Bio111 Proteins
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
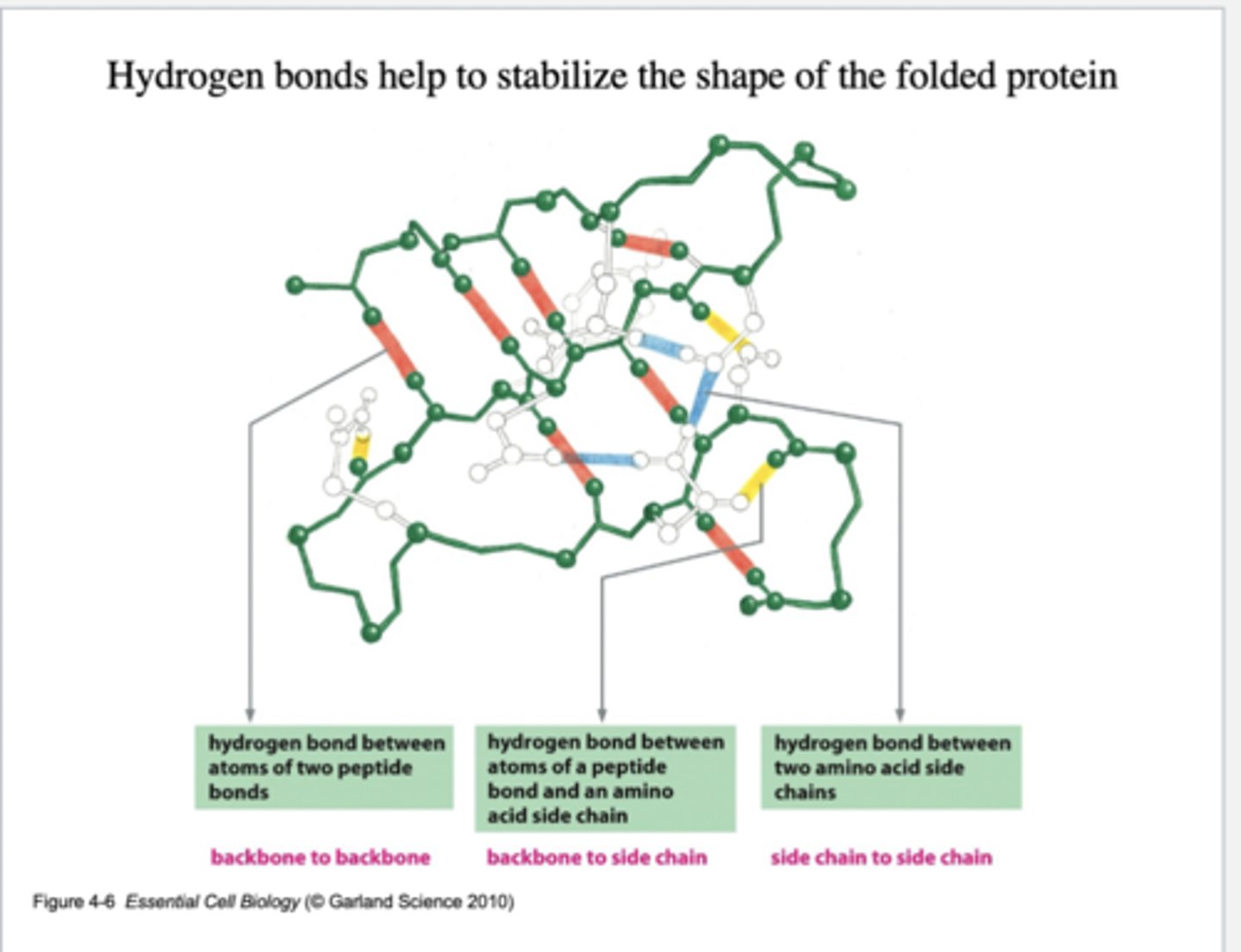
Hydrogen Bond
weak attraction between a hydrogen atom that is part of a polar covalent bond and another electronegative atom like oxygen or nitrogen
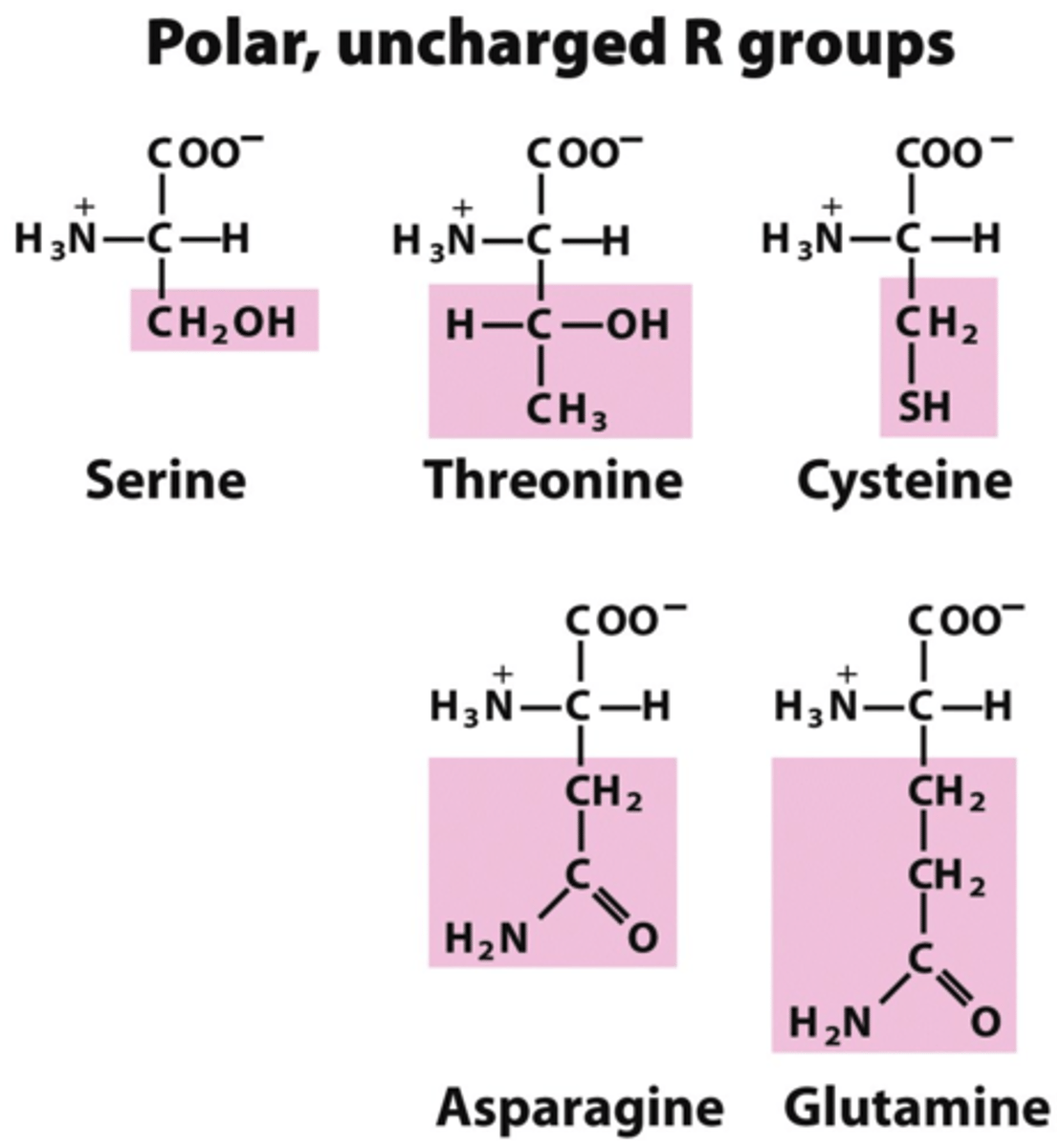
Polar
Molecule with partial charges due to the presence of an electronegative atom like oxygen, nitrogen, phosphorous, or sulfur. Mixes with water.
