electron transport, oxidative phosphorylation-BC2
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
aerobic cellular respiration
oxygen-coupled breakdown of food molecules and the production of ATP
cells uptake O2 and release CO2
3 major stages
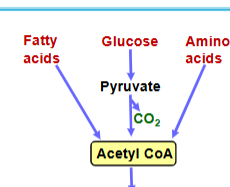
stage 1 of aerobic cellular respiration
oxidation of glucose, fatty acids, and amino acids to yield acetyl coA
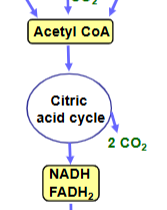
aerobic cell resp. stage 2
Oxidation of acetyl groups in citric acid cycle produces NADH, FADH2, and CO2
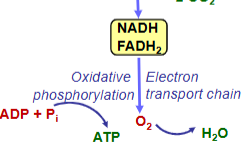
cell resp. stage 3
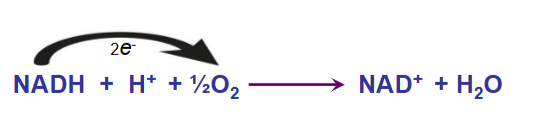
Electrons from NADH and FADH2 are transferred via electron transport chain to oxygen by which O2 is reduced to H2O and ATP is produced
fuel molecules + O2 > CO2 + H2O + ATP
enzymes help this reaction proceed
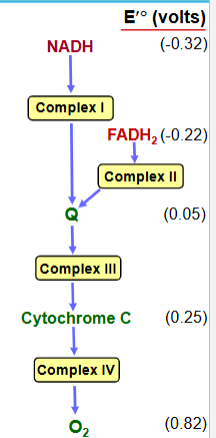
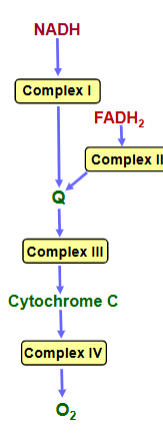
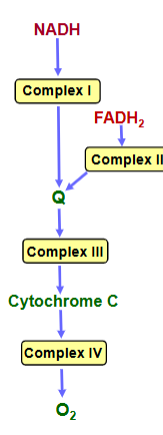
Overview of Membrane-associated Electron Transport
-Four protein complexes (I-IV) involved in electron transport are found in the inner mitochondrial membrane
-Electrons from NADH or FADH2 are transferred via a series of electron carriers to molecular oxygen to produce water
-Spontaneous flow of electrons through complexes I, III, and IV is coupled to ejection of H+ from the mitochondrial matrix
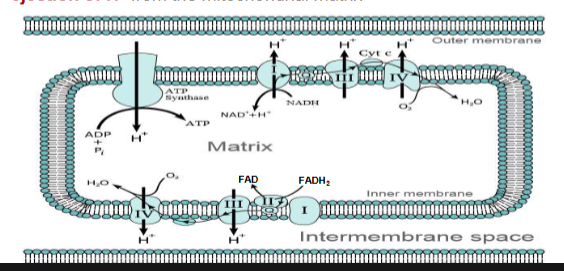
electron flow through complexes 1-4 (membrane-associated electron transport)
-Electrons flow through the electron transport components in the direction of increasing reduction potentials
-NADH (strong reducing agent)
-O2 (terminal oxidizing agent)
complexes 1-4: coenzymes (membrane-associated electron transport)
Mobile coenzymes: ubiquinone (Q) and cytochrome c serve as links between the complexes
complex 4 (membrane-associated electron transport)
-Complex IV reduces O2 to water
reduction potential
measure (in volts) of the affinity for electrons
standard reduction potential is established under standard conditions (1 M of each species, pH=7), its unit is volt
reduction potential: The more positive the value,
the more likely the molecule is to accept electrons, or serving as the oxidant
reduction potential: electrons flow from
lower to higher reduction potential
or they flow in the direction of increasing reduction potential
Four complexes of the electron transport chain function in order of
increasing reduction potential
(E’), because electrons tend to flow carriers of lower E’ to carriers of higher E’
mobile coenzymes: electron transport
ubiquinone (Q) and cytochrome c serve as links between the complexes
electron transport: oxygen
Oxygen is the ultimate acceptor of electrons from NADH and FADH2. Thus, oxygen is reduced to H2O
electron transport: free energy
A large amount of free energy is liberated in the process and it is used to drive the phosphorylation of ADP to ATP
directionality of electron transport chain: photo
NADH: NADH > Complex 1 > Q > Complex 3 > Cytochrome c > Complex 4 > O2
FADH2: FADH2 > Complex 2 > Q > Complex 3 > Cytochrome c > Complex 4 > O2
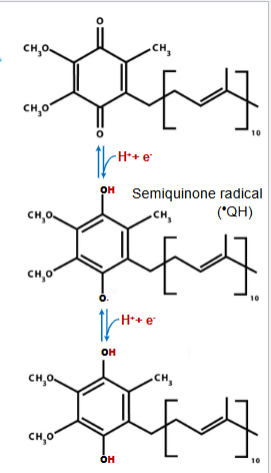
ubiquinone (Q)
-Ubiquinone (also called coenzyme Q, or Q) is a lipid-soluble molecule with a long isoprene side chain
-Because Q is small and hydrophobic, it is freely diffusible within the inner mitochondrial membrane
-Q can accept one electron to become the semiquinone radical (*QH) or two electrons to form ubiquinol (QH2)
cytochromes (characteristics)
-Cytochromes are proteins with characteristic strong absorption of visible light, due to their iron-containing heme prosthetic groups
