Oxidation/Reduction/Ether/Epoxides Reactions and Reactants
1/94
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
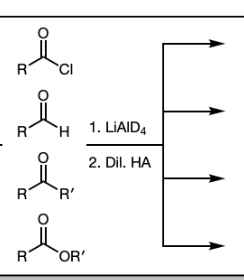
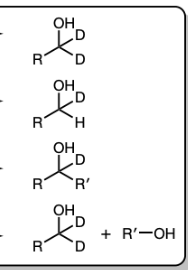
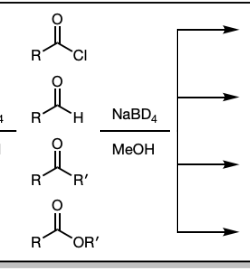
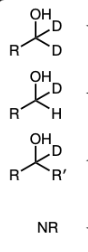
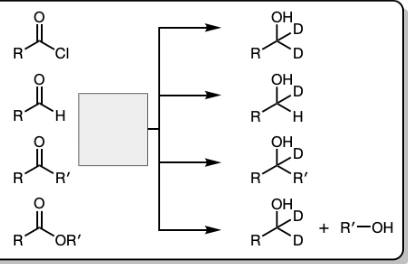
1) LiAlH4
2) Dil HA
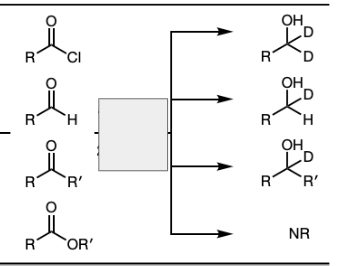
NaBH4 and MeOH
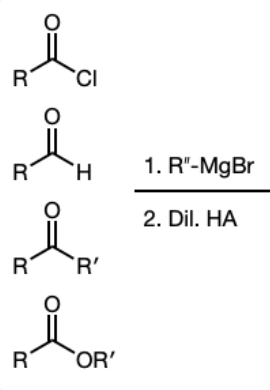
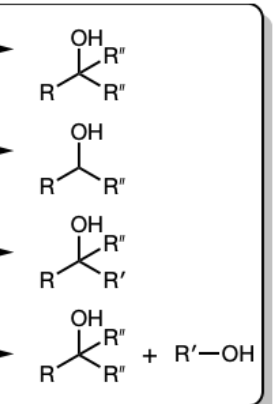
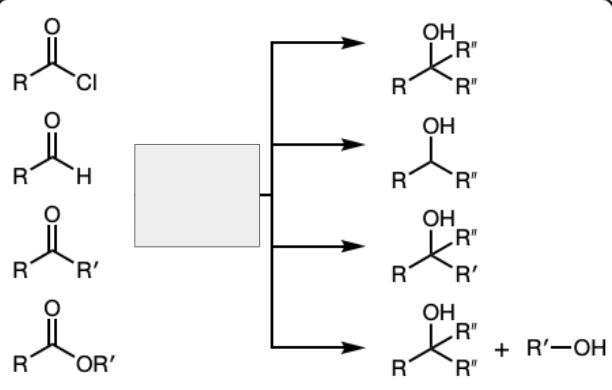
1) R’’—MgBr
2) Dil HA
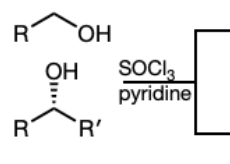
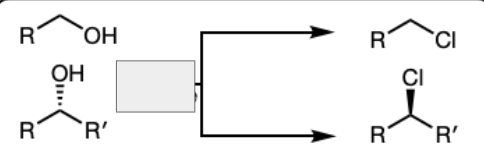
SOCl3 in pyridine
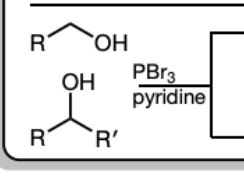
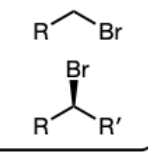
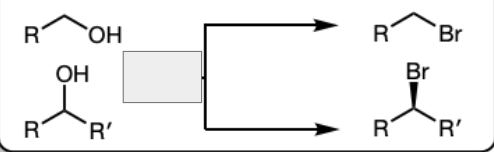
PBr3 in Pyridine
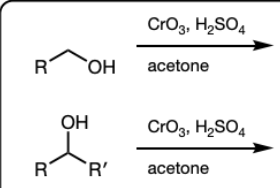
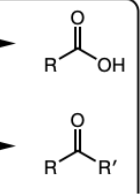
CrO3, H2SO4, acetone
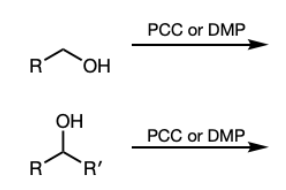
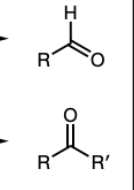

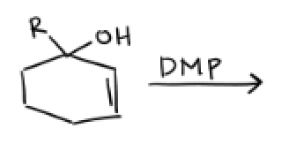
PCC or DMP
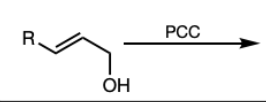
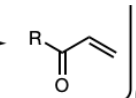

PCC
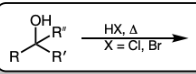
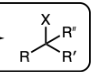