Acids
substances that release hydrogen ions (H+) when dissolved in water; cause pH to decrease
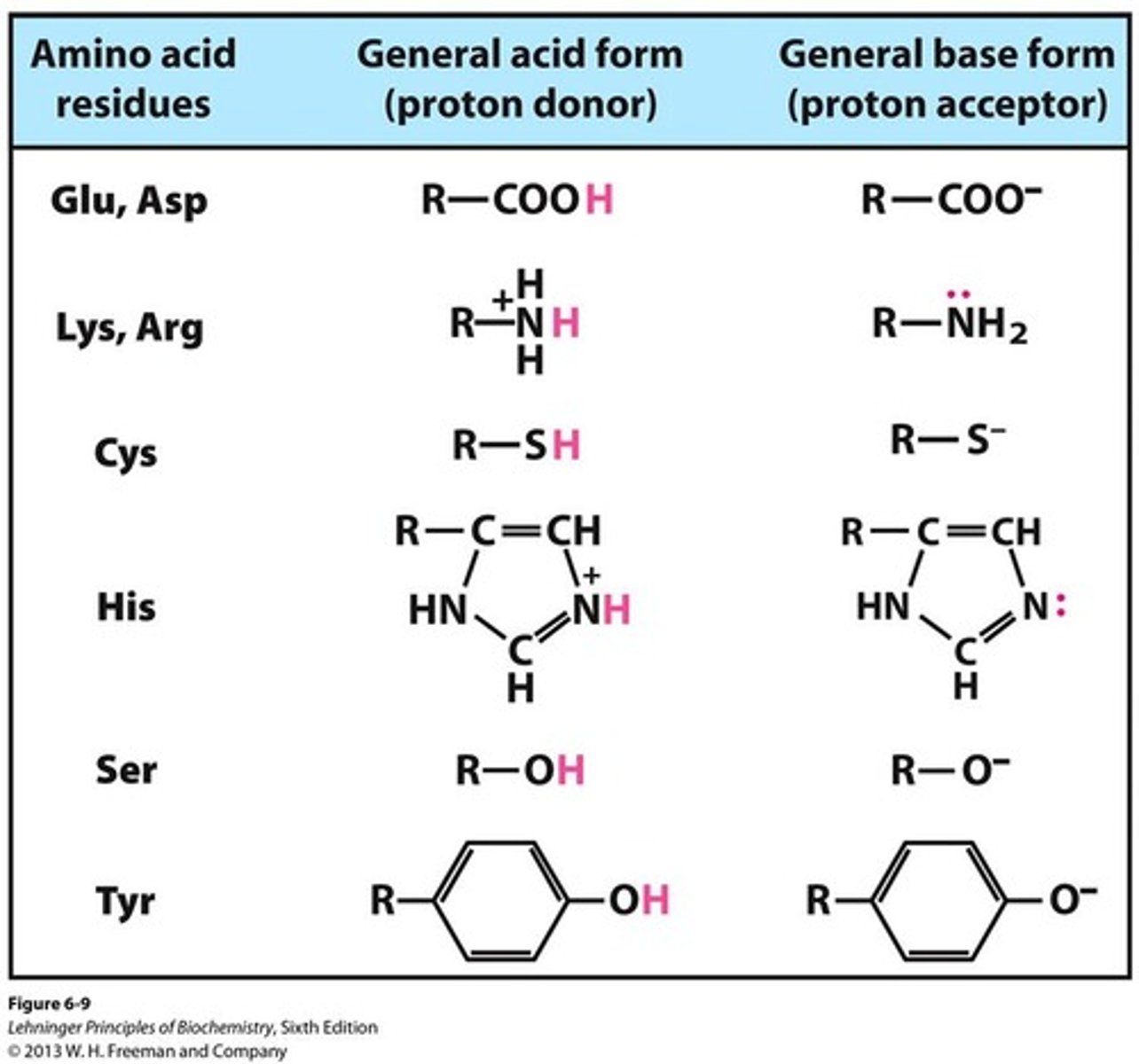
Base
A substance that decreases the hydrogen ion concentration in a solution, while increasing the hydroxide ion (OH-) concentration.