-The iron is bonded to 4 nitrogen atoms of the porphyrin ring of heme
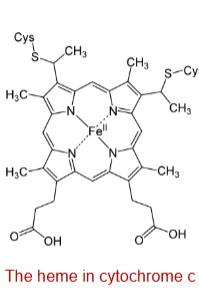
cytochromes: iron
The iron in cytochromes can undergo reversible one-electron oxidation-reduction (Fe2+ / Fe3+ + e-)
classes of cytochromes
a, b, c
Cytochromes a and b are integral proteins of the inner mitochondrial membrane
Cytochrome c is a small, water-soluble protein
complex 1
-Complex I, also called NADH:ubiquinone oxidoreductase, is the largest component of the inner membrane.
-It contains some 42 subunits, among them are a flavoprotein, several iron-sulfur (Fe-S) proteins
complex 1 catalyzes the…
-transfer of electrons from NADH to Q (ubiquinone)
NADH + H+ + Q > NAD+ + QH2
-Some of the energy released in this reaction is used to move H+ from the matrix into the intermembrane space
-Four H+ are pumped from the matrix into the intermembrane space
iron sulfur proteins
-Iron-sulfur proteins are electron-transfer proteins containing one or multiple iron-sulfur centers
-The electron carrier in Fe-S proteins is the iron ion
-Fe-S proteins participate in one-electron transfers in which 1 iron atom of the Fe-S center is oxidized or reduced (Fe3+ or Fe2+)
-Present in complexes I, II, and III
complex 2
Complex II, also called succinate dehydrogenase, consists of the citric acid cycle enzyme succinate dehydrogenase and two iron-sulfur proteins
complex 2 oxidizes…
-FADH2 to FAD. The oxidizing agent is ubiquinone (Q), which is reduced to QH2
FADH2 + Q > FAD + QH2
-Complex II does not contribute to proton gradient, but supplies electrons from succinate
complex 3
Complex III, also called ubiquinone:cytochrome c oxidoreductase, contains 11 subunits, including cytochrome b, cytochrome c1, and Fe-S proteins
complex 3 delivers…
-electrons from QH2 (through cytochrome b, Fe-S centers, cytochrome c1) to cytochrome c (Cyt c)
-Four H+ are pumped from the matrix into the intermembrane
space
-QH2 + 2Cyt c (oxidized) > Q + 2H+ + 2Cyt c (reduced)
complex 4
Complex IV, also known as cytochrome oxidase, carries electrons from cytochrome c to molecular oxygen, reducing it to H2O
complex 4 reaction
2 Cyt c (reduced) + 2 H+ + ½ O2 > 2 Cyt c (oxidized) + H2O
-Electron transfer goes from cytochrome c to a copper-thiol center and finally to O2, which in turn is converted to H2O
-The energy of the reaction pumps two protons outward into the intermembrane space for each pair of electrons that passes through
electron transport chain: NADH
-Complex I (NADH:ubiquinone oxidoreductase): pumps out 4 H+
NADH + H+ + Q > NAD+ + QH2
-Complex III (ubiquinone cytochrome c oxidoreductase): pumps out 4 H+
QH2 + 2Cyt c (oxidized) > Q + 2H+ + 2Cyt c (reduced)
-Complex IV (cytochrome oxidase): pumps out 2 H+
2Cyt c (reduced) + 2H+ + ½O2 > 2Cyt c (oxidized) + H2O
electron transport chain for FADH2
-Complex II (succinate dehydrogenase)
FADH2 + Q > FAD + QH2
-Complex III (ubiquinone cytochrome c oxidoreductase): pump out 4 H+
QH2 + 2Cyt c (oxidized) > Q + 2H+ + 2Cyt c (reduced)
-Complex IV (cytochrome oxidase): pump out 2 H+
2Cyt c (reduced) + 2H+ + ½O2 > 2Cyt c (oxidized) + H2O
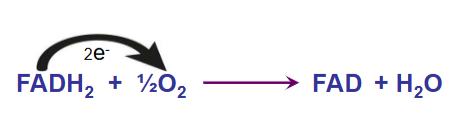
four complexes of the electron transport chain: ubiquinone Q
-diffuses in the mitochondrial membrane, shuttling electrons from complex I to III
-Q also carries electrons from FADH2 generated from the succinate dehydrogenase reaction thus Q can shuttle electrons from complex II to III
four complexes of the electron transport chain: cytochrome C
-small mobile protein that shuttles electrons from complex III to IV
-Cytochrome c oxidase catalyzes the reduction of O2 to H2O
electron transport inhibitors: rotenone
blocks electron transfer in complex 1, causes paralysis
