PT AC (Class 1): VTE - Pathophysiology and Pharmacology
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Define thrombus and embolus.
Thrombus
- Blood clot that remains where it was formed
- Final product of the blood coagulation step in hemostasis
- Two major components: Aggregated platelets (form a platelet plug) and mesh of cross-linked fibrin protein
Embolus
- Blood clot or other material (e.g., fat, air, fluid) that travels through the circulation
- Eventually lodges in a smaller vessel, causing a blockage (embolism)
Describe how arterial clots differ from venous clots.
Arterial thromboembolism
- Clot formation in the arterial circulation
- Platelet-rich (“white thrombi”)
- Often due to ruptured atherosclerotic plaques and platelet activation
- Manifestations: MI or stroke
Venous thromboembolism (VTE)
- Clot formation in the venous circulation
- Fibrin- and RBC-rich (“red thrombi”)
- Often due to stasis of blood flow and hypercoagulability
- Manifestations: DVT (rarely fatal) and PE (can be fatal)
- Long-term complications: Post-thrombotic syndrome (PTS) and chronic thromboembolic pulmonary HTN (CTPH)
How does Virchow's Triad explain the pathogenesis of DVT and PE?
1. Venous stasis: Slowed or stagnant blood flow
- Immobilization
- Surgery (especially general anesthesia)