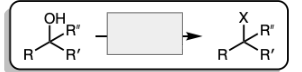
H-X and heat (X=Cl, Br)
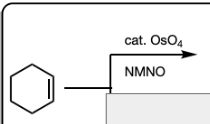
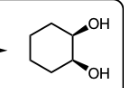
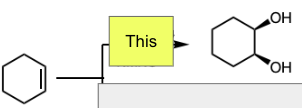
Cat. OsO4 and NMNO
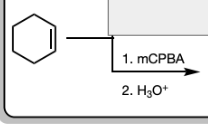
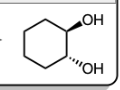
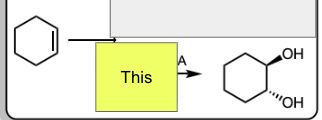
1) mCPBA
2) H3O+
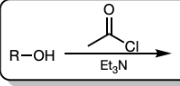
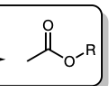
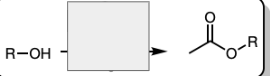
and Et3N
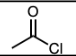
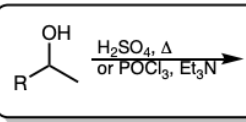
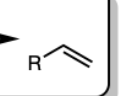
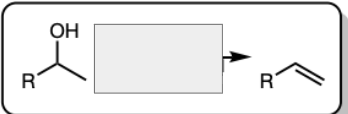
H2SO4 and Heat
OR
POCl3 and Et3N
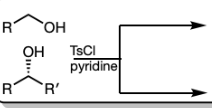
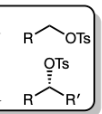
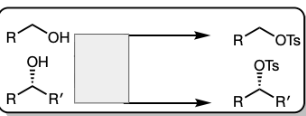
TsCl in pyridine
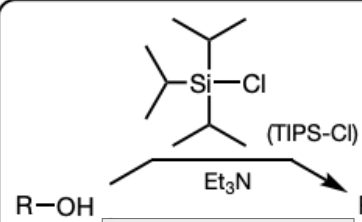

TIPS-Cl and Et3N
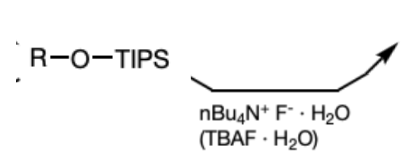
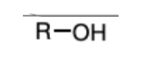
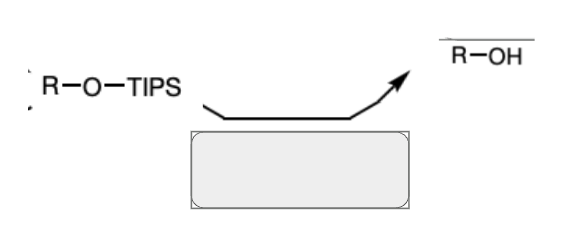
nBu4N+F- and H2O
(TBAF and H2O)
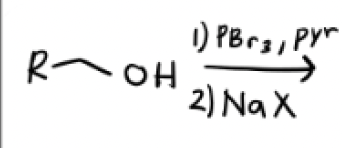

1) PBr3 in Pyridine
2) Na—x
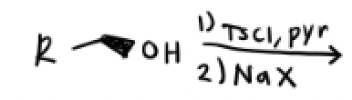
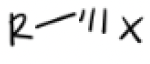
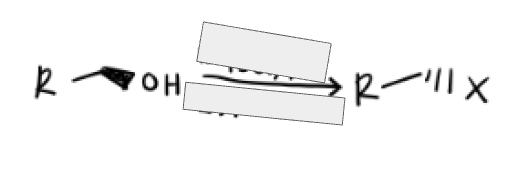
