules increases
OR
surroundings as its temperature rises
As energy lost and transferred to Surroundings
*(the higher the temperature difference between the surroundings and the object the higher, the rate of loss of heat to the surrounding) referred to the previous flash card
When a liquid is at its boiling point. Internal energy.
when a liquid is at its boiling point, the work done against atmosphere by the water vapor is equal no matter how much energy is given by the heater.(for example, 1 heater provides 20,000 J of energy and another heater provides 30,000 J of energy, the rate of heat loss hence the heat loss in a given time will be the same. Also, the work done.)
The energy loss is also equal
Hence, the energy input by the heater is equal to the energy used by the water, hence the increase in internal energy of the water
So energy input=Pt=VIt=energy used=U=ml (energy loss and work done cancels out)

State one change that could be made in a liquid in glass thermometer that would enable to give a more accurate measurement of temperature?
Using a liquid with lower specific heat capacity. (mercury or alcohol)
OR
Use a smaller mass of the liquid
Explain why a liquid in glass thermometer does not provide a direct measurement of thermodynamic temperature.
it depends on properties of a real substance (at absolute zero atoms stop vibrating as they have no kinetic energy at all so at absolute zero mercury or alcohol or any liquid will be frozen hence cannot expand a contract)
0°C is not absolute zero (thermodynamic temperature means temperature in kelvin. We cannot measure the temperature directly in kelvin using this thermometer as the thermometer it’s not starting from -273.15°C meaning it is not 0 Kelvin)
(Gas thermometer(ideal gas thermometer) a constant-volume gas thermometer can approximate thermodynamic temperature very closely in limit of low pressure approaching and ideal gas. It matches the thermodynamic temperature so measuring pressure at fix volume. You can infer the thermodynamic temperature.)
(i) in what way the absolute scale of temperature differs from other temperature scales
(ii) what is meant by the absolute zero of temperature
(i) it does not depend on the physical property of a substance
(ii) temperature at which the atoms have minimum or zero energy
What is the basic principle by which temperature is measured and using your answer explain why two the thermometers may not give the same temperature reading for an object. [3]
The basic principle by which temperature is measured: uses property of a substance that changes with temperature
Temperature scale assumes that the property of a substance changes linearly with temperature
Physical properties may not vary linearly with temperature(most cases, some may travel faster some slower)
agrees only at fixed points.(Because their "curves" are different, they will only agree at the fixed points where we forced them to match. At any temperature in between, they will disagree.)
Explain why the internal energy of ideal gases equal to kinetic energy
Internal energy equals to sum of kinetic energy of molecules and potential energy molecules
No intermolecular forces (PE is due to intermolecular forces like attraction and repulsion)
Potential energy of molecules is zero
Internal energy of ideal gases. (Q15c)
As we know, internal energy and idle gases equals to the total kinetic energy of the particles in the system (is there some molecular force, hence no potential energy)
But there’s still work being done and for work to be done volume needs to change. (Now the confusion is that potential energy depends on the variation of the separation of the molecules, but as there is no intermolecular forces, we are going to take potential energy zero)
changing the volume by doing work forces the atoms to gain or lose Kinetic Energy.
Expansion: Gas does work
Particles lose KE, Internal Energy decreases (unless you add heat!).
Compression: Work is done on gas Particles gain KE (like being hit by a moving bat) Internal Energy increases.
What is the first law of thermodynamics?
Energy cannot be created or destroyed, only transferred or converted from one form to another.
(U=Q+W)
A Ideal gas is supplied with thermal energy Q. Explain with reference to the first law of thermodynamics why the increase in energy in internal energy of the gas is Q. [2]
Increasing internal energy = Q + work done
Constant volume, so no work done.
Specific heat capacity of ideal gas
Q = kinetic energy

There is one container that has a gas with fixed the volume and another container has a gas that does not have a fixed volume. both supplied with thermal energy given that the one supplied with the gas that does not have fixed volume has constant pressure. Suggest, with a reason how the specific heat capacity of the gases in the two containers would compare.
as gas expands, it does work against the atmosphere.