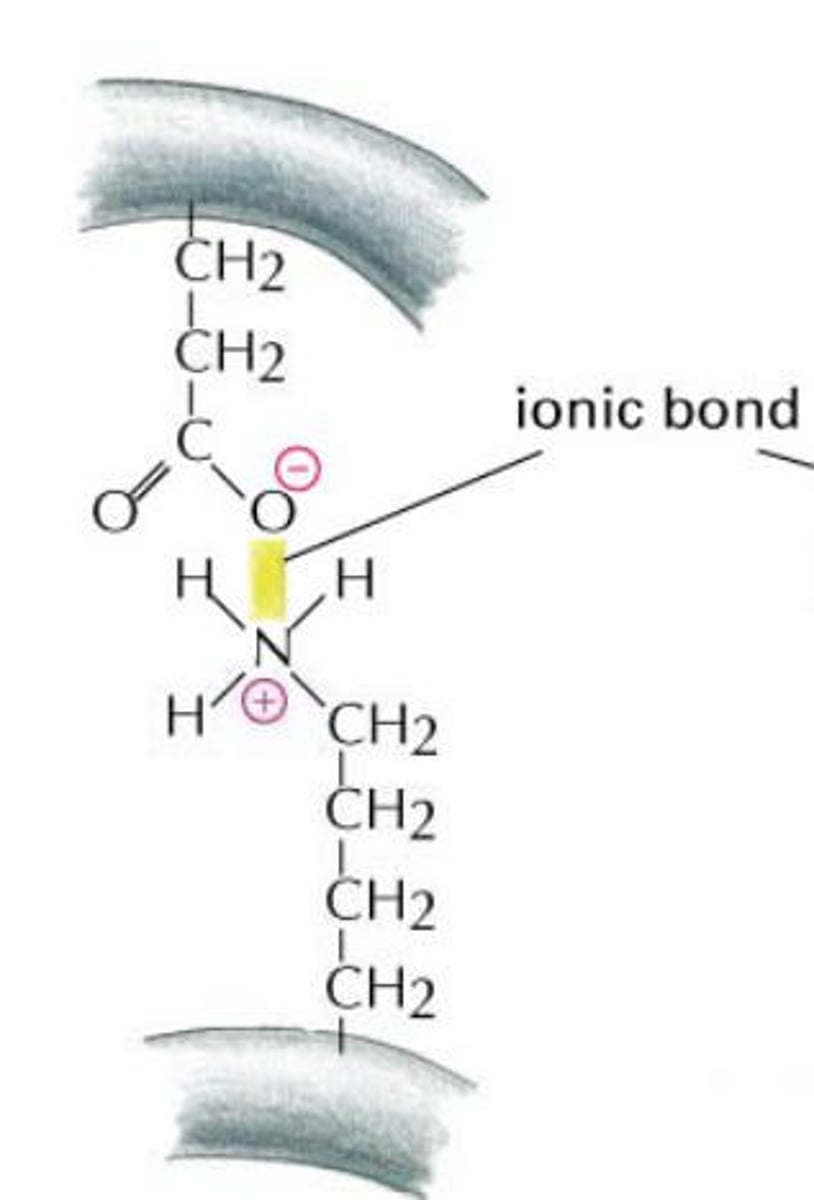
Ionic bond
A chemical bond resulting from the attraction between oppositely charged ions.
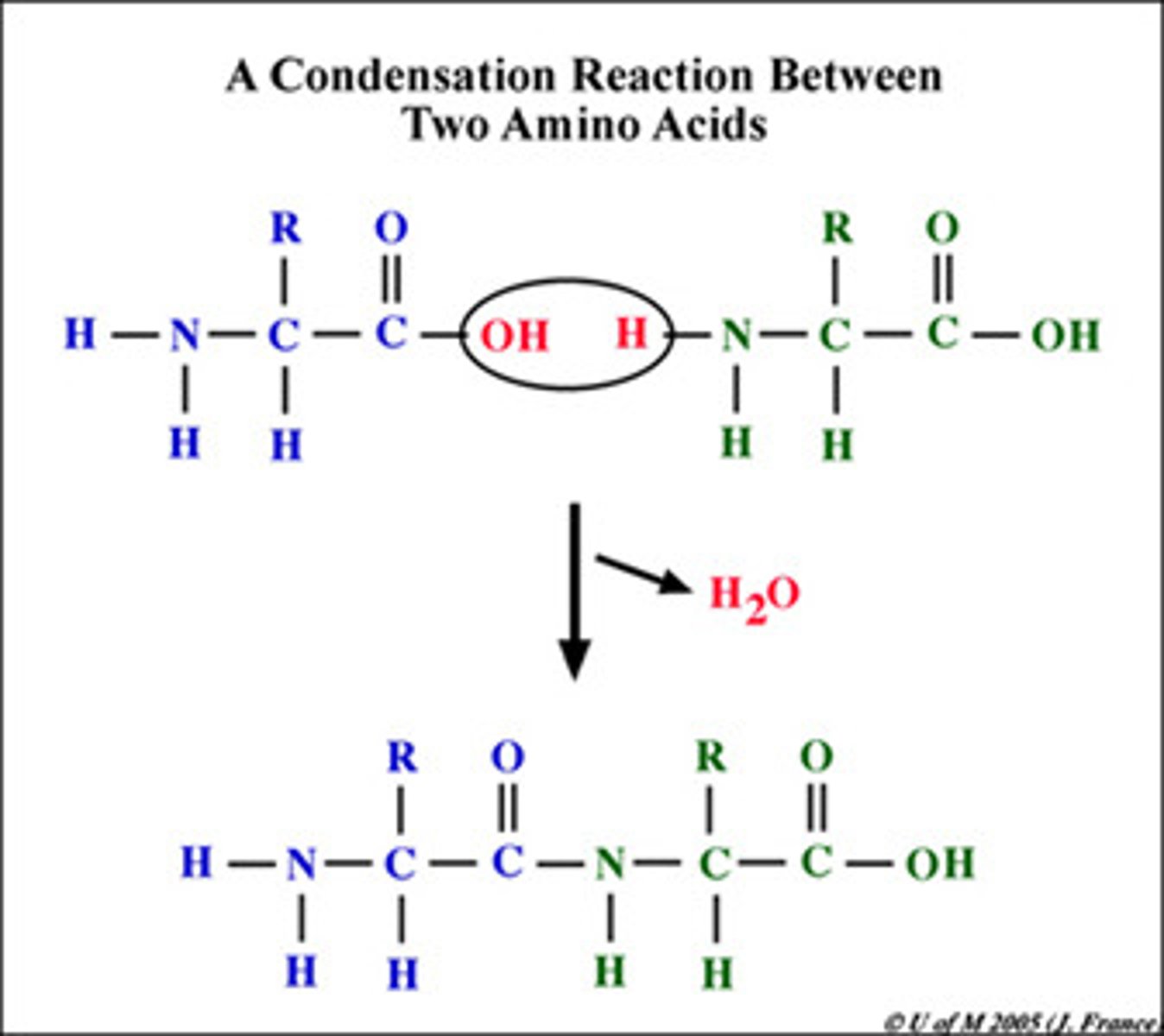
Dehydration Synthesis
A chemical reaction in which two molecules covalently bond to each other with the removal of a water molecule.