- Damage to venous valves
- Obesity
2. Endothelial injury: Damage to inner lining of blood vessels
- History of DVT/PE
- Major orthopedic surgery
- Trauma (e.g., fractures of pelvis, hip, or leg)
- Indwelling catheters
3. Hypercoagulability: Increased tendency for blood to clot
- Obesity/diabetes
- Cancer
- Clotting factor changes (e.g., inheritable mutations, overexpression)
- Pregnancy
- Drugs (e.g., estrogen-containing contraceptives)
What is hemostasis?
- Process responsible for maintaining circulatory system integrity following blood vessel damage
- Prevents excessive bleeding upon blood vessel damage
Outline the steps leading to platelet aggregation.
Vascular injury exposes endothelium, triggering the process of primary hemostasis (platelet plug formation)
Adhesion: Platelets adhere to exposed collagen primarily via Von Willebrand factor (vWF), which acts as “molecular glue”
Activation/aggregation: Platelets change shape and release factors that promote platelet aggregation, such as…
- Adenosine diphosphate (ADP)
- Serotonin (5-HT)
- Thromboxane A2 (TXA2)
Results in formation of an unstable platelet plug (i.e., platelet thrombus)
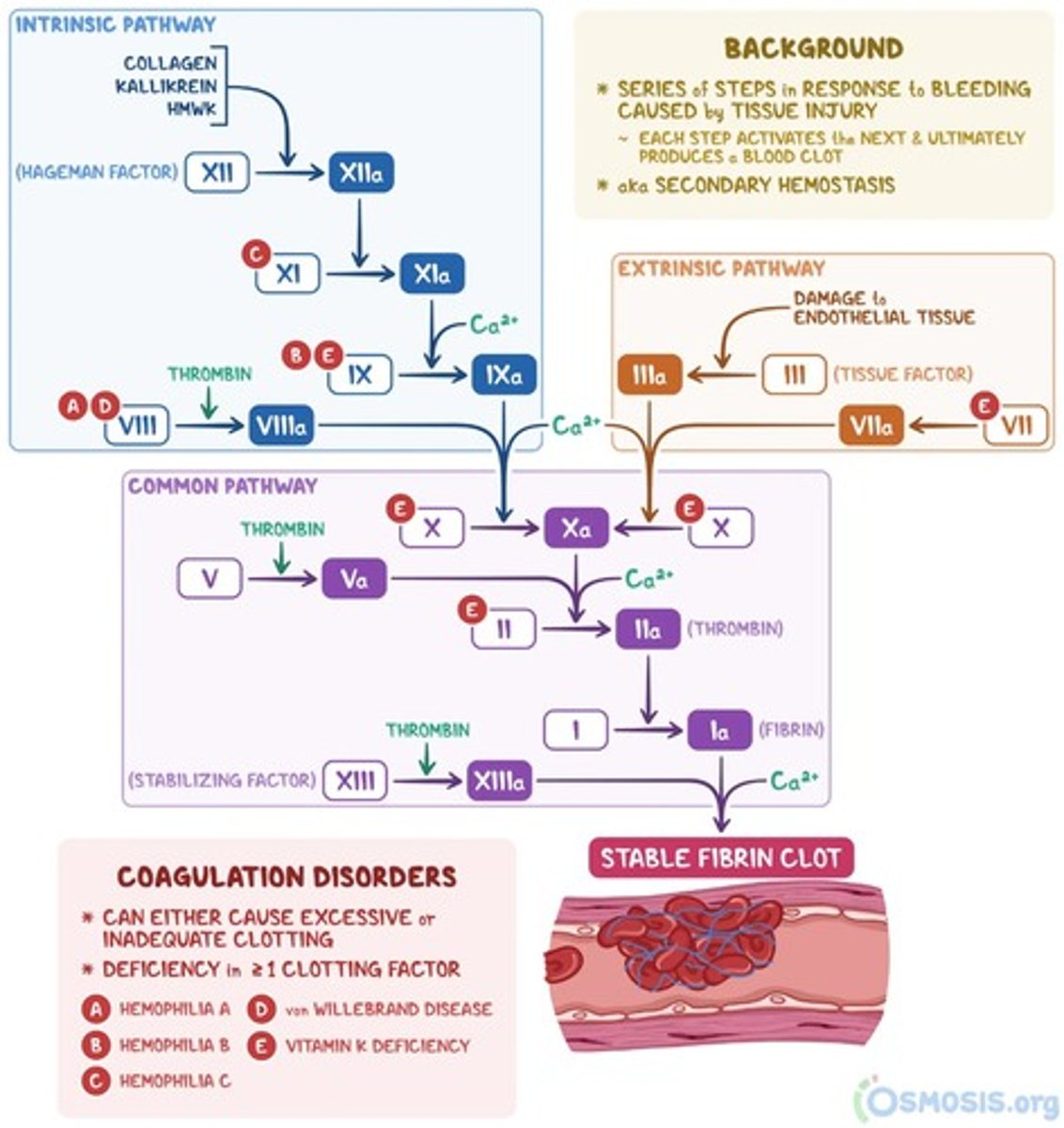
Outline the steps resulting in fibrin clot formation via the intrinsic pathway of coagulation.