For the same temperature rise more thermal energy is needed in the container with a gas with no fixed volume so has larger specific heat capacity
(Change in internal energy U = Q + W
In fixed volume
U = Q only, Q=U
And unfixed volume
U = Q - W (vol increases so W -ve)
Q = U + W
Hence more thermal energy required by gas of unfixed vol
Simply put, when volume is fixed, all the thermal energy supplied goes into increasing the internal energy and no energy is being wasted to do work, and when the volume is not fixed, extra energy is wasted to do work against atmosphere)
Use the first law of thermodynamics to suggest, with a reason, how the specific heat of vaporization of water at the pressure greater than atmospheric pressure compared with its value at atmospheric pressure. [2]
Work done by gas against atmosphere is more
So specific latent heat of vaporization is more
(Change in internal energy U= Q - W
More work done
Q = U + W
So more thermal energy required, and more latent of vaporization, Q=ml)
Use the expression p=1/3p<c2> (p as in rho/density) to show that the mean kinetic energy <Ek> of the atoms of an ideal gas is given by the expression <Ek>= 3/2kT
Explain any symbols that you use. [4]
(When told to ‘explain any symbols that you use’ always explain the symbols that represents a constant for example, Bozeman constant universal gravitational, constant or blank, constant or permittivity of free space constant, and etc., something that doesn’t represent a physical unit)
(also know that mean kinetic energy is this, but when you’re trying to find a total internal energy, you have to multiply the kinetic energy with the number of atoms in the system) 9702/41/MJ/10/Q2c(ii & iii)
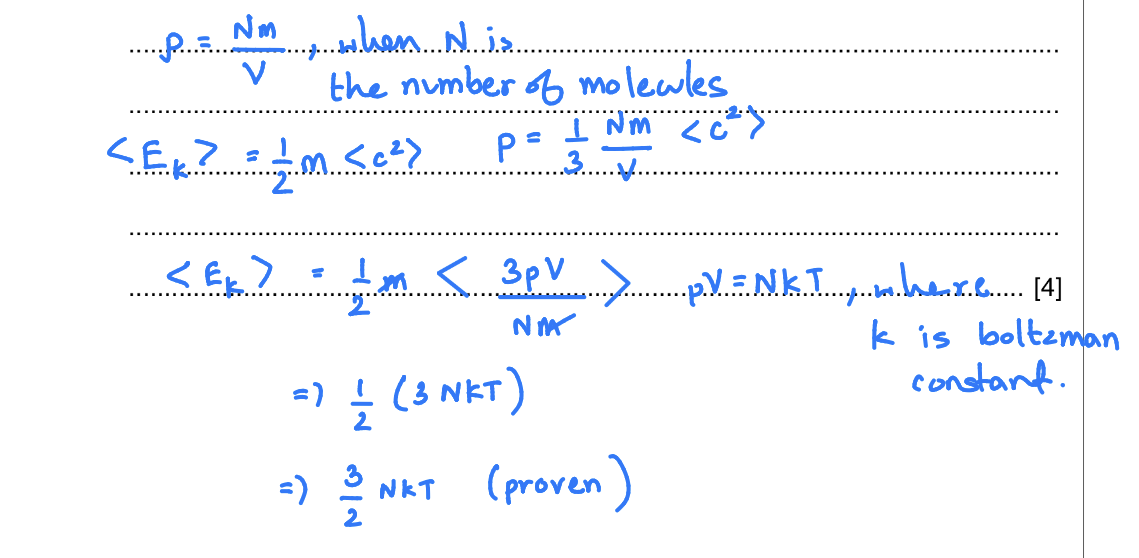
When the relationship between two variables is said to be varied linearly with each other, then it follows the equation
y = mx + c
For example, the relationship between the resistance of the thermistor and temperature:
(note that the thermodynamic scale does not depend on property of a substance so change in resistance of thermistor with temperature is non-linear)

How to choose thermometers?
range, sensitivity, linearity, accuracy, responsiveness, robustness, remote measurement
(i) the temperature of the flame of a Bunsen burner - thermocouple or resistance thermometer due to their wide ranges
(ii) they change the temperature of a small crystal when it is exposed to a pulse of ultrasound energy - thermocouple (can measure temp of very small objects due to their small sensors) or thermistor (very sensitive, even a tiny temperature rise causes a noticeable change in resistance)
A resistance thermometer, and a thermocouple thermometer are both used at the same time to measure the temperature of a water bath.