1) TsCl in Pyridine
2) Na—x
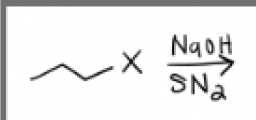
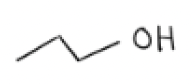
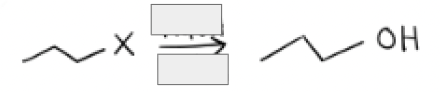
NaOH + Sn2 conditions
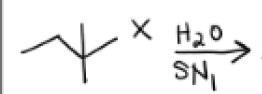
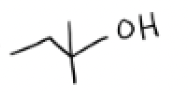
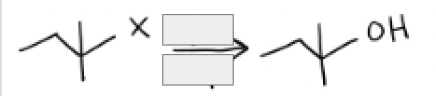
H2O + SN1 Conditions
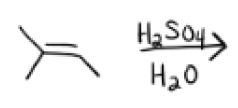
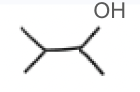
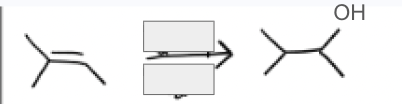
H2SO4, H2O
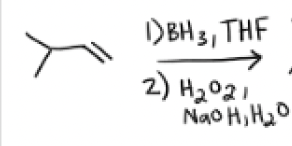
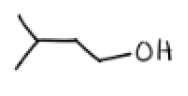
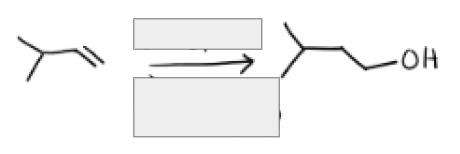
1) BH3, THF
2) H2O2, NaOH, H2O
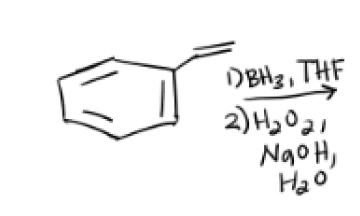
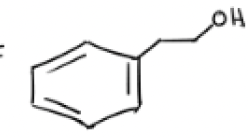
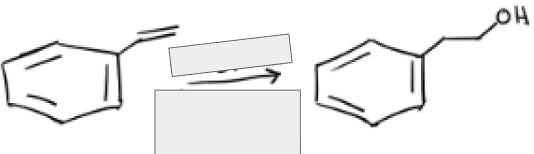
1) BH3, THF
2) H2O2, NaOH, H2O
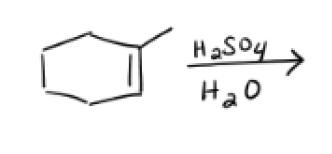
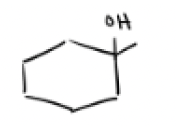
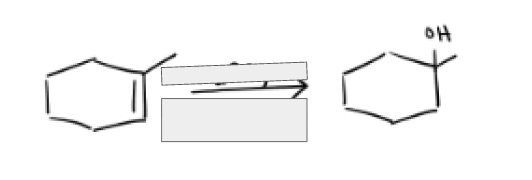
H2SO4, H2O
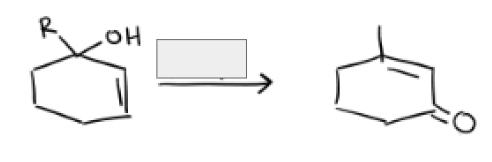
DMP
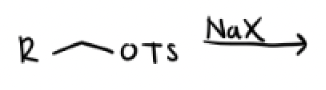
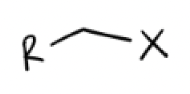

Na—X
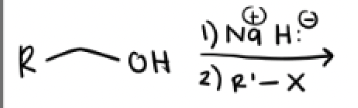
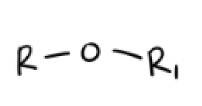
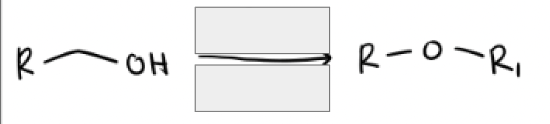
1) Na + and H-
2) R’—X