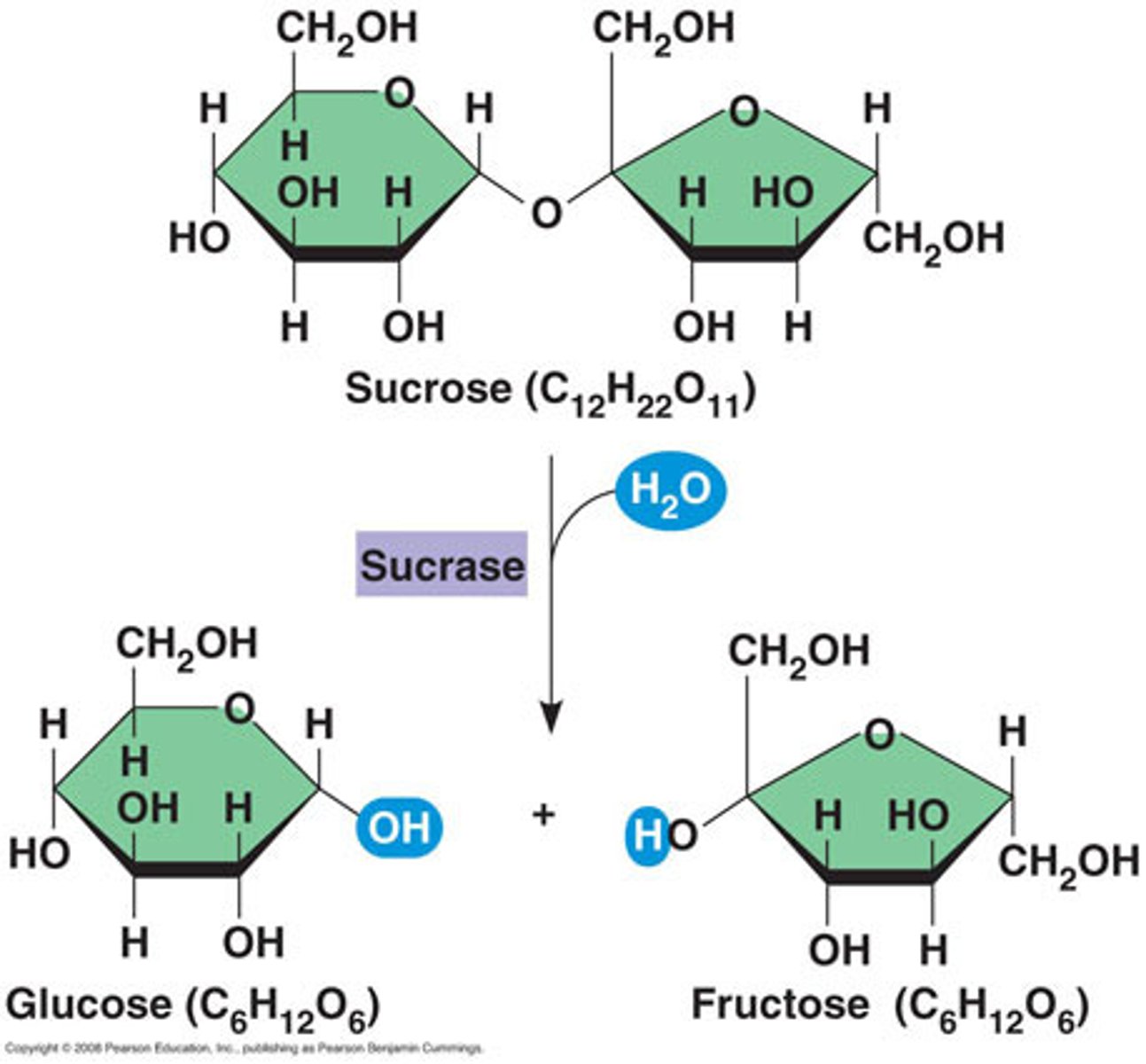
Hydrolysis
A chemical process that splits a molecule by adding water.
Monomer
small chemical unit that makes up a polymer
Polymer
molecules composed of many monomers; makes up macromolecules
Hydrophilic
water loving, polar molecule
Hydrophobic
water hating, nonpolar molecule
Covalent Bond
A chemical bond formed when two atoms share electrons