Explain why all the both temperatures have been calibrated correctly and are at equilibrium they may record different temperatures. [2]
Q14
The scale is calibrated, assuming that there is a linear change of property with temperature (when we say that a thermometer is calibrated assuming linear change, it means temperature scale as a straight line and we take two fixed points like 0°C, and 100°C, draw straight line between them and divide them into hundred equal parts assuming that there is linear change)
But neither property vary linearly with temperature. (a resistance thermometer relies on the change of resistance in a metal and a thermocouple rely on EMF generated between two junctions. None of this thermometric-properties varies linearly with temperature. Also resistance and EMF follow two different mathematical curves so their physical properties vary between the two fixed points, and agreed only at that fixed points)

An alternative to the thermocouple thermometer is the resistance thermometer.
State advantages that a thermocouple thermometer has over a resistance thermometer.
can measure temperature change of small objects / short response time
Can be used to measure temperature difference
No power supply needed
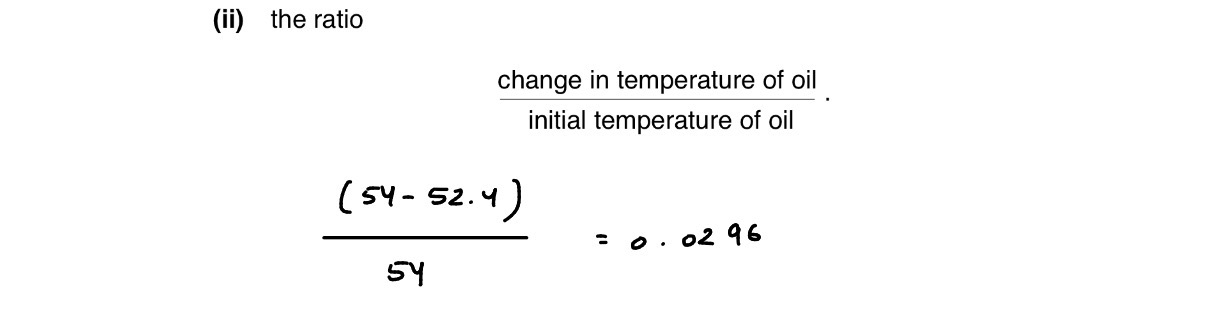
Suggest, with an explanation, a type of thermometer that would be likely to give a smaller value for the ratio calculated.
Thermistor thermometer or resistance thermometer
Because small mass or thermal capacity (Small Mass: A thermistor is an incredibly tiny electronic component compared to a bulky glass thermometer bulb filled with mercury or alcohol. Small Thermal Capacity: Because its mass 𝑚 is so small, its total thermal capacity (𝑚𝑐) is very low. This means it requires a negligible amount of heat energy to raise its temperature to match the oil. Effect on the Oil: Since the thermistor only absorbs a tiny, negligible amount of heat, the oil loses almost no heat. Result: The change in temperature (Δ𝜃) of the oil will be extremely small, making the ratio give a much smaller, more accurate value.)
A thermocouple does not directly measure absolute temperature. It generates an electromotive force (e.m.f.) based on the temperature difference between its measurement junction (the hot end in the oil) and a reference junction (the cold end). Choose a Thermocouple only if the question specifies that the temperature is changing very rapidly or is extremely high (like a furnace).