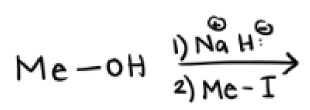
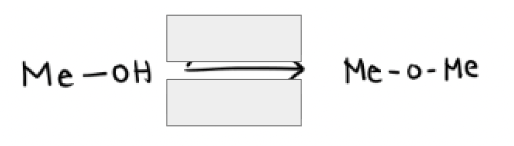
1) Na + and H-
2) Me—X
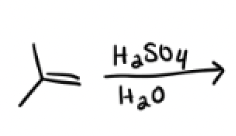
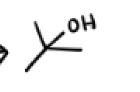
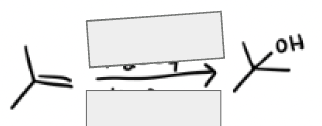
H2SO4, H2O
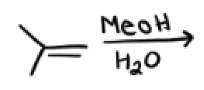
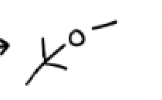
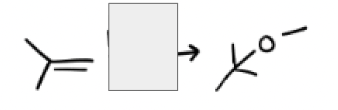
MeOH, H2O
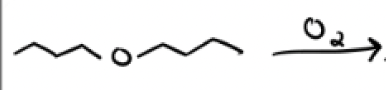
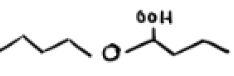
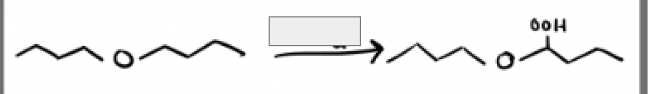
O2
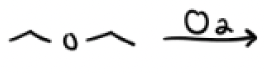
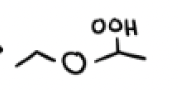

O2
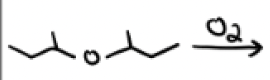
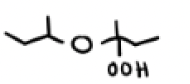

O2
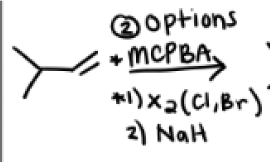
MCPBA
Or
1) X2 (Cl or Br)
2) NaH
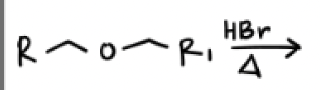
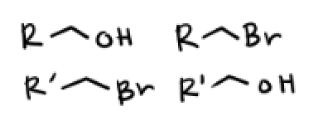
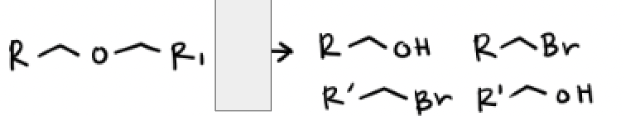
HBr and Heat
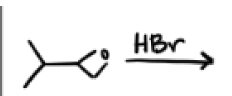
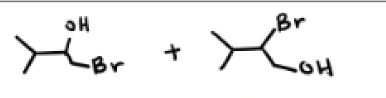
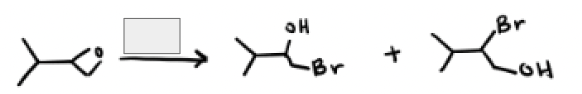
HBr
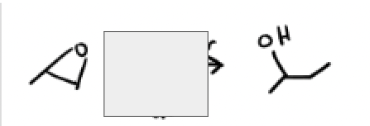
1) Me—Mg—Br
2) dil acid
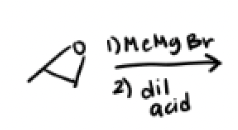
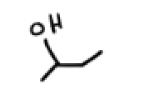
The grignard reaction fails under the presence of what?