Protein
macromolecule that made of amino acids that contain carbon, hydrogen, oxygen, and nitrogen; needed by the body for growth and repair
Amino Acid
Building blocks of protein; includes an amino group, carboxyl group, and variable R group
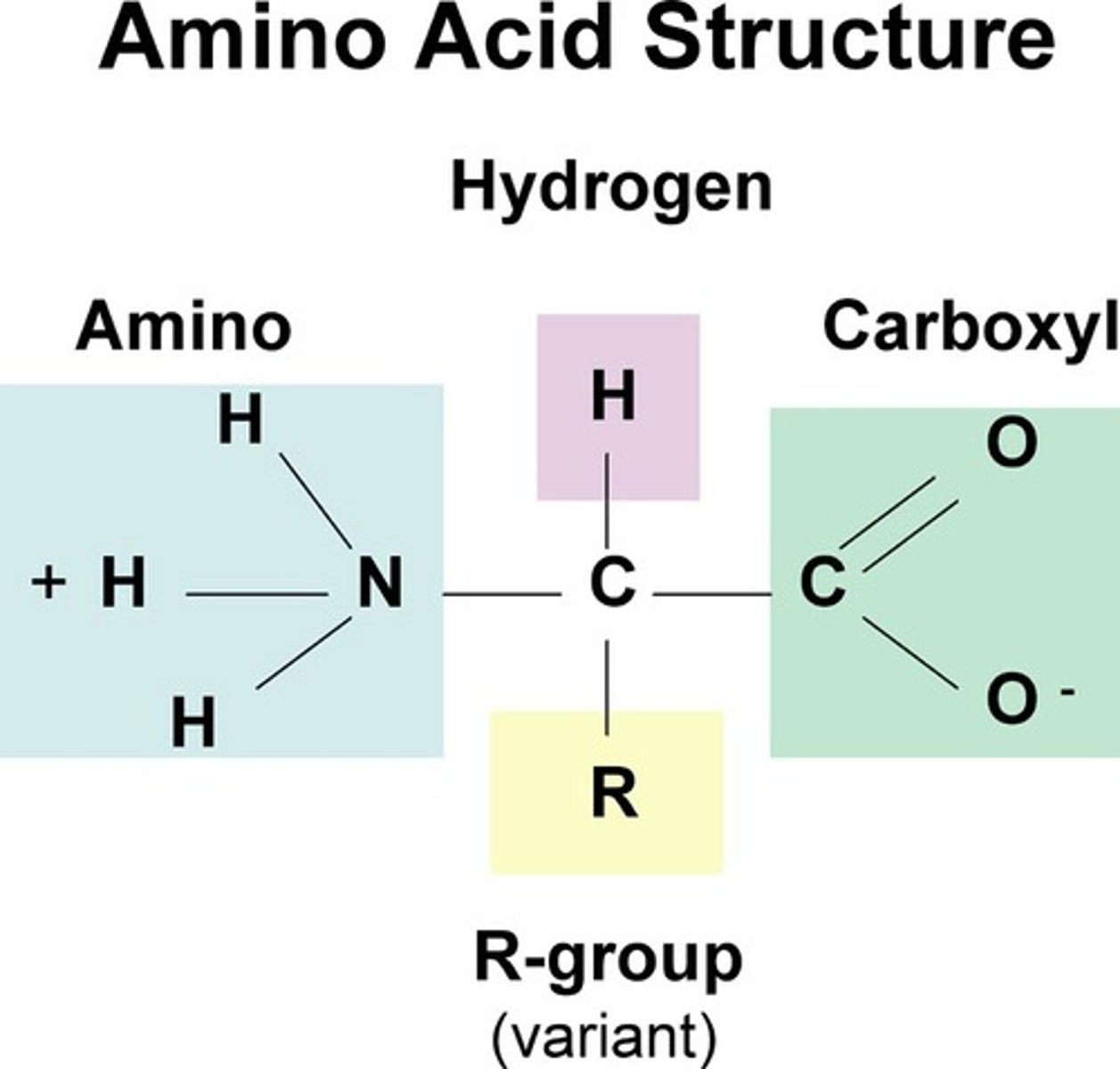
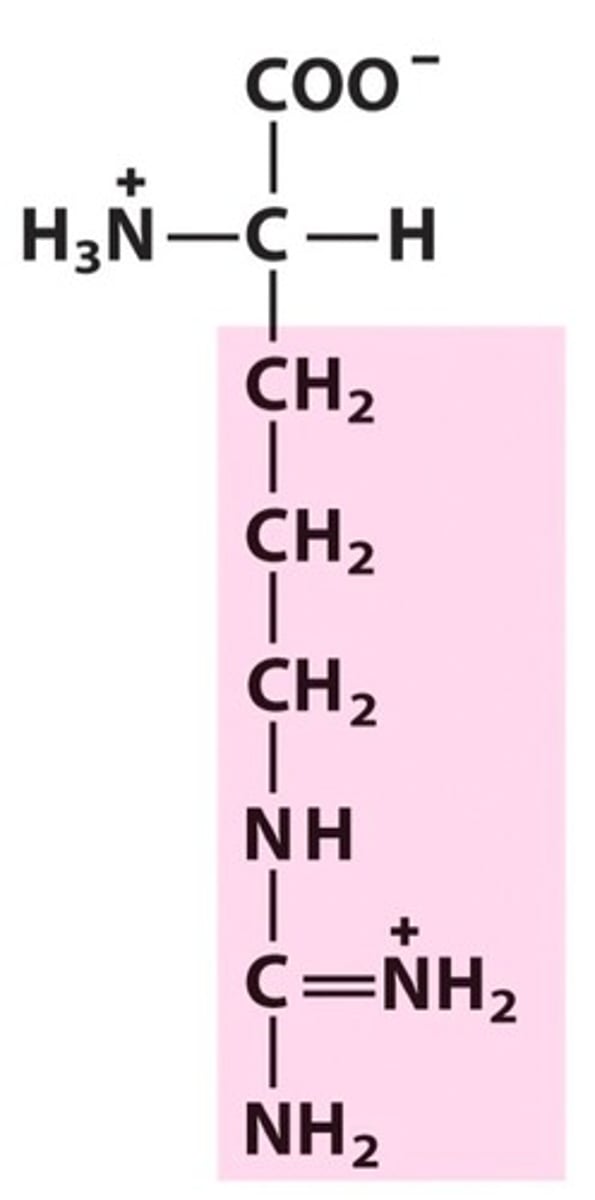
R group