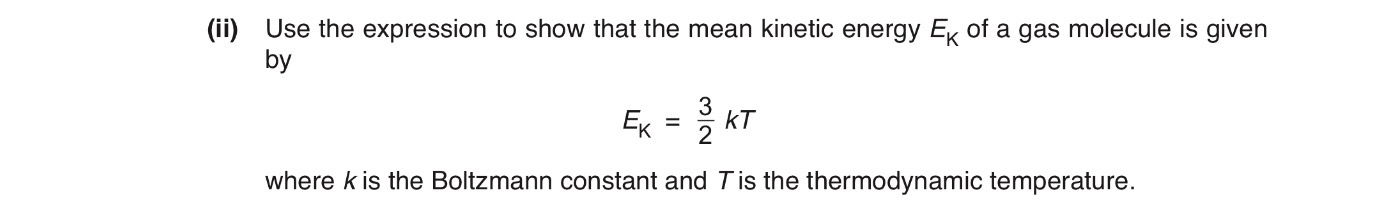
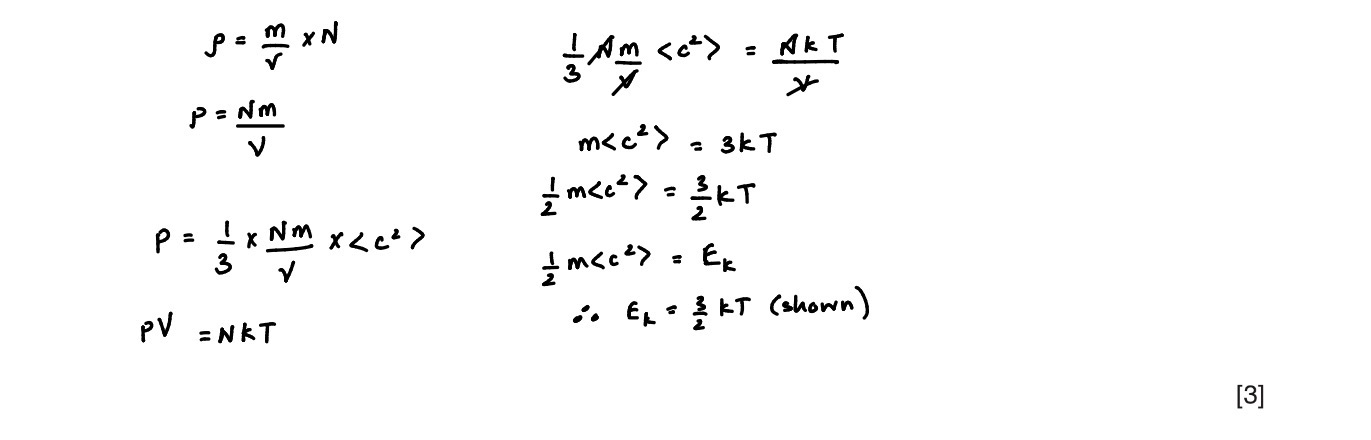

The expression in a(ii) is Ek= 3/2kT

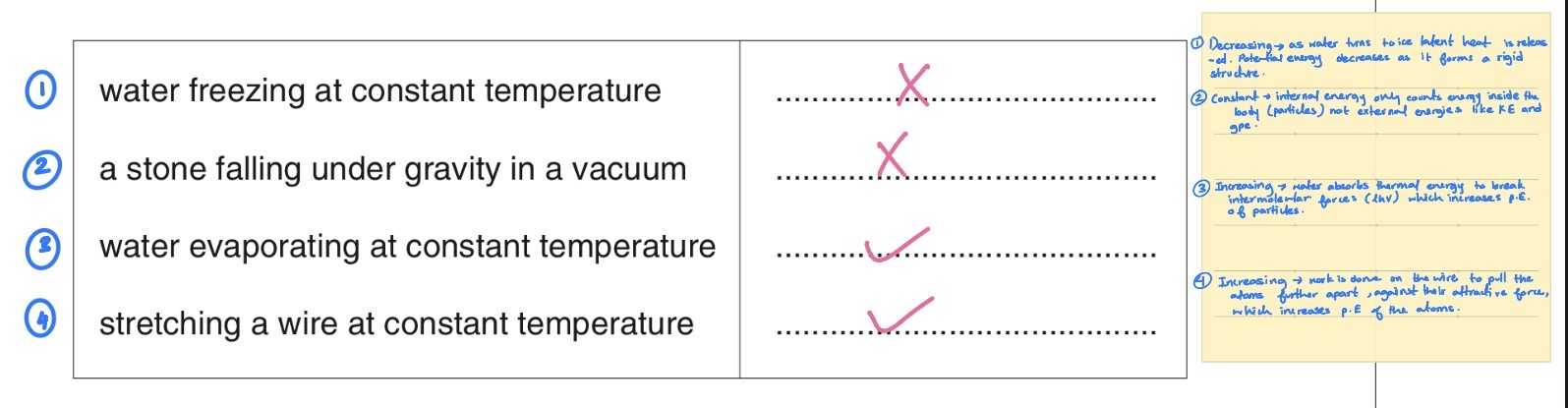
During melting, a solid becomes liquid with little or no change in volume use kinetic theory to explain why during the melting process Thermal energy is required, although there is no change in temperature.[3]
during melting bonds between atoms are broken
Potential energy of atoms is increased (as distance between atoms increased)
