Chap 23A - Carboxylic acid
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
State general formula, functional group and lactic acid formula
General formula of monocarboxylic acids: CnH2n+1COOH
Functional group: carboxyl group
Example: 2-hydroxypropanoic acid (lactic acid)
Describe bp of carboxylic acid + why less volatile than alcohols
Lower members are liquids with pungent odours
Less volatile (high bp) than alcohols of similar Mr
Carbo has stronger HB than alcohols as -OH bond is more polarised due to the presence of electron–withdrawing inductive C=O group
Carbo also dimerise in the liquid state and in non–polar solvents, forming 2 HB between each pair of molecules
In polar solvent: RCO2H ⇌ RCO2– + H+
Describe solubility of carboxylic acid
Lower acids are completely miscible with water because –CO2H can form HB with water
As the length of the non-polar hydrocarbon chain increases -> solubility in water decreases but solubility in non-polar solvent increase
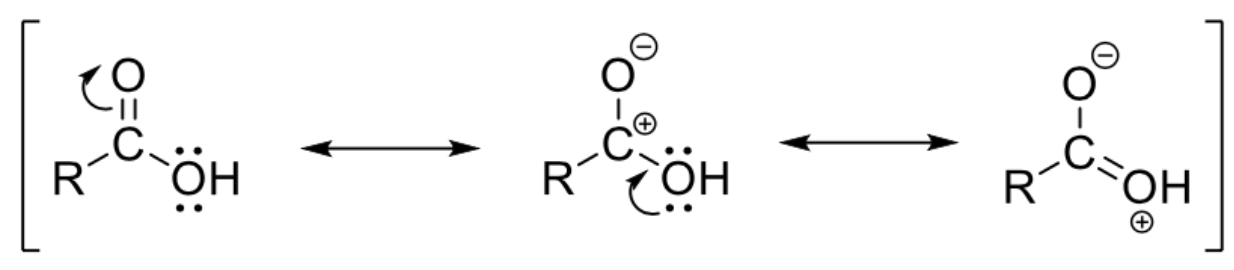
Draw resonance structures of RCOOH and RCO2-
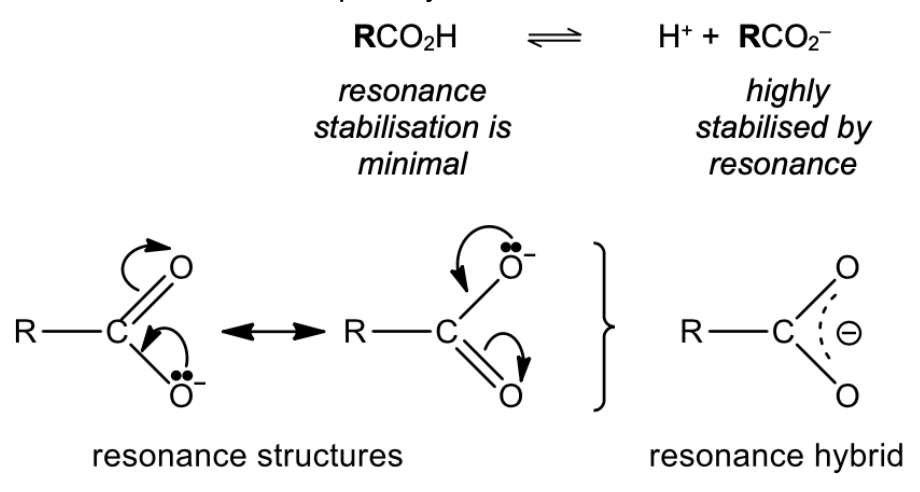
Explain why carboxylate anion is stabalised
Carbo is weak acid, dissociate partially: RCO2H ⇌ RCO2– + H+
C–O bonds are equal in length -> negative charge is equally distributed between the 2 O atoms, leading to a stabilisation of the carboxylate anion
An electron–withdrawing inductive group in RCO2– reduces the electron density of the negative charge on the carboxylate anion by charge dispersal -> stabilises the RCO2– and increases the strength of the acid
Why are carboxylic acids stronger acids than alcohols
An electron–withdrawing inductive group in RCO2– reduces the electron density of the negative charge on the carboxylate anion by charge dispersal -> stabilises the RCO2– and increases the strength of the acid
The RO- ion is destabilised by electron- donating inductive effect of the R group -> negative charge is intensified
The first equilibrium position lies greater to the right compared to the second -> hence RCOOH is stronger acid than ROH