a functional group that defines a particular amino acid and gives it special properties.
Peptide bond
covalent bond formed between amino acids
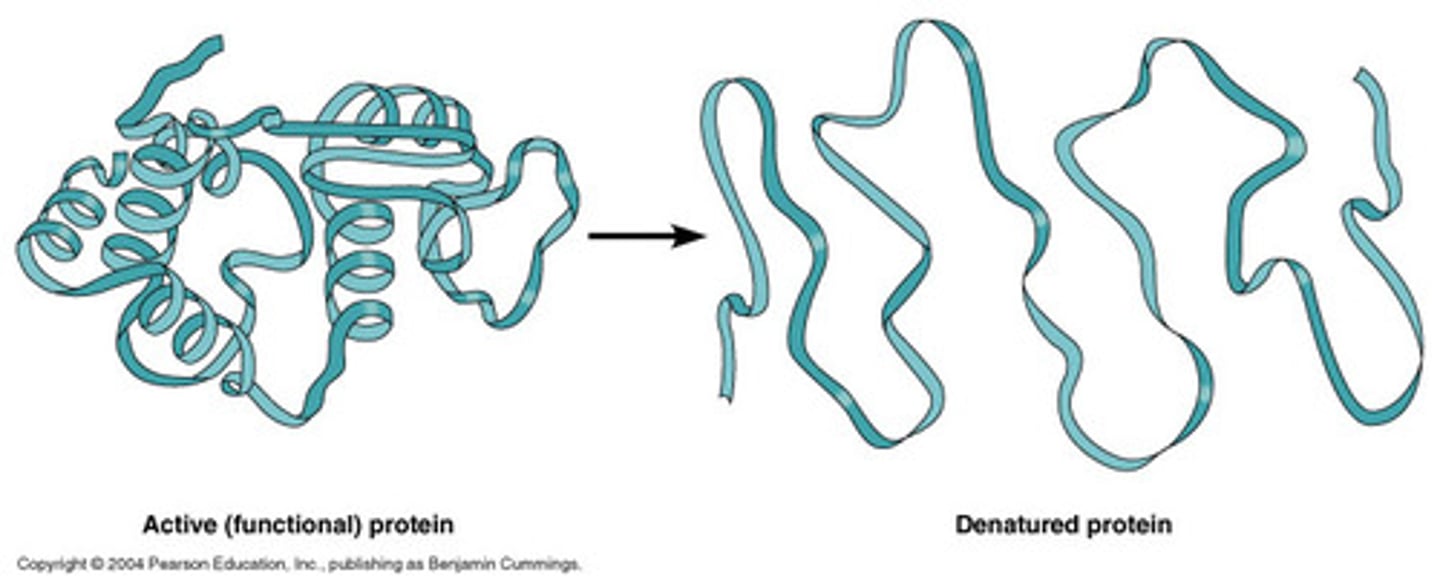
Denaturation
loss of normal shape of a protein due to heat or other factor
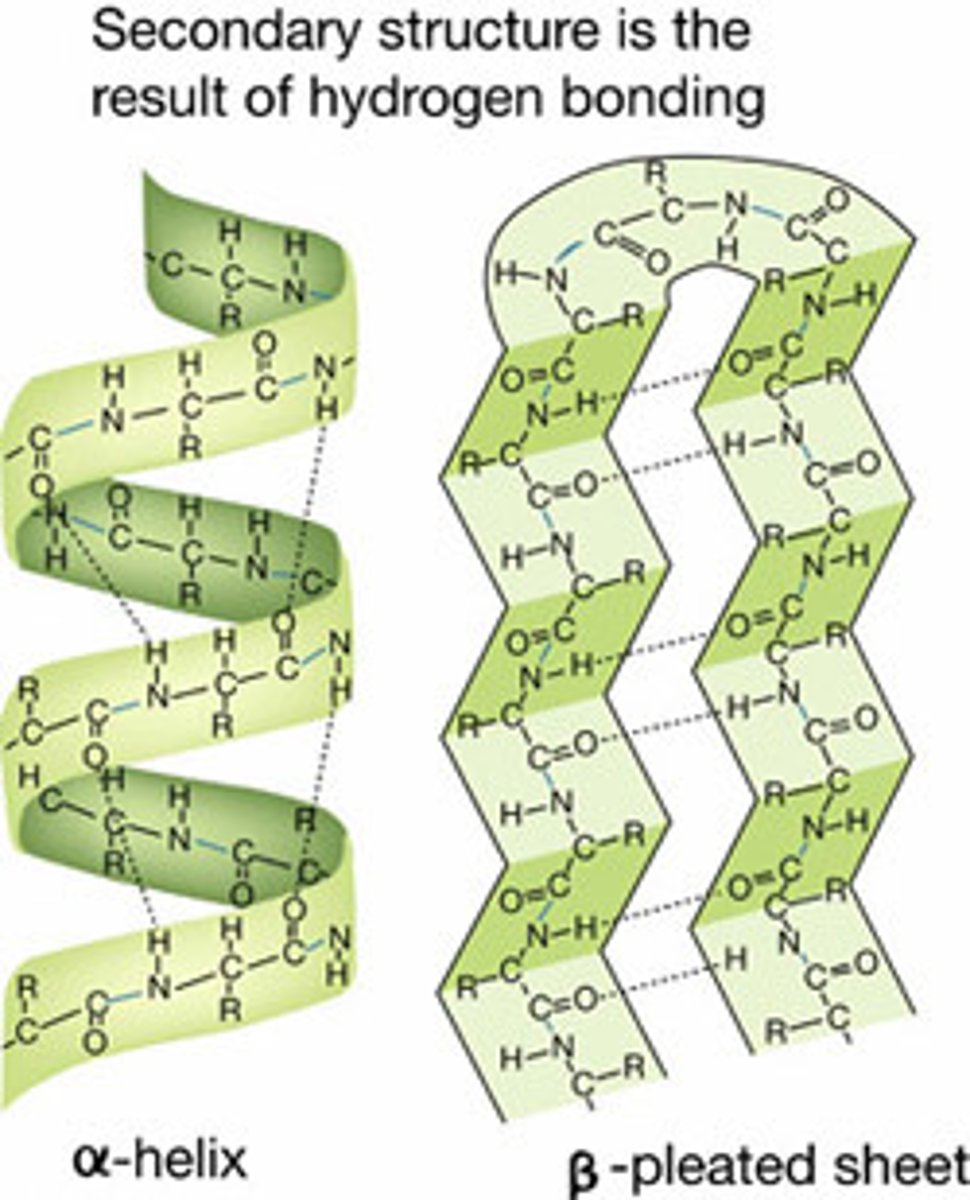
Alpha Helix
A spiral shape constituting one form of the secondary structure of proteins, arising from a specific hydrogen-bonding structure.