common rat poison
electron transport inhibitors: antimycin A
(used as a fungicide, insecticide, and miticide) blocks e- transfer in complex III
electron transport inhibitors: cyanide or carbon monoxide
block e- transfer in complex 4
inhibitors of electron transport
These inhibitors are extremely toxic because they block e- transport and inhibit
ATP synthesis (electron transfers provide most of the energy for living things)
oxidative phosphorylation
enzymatic phosphorylation of ADP to form ATP coupled to electron transfer from a reducing molecule (NADH or FADH2) to molecular oxygen
Oxidative phosphorylation is the enzymatic phosphorylation of ADP to form ATP as a result of the
transfer of electrons from NADH and FADH2 to O2 by a series of electron carriers
oxidative phosphorylation: Electrons from FADH2 and NADH are transferred in a series of
oxidation-reduction reactions from one complex to the next until they reach O2
O2 is reduced to H2O
oxidative phosphorylation: As a result of electron transport,
-protons are pumped to the intermembrane space and thus an electrochemical potential across the inner membrane is produced
-This electrochemical potential drives ATP synthesis
chemiosmotic theory
-Proposed by Peter Mitchell in the 1960’s (Nobel Prize 1978)
-Electron transport and ATP synthesis are coupled by a proton gradient across the inner mitochondrial membrane
chemiosmotic theory: proton motive force
-The proton gradient generated by the oxidation of NADH and FADH2 is called the proton-motive force
-The proton-motive force powers the synthesis of ATP
-Oxidation (electron transferring) and phosphorylation of ADP to ATP are coupled through the proton motive force
oxidative phosphorylation: Electrons are carried by NADH and FADH2 to the
mitochondria, where they then pass through a chain of electron carriers in the inner membrane
-The flow of electrons is accompanied by proton transfer across the inner membrane, producing a proton gradient
-As the protons travel from the intermembrane space to the matrix, they provide energy for phosphorylating ADP to form ATP (oxidative phosphorylation)
ATP synthase forms
ATP from ADP and phosphate during oxidative phosphorylation
ATP synthase has two distinct components:
F0 and F1
F0
spans inner mitochondrial membrane and makes up the H+ channel
F1
projects into the matrix and contains the catalytic subunits for ATP synthesis
ATP synthase: each of the 3 beta subunits has
one catalytic site for ATP synthesis
ATP synthase visual
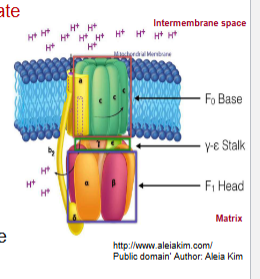
ATP synthase: The c-e-y unit forms a
rotor and H+ passage through the F0 channel causes the rotor to spin in one direction
rotation induces structural changes in the beta subunit that results in the synthesis and release of ATP from the enzyme
ATP synthase: Proton gradient powers the synthesis of
ATP (H+ flow through the F0 channel to F1 where ATP is made)
P/O ratio
“ATPs per oxygen” or ATP/1/2O2
-The number of ATP formed in oxidative phosphorylation per two electrons flowing through the electron-transport chain
-The number of ATP formed in oxidative phosphorylation per ½ O2 reduced
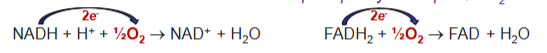
what is required for each ATP produced by ATP synthase?
translocation of three H+
what is needed for transport of Pi, ADP, and ATP across the mitochondrial membrane?
one H+
H+ translocated for each 2e- (for each complex)
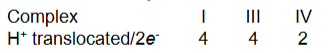
P/O ratio for two electrons from NADH
2.5
P/O ratio for two electrons from FADH2
1.5
total ATP for electron transfer and oxidative phosphorylation (aerobic cell respiration)
32 ATP
fermentation total ATP (anaerobic)
2 ATP