Acidic protons such as phenols, amines, carboxylic acids, terminal alkynes and alcohols. They immediately protonate the reagent and destroy it.
Why can’t tertiary alcohols be oxidated?
Oxidation reactions are the loss of an alpha hydration. Tertiary alcohols lack the alpha hydrogen, so they are resistant.
Oxidation rate of alcohols
2 > 1 > 3
secondary alcohols just oxidize to ketones and stop
primary alcohols go through an aldehyde, then a carboxylic acid
Draw mCPBA
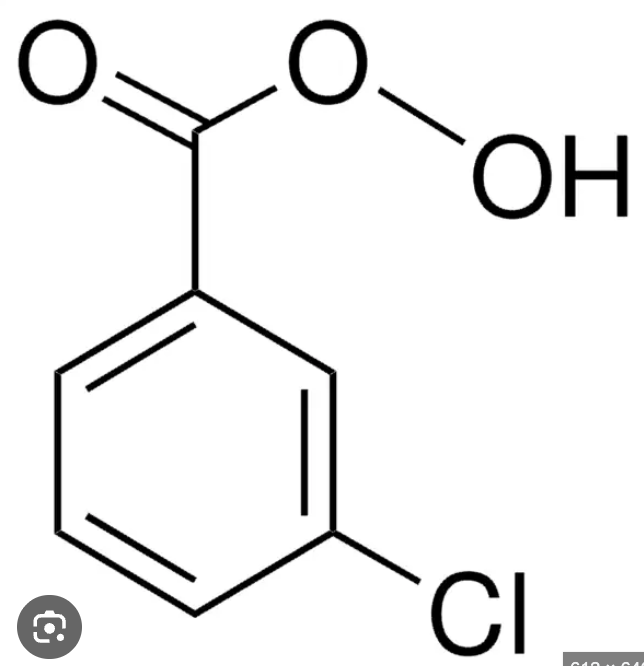
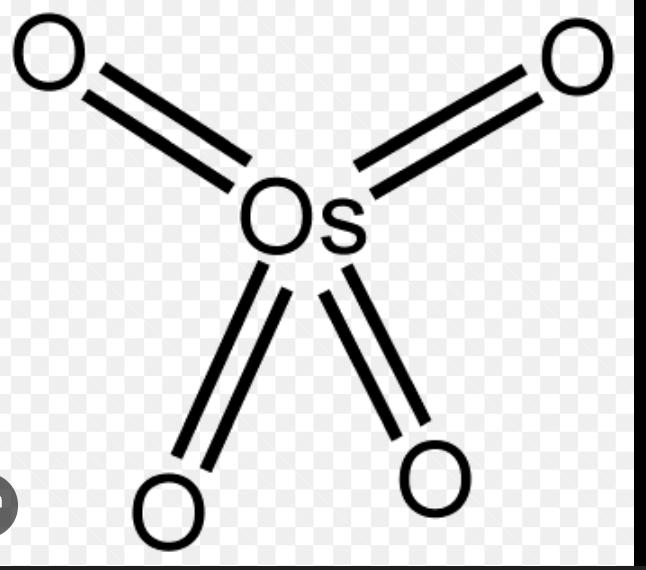
Draw O5O4
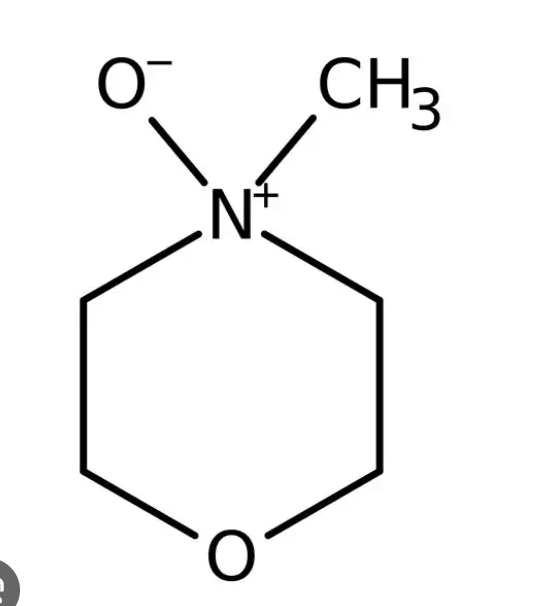
Draw NMO
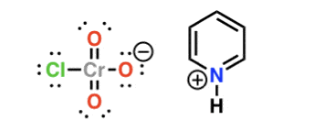
Draw PCC
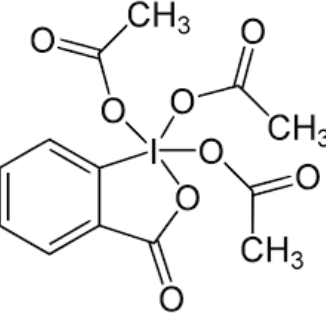