1. XII → XIIa: XII (Hageman Factor) is activated via exposure of blood to negatively charged surfaces (e.g., subendothelial collagen)
2. XI → XIa: XIIa activates Factor XI
- Thrombin also plays a part in XI activation
3. IX → IXa: XIa activates Factor IX
4. X → Xa: IXa forms a complex with Factor VIIIa, which activates Factor X
- Phospholipids from aggregated platelets (PL) and Ca2+ are necessary for full activation of X
- Thrombin plays a part in VIII activation
- Xa then enters the common pathway (steps below)
5. II → IIa: Xa (along with FactorVa) convert prothrombin (II) to thrombin (IIa)
- Thrombin plays a part in V activation
6. I → Ia: Thrombin converts soluble fibrinogen (I) to insoluble fibrin (Ia)
- In addition, thrombin amplifies intrinsic coagulation cascade by activating Factors XI, VIII, and V
7. Factor XIIIa cross-links fibrin to form stable fibrin clot
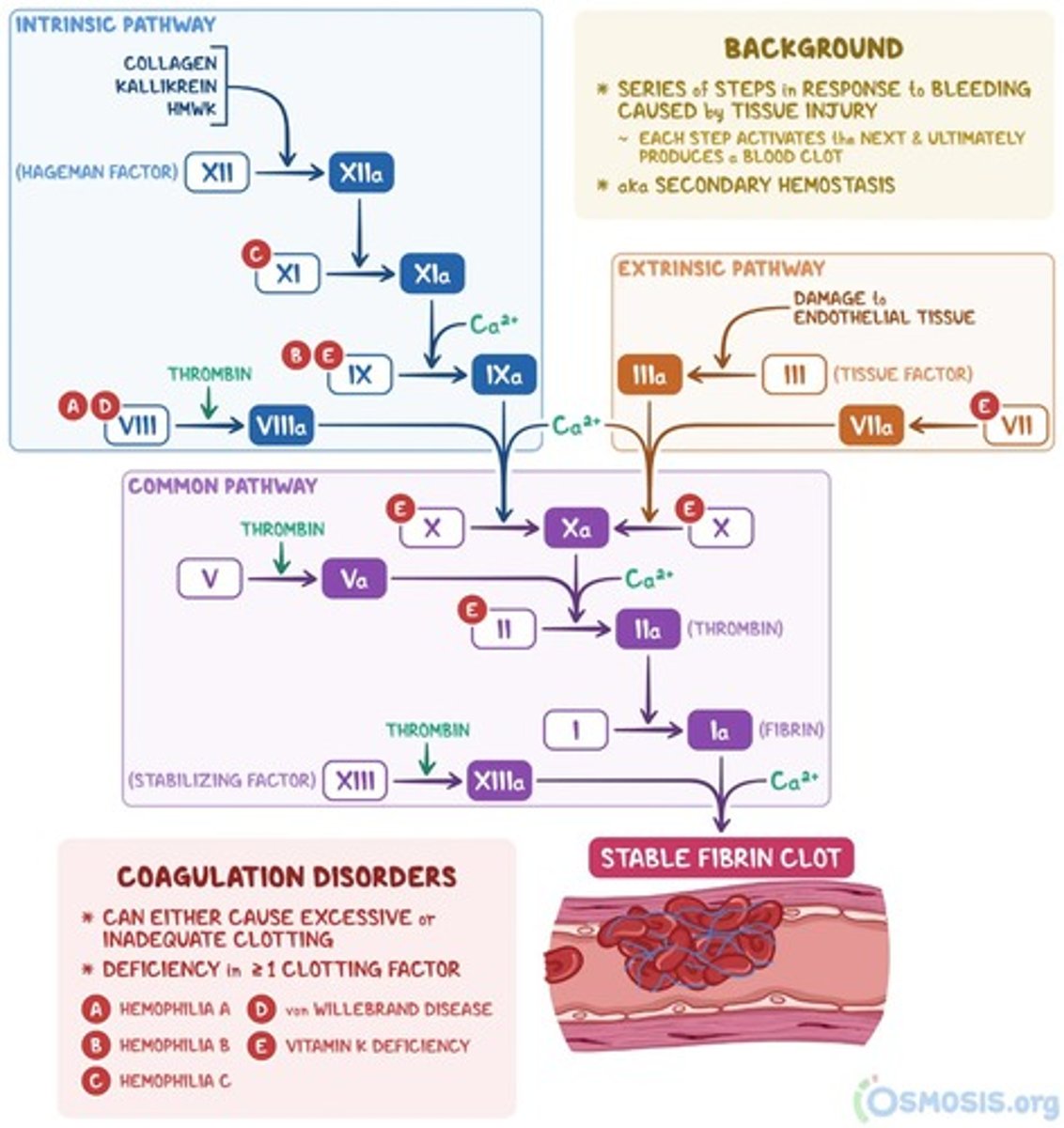
What is tissue factor (TF)?
- Also called thromboplastin
- Expressed on the surface of subendothelial components (e.g., smooth muscle cells, fibroblasts)
- Also found in microparticles released from activated platelets and monocytes
- Initiates extrinsic coagulation pathway by binding to and activating Factor VII
Outline the steps resulting in fibrin clot formation via the extrinsic pathway of coagulation.