No work done so required input of energy is thermal. (we need thermal energy to increase potential energy as there is no work being done on the gas or work is done by the gas as volume remains constant, so the only way to increase potential energy is by providing thermal energy)
(when kinetic theory is mentioned, always remember it is a model based on the microscopic motion of atoms or molecules of a gas so talk about what is happening to atoms or molecules or particles and their bonds)
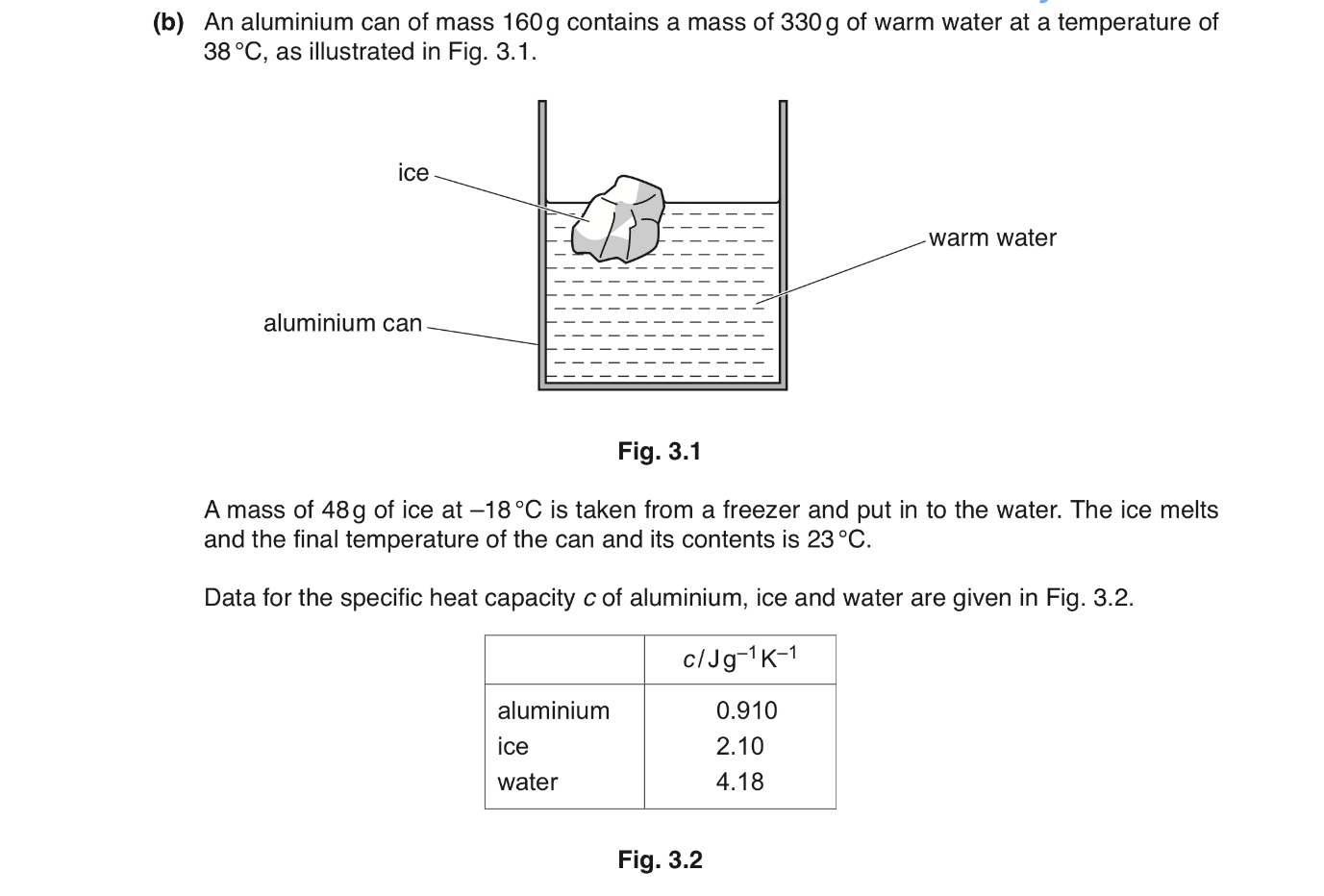


2023 41 MJ Q3