Draw DMP
Conversion of alcohols to esters rates?
1 > 2 > 3
depends on steric hindrance.
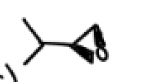
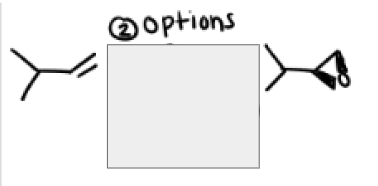
From an epoxide reaction formation, when should mCPBA be used?
the two options to form an epoxide are:
mCPBA or 1) X2 2)Nah
It should be used if a molecule has acid sensitive groups or can be destroyed by water or x2
Why are smaller rings happier to open?
Severe ring strain, the ideal bond angle is 109.5 degrees and it’s being forced into 60 degree angle
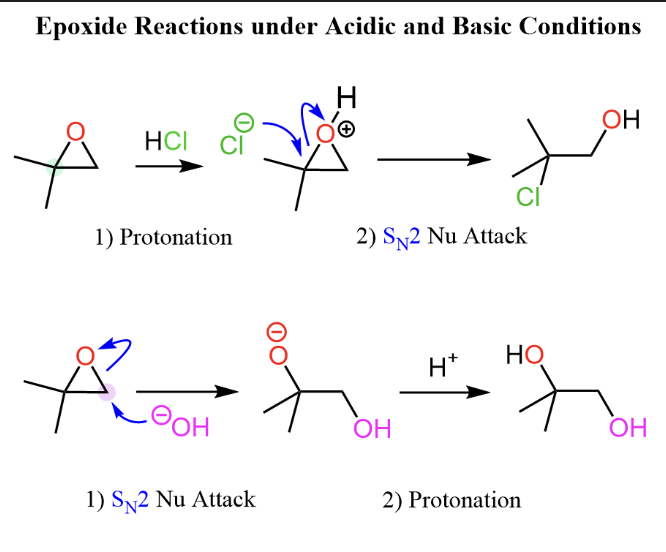
In acid catalyzed ring-opening of epoxides, how do major and trace amount products change in acidic and basic reactions?