1. External trauma causes TF release from damage cells
2. VII → VIIa: TF binds to and activates Factor VII
3. X → Xa: TF-VIIa complex activates Factor X
- TF-VIIa complex also activates IX to IXa in intrinsic pathway
- Xa then enters the common pathway (steps below)
5. II → IIa: Xa (along with FactorVa) convert prothrombin (II) to thrombin (IIa)
6. I → Ia: Thrombin converts soluble fibrinogen (I) to insoluble fibrin (Ia)
7. Factor XIIIa cross-links fibrin to form stable fibrin clot
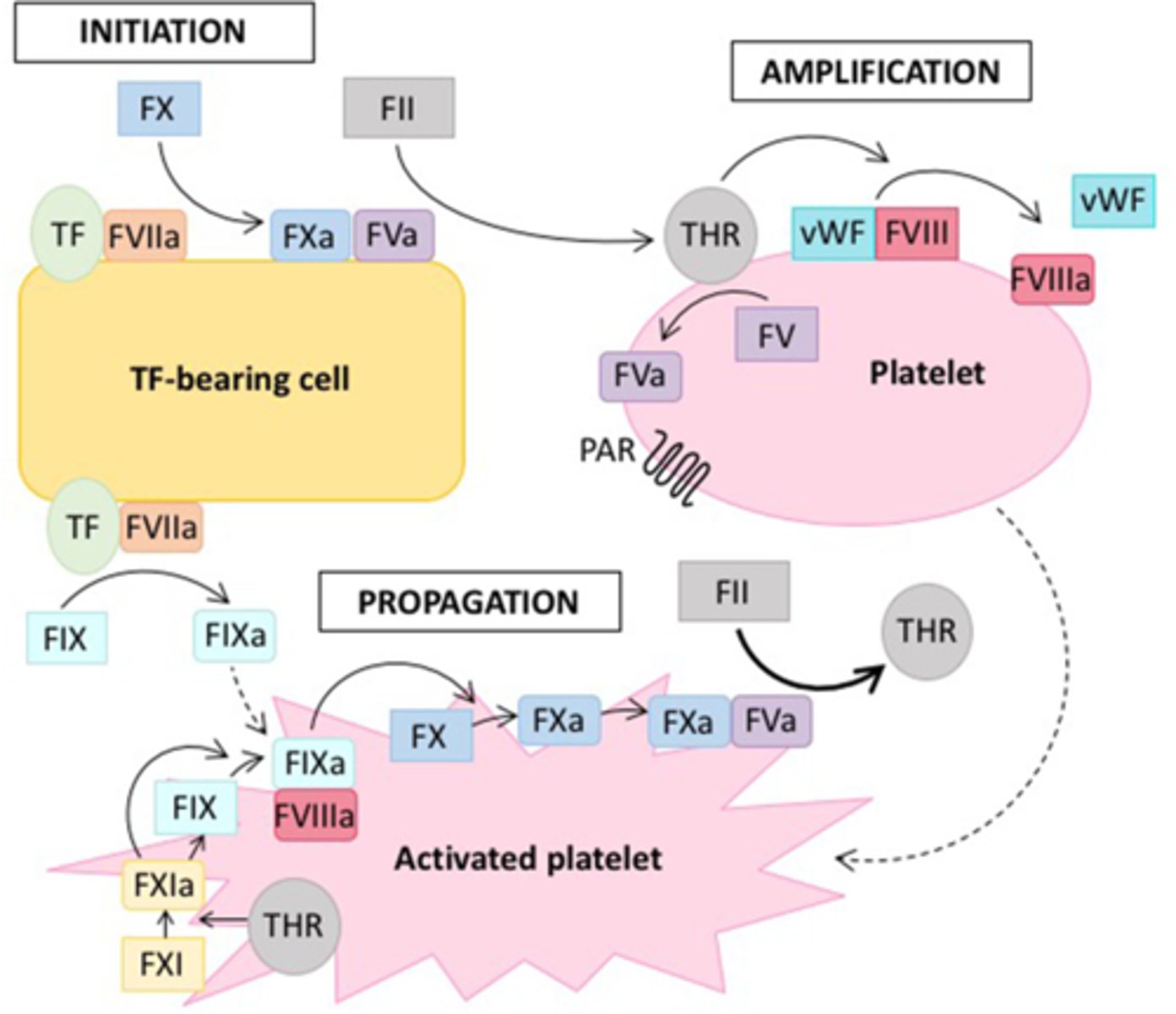
Describe the processes occurring during the initiation phase of the cellular model of coagulation.
Occurs on TF-bearing cells
1. VII → VIIa: TF is released from damaged cells, and binds to/activates Factor VII
2. TF-VIIa (extrinsic tenase) complex activates limited amounts of…
- Factor X (X → Xa): Xa-Va (prothrombinase) complex cleaves prothrombin (II) to generate a small (i.e., picomolar) amount of thrombin (IIa)
- Factor IX (IX → IXa): IXa moves from TF bearing-cells to the surface of activated platelets in the growing platelet thrombus
3. Tissue factor pathway inhibitor (TFPI) rapidly terminates the initiation phase
- Inhibits circulating Xa
- TFPI-Xa complex also inhibits TF-VIIa complex