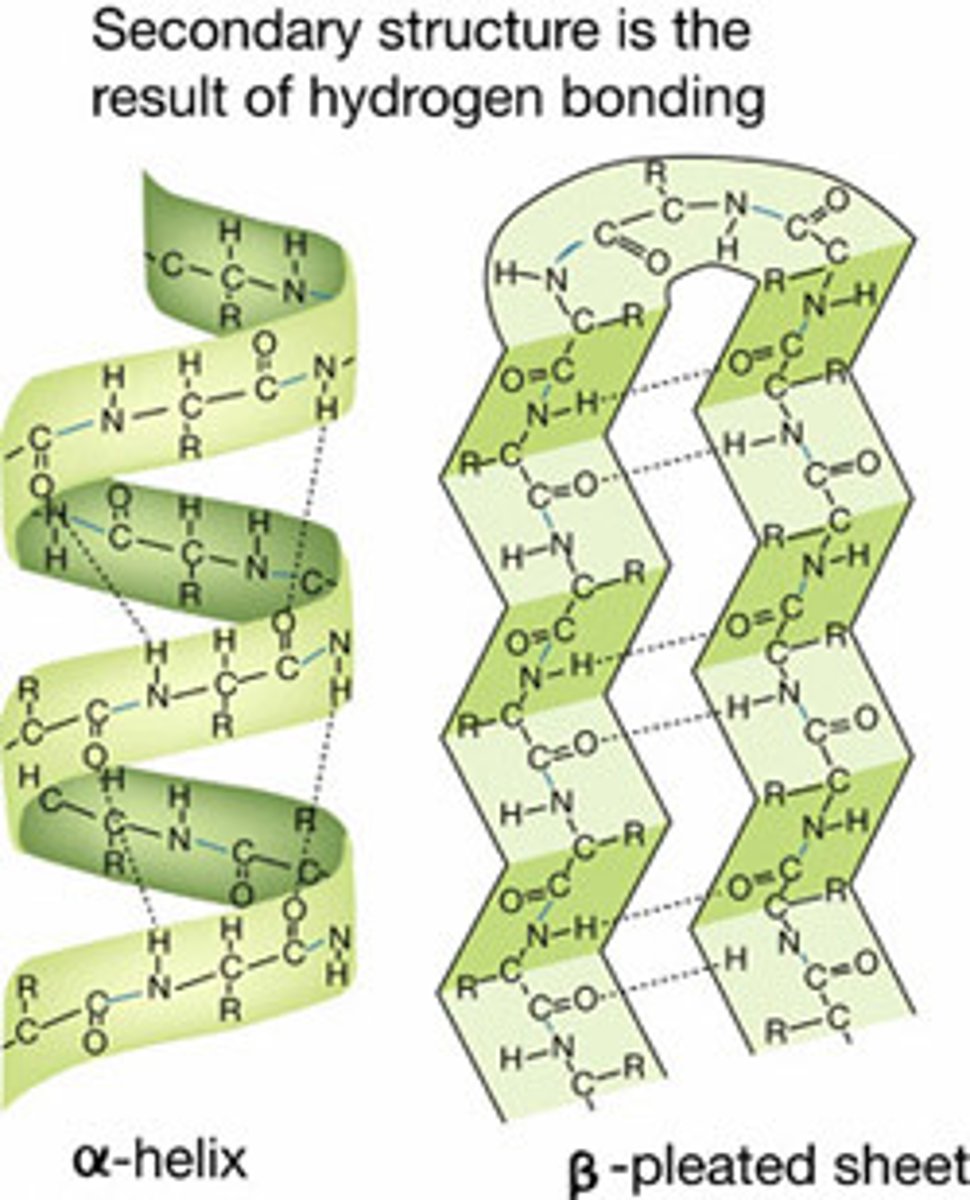
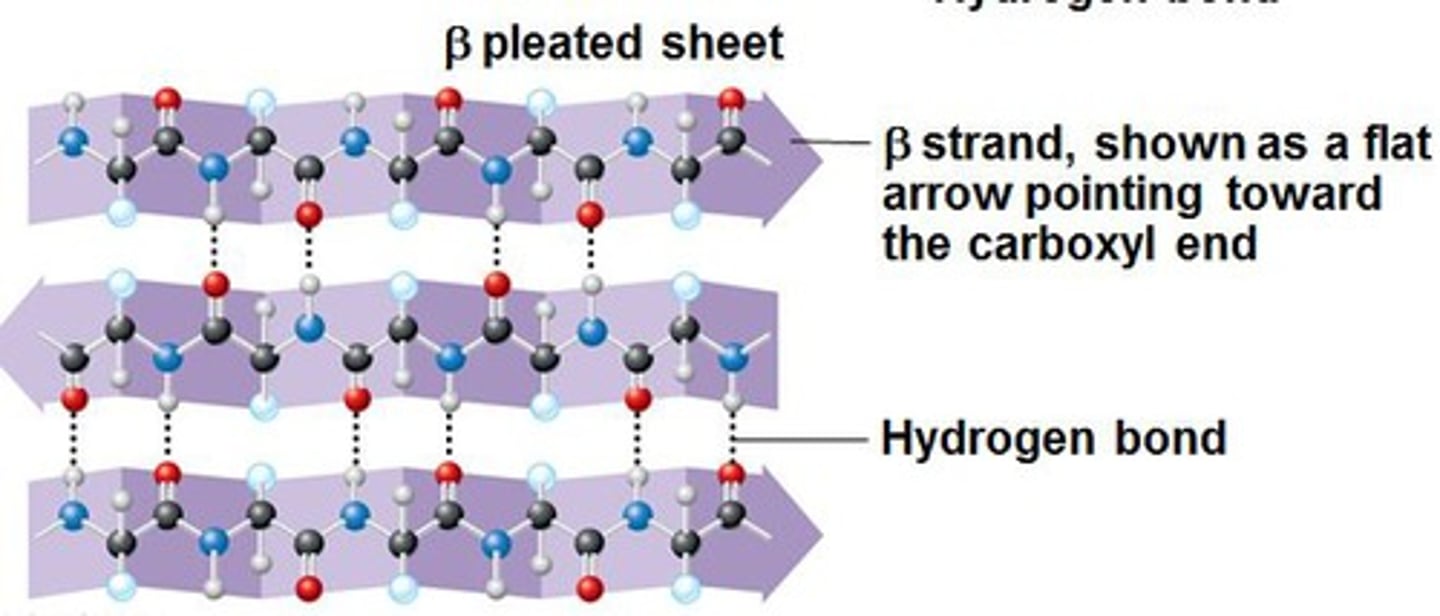
Beta-Sheet
Folding pattern found in the secondary structure of many proteins in which neighboring regions of the polypeptide chain associate side by side with each other through hydrogen bonds to give a rigid, flattened structure.