State ways to prepare carboxylic acids
Oxidation of Primary Alcohols and Aldehydes |
Hydrolysis of Nitriles |
Side-chain oxidation of alkylbenzene to benzoic acid |
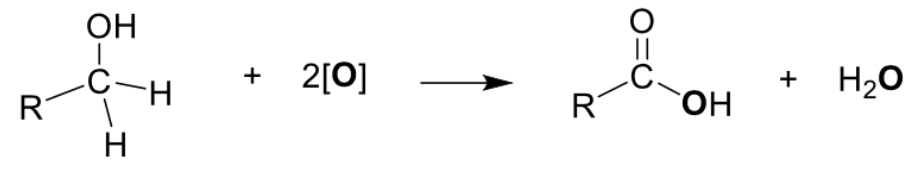
Describe preparation of carboxylic acid (Oxidation of Primary Alcohols and Aldehydes) + equation
Oxidation of alcohol
Type of reaction: Oxidation
Reagents and conditions: K2Cr2O7(aq), H2SO4(aq), heat under reflux OR KMnO4(aq), H2SO4(aq), heat
CH2OH -> COOH
Oxidation of aldehydes
Type of reaction: Oxidation
Reagents and conditions: K2Cr2O7(aq), H2SO4(aq), heat OR KMnO4(aq), H2SO4(aq), heat
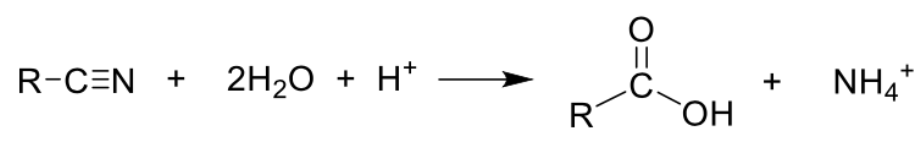
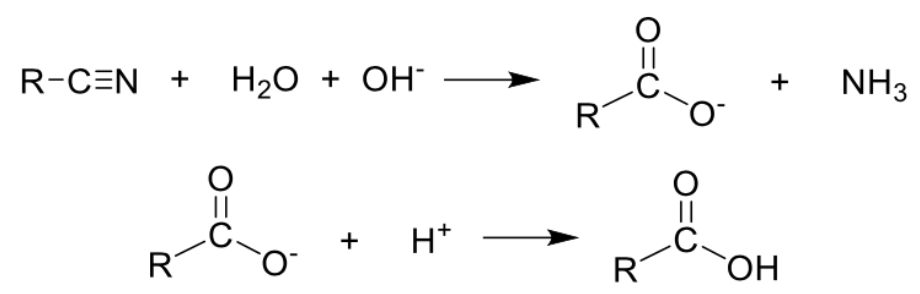
Describe preparation of carboxylic acid (ACID hydrolysis nitrile) + equation
Type of reaction: Hydrolysis
Reagents and conditions: H2SO4(aq), heat
Describe preparation of carboxylic acid (ALKALINE hydrolysis nitrile) + equation
Type of reaction: Hydrolysis
Reagents and conditions: NaOH(aq), heat, followed by H2SO4(aq)
Distinguishing test as NH3 produced turns damp red litmus paper blue
Explain how alkaline hydrolysis can take place via amide (with equations)
Reaction can take place via the amide
RCN + H2O → RCONH2
Same as above:
RCONH2 + H2O → RCO2–NH4+
RCO2−NH4+ + H+ → RCO2H + NH4+
RCO2−NH4+ + OH– → RCO2– + NH3 + H2O
Describe side-chain oxidation of alkylbenzene to benzoic acid
Type of reaction: Oxidation
Reagents and conditions: KMnO4(aq), H2SO4(aq), heat OR KMnO4(aq), NaOH(aq), heat, followed by H2SO4(aq)
Explain why C=O does NOT undergo addition
C=O does NOT undergo addition
Reason: Lone pair of electrons in p orbital in O of -OH group interacts with the p orbital of the carbonyl C atom -> results in an increase in delocalisation of electrons and greater stability
This stability would be lost if a reagent is added to the carbonyl group -> carboxylic acids are more resistant to addition than aldehydes and ketones
Draw delocalisation of electrons in RCOOH