In acidic conditions (H3O+, H2SO4, H-X):
Nucleophile attacks the most substituted carbon or benzylic carbon and alcohol goes into the least substituted carbon
Protonation of the oxygen makes it a good leaving group, creating a partial positive charge on the more substituted carbon
In basic conditions(OH-, OR-, CN-):
Nucleophile attacks the less substituted carbon and alcohol goes on the most substituted carbon
Strong nucleophiles prefer the less hindered carbon via an SN2 mechanism
Electron-withdrawing groups (EWG) do what to pka?
lower it —- increases acidity
Stabilize and result in a stronger acid because acidity depends on the stability of the conjugate base
Electron-donating groups (EDG) do what to pka?
increase pKa — lowers acidity
Destabilize and result in a weaker acid because acidity depends on the stability of the conjugate base
What are some solvent for SN2 reactions?
polar aprotic
(acetone, DMSO, DMF, MeCN, HMPA, THF, acetonitrile, dichloromethane)
What are some solvent for SN1 reactions?
polar protic
water, alcohols, and carboxylic acids
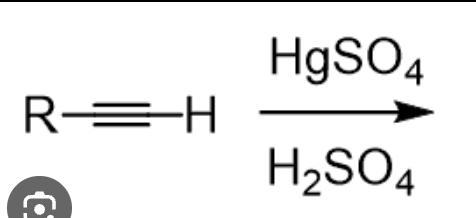
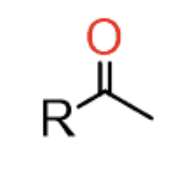

HgSO4
H2SO4
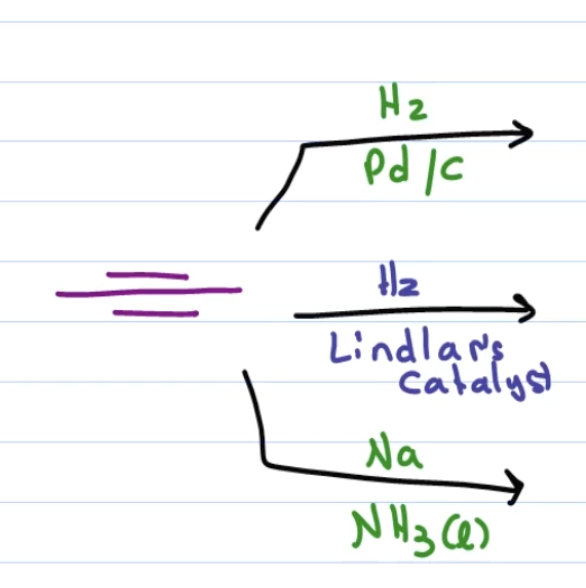
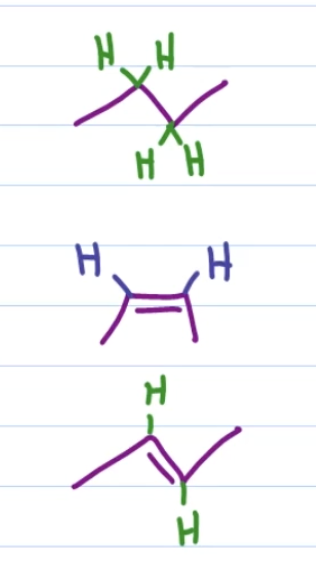
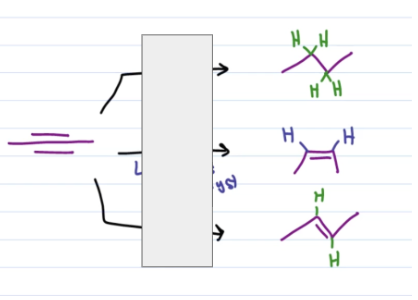
first: H2, pd/c
second: H2, lindlar’s catalyst
third: Na, NH3 (l)
Gringards reagent can also pop open epoxides, what determines major and minor?
Sterics, nucleophile will attach itself to the least substituted carbon