Polypeptide
long chain of amino acids that makes proteins
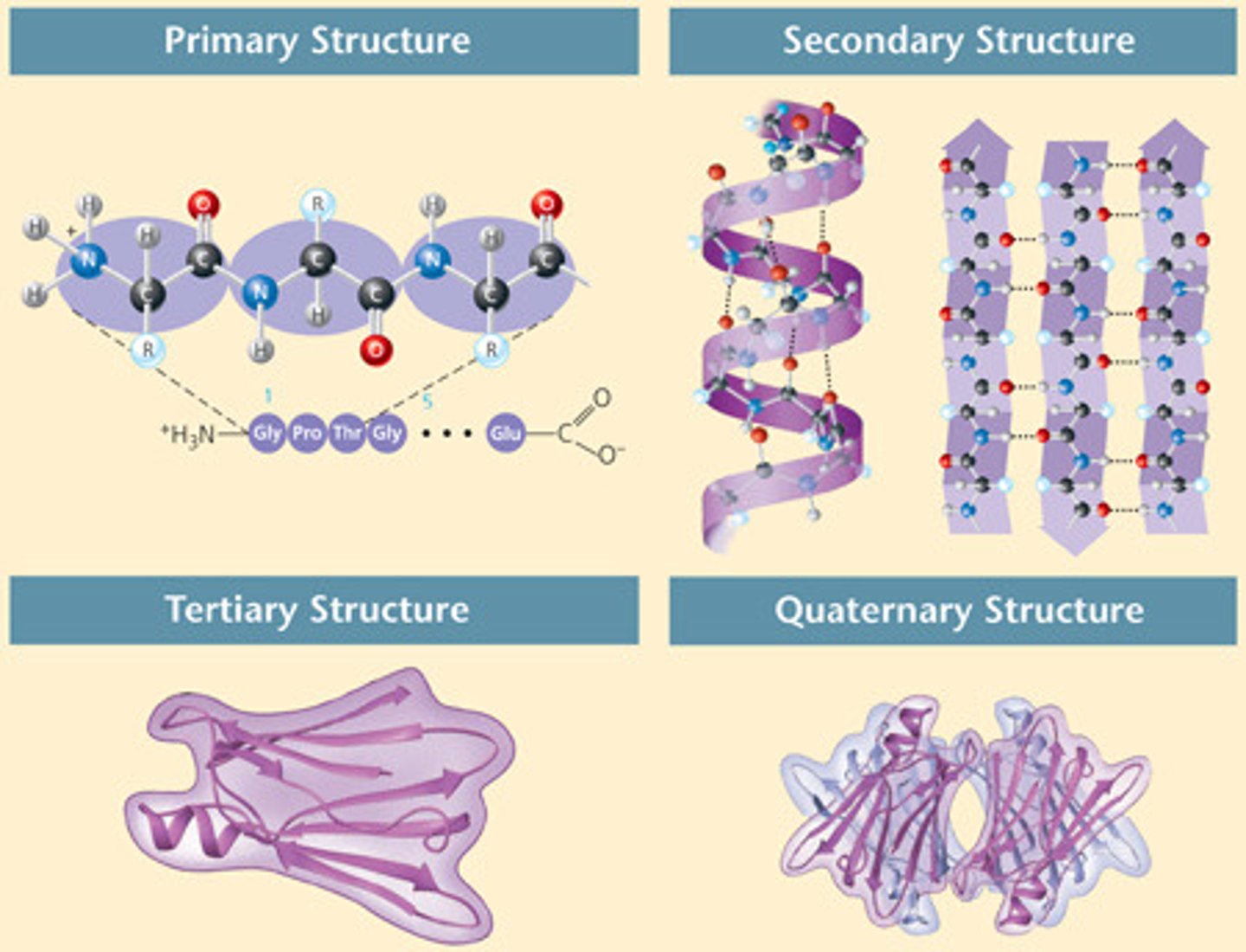
Primary Structure
The first level of protein structure; the specific sequence of amino acids making up a polypeptide chain.

Secondary Structure
the regular local patterns of coils or folds of a polypeptide chain that results from hydrogen bonding between amino and carboxyl groups in the peptide backbone
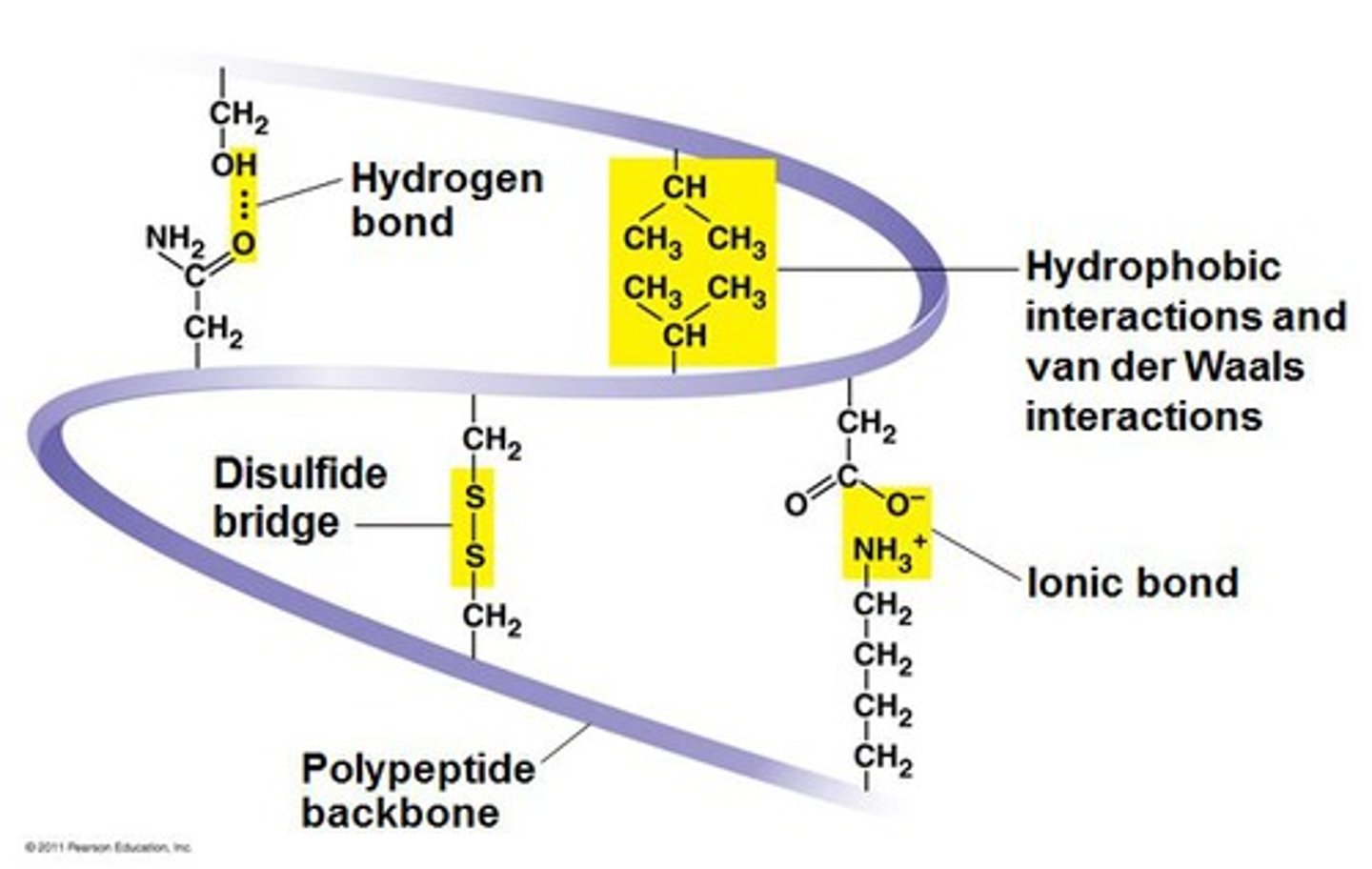
Tertiary Structure
Level of protein folding that results from interactions between side chains, including hydrophobic, covalent, ionic, and hydrogen bonds
Quaternary Structure
Combination of two or more polypeptide subunits to form a fully functional protein