P1.3 - Pressure
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What is Brownian Motion?
constant random motion at high speeds (undergoes sudden change when colliding with container walls and colliding with molecules)
Define Pressure:
force exerted per unit area
Formula for pressure:
P = F/A
Pressure = pa - N/m2
F = N
A = m2
How does temp affect motion of molecules?
As temp of gas increases, average speed of molecules and vice versa
How does temp affect KE?
The hotter the gas, the higher the average kinetic energy
The cooler the gas, the lower the average kinetic energy
How does temp affect pressure?
This means they will collide with the walls more often
This creates an increase in pressure
Therefore, at a constant volume:
an increase in temperature increases the pressure of a gas and vice versa
If gas temp is constant, then how does volume affect pressure?
Compressed – decreases the volume which increases the pressure
Expanded – increases the volume which decreases the pressure
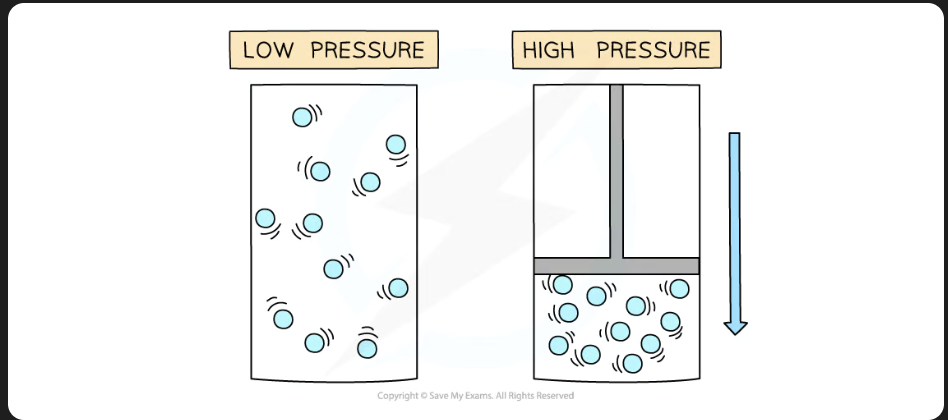
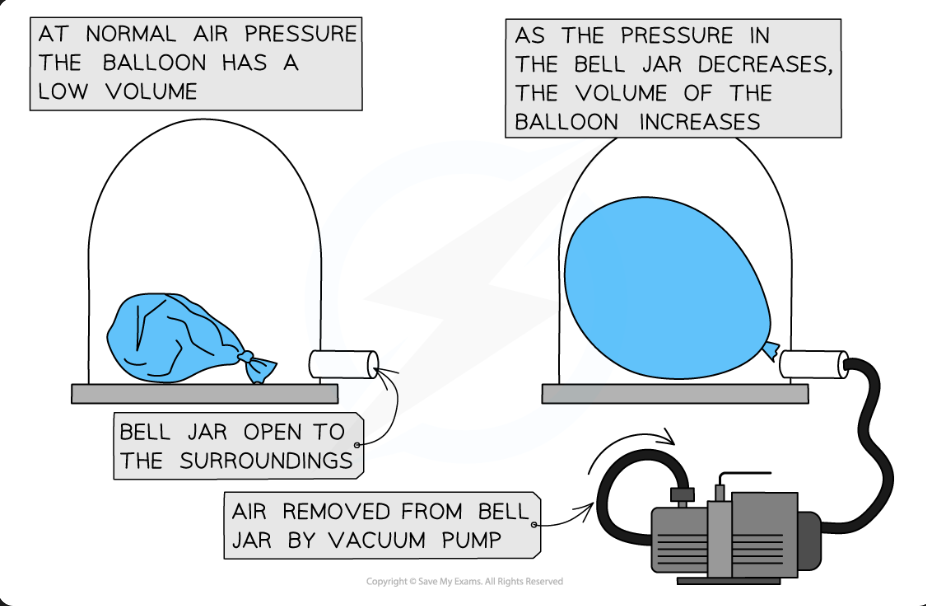
How can a change in pressure cause a change in volume ? (balloon example)
If air is removed from bell jar vacuum, pressure decreases in bell jar (since air is removed) and this increases the Balloon’s volume.
KEY: Less air = less pressure, No air = No pressure
How can gas being compressed (less volume), lead to greater pressure ?
If gas is compressed, molecules will hit wall of container more frequently , this creates a larger overall net force on the walls which increases the pressure
How does pressure produce a force on a surface?
Gas particles move about randomly they collide with container’s net walls, these collisions produce a net force at 90o to wall of the container
Hence gas at high pressure has more frequent collisions with container at higher force.
So higher the pressure, the higher the force exerted per unit area
What happens when gas compresses to volume ?
Volume decreases, because density of gas increases , because container size decreases but number molecules stays same
Allows more frequent collisions of molecules on container wall
So they hit wall with more force and therefore increase pressure
same steps but opposite when gas expands which means volume increases
Summarise relationship between pressure and volume:
When the volume decreases (compression), the pressure increases
When the volume increases (expansion), the pressure decreases
key assumption is that the temperature and the mass (and number) of the particles remains the same
The 2 are inversely proportional
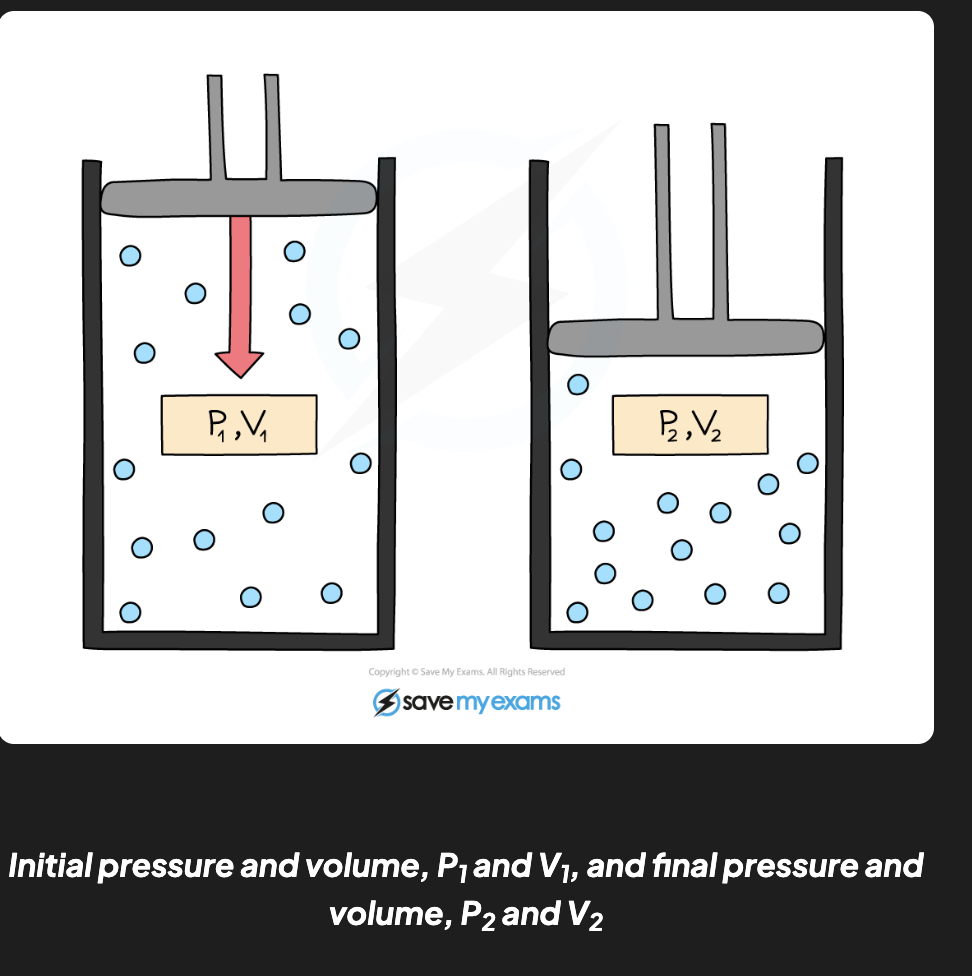
Comparing pressure and volume formula: (used in calculations)
P1V1 = P2V2
P1 = initial pressure in pascals (Pa)
V1 = initial volume in metres cubed (m3)
P2 = final pressure in pascals (Pa)
V2 = final volume in metres cubed (m3)
Define Work:
Transfer of energy by force
What’s the relationship between work done and gas compression /expansion ?
Work can be done on a gas by compression
Gas will do the work instead if allowed to expand
Doing work on a gas involves transfer of energy (increase in internal energy can cause increase in temp)
Explain how the work done by gas and vice verse actually works:
When compressed, a force is needed to compress gas so the force does the work - hence work is done on the gas causing the temp to rise
When a gas expands it does the work itself which does cause gas to lower its internal energy which causes a lower temperature
How does a bicycle pump cause an increase in temp?
If hand is placed over pump hole, and quickly compressed several times, the pump will feel very warm.
because work is done on a gas (force from hand to compress) it causes temp to increase
How does a fire extinguisher cause a decreasing temperature ?
When pressurised carbon dioxide is released from a high pressure cylinder, gas does the work so it loses energy
which causes CO2 to freeze (lowers temp) forming dry ice
Relationship between temperature and work done (gas):
Work done on a gas (compresses) - temp falls
Work done by a gas (expands) - temp increases
Why does an object experience more pressure underwater ?
It experiences atmospheric pressure
It experiences the liquid pressure aswell
(Both are sources of fluid pressure)
What is a fluid?
A substance that can flow and take the shape of its container (usually liquid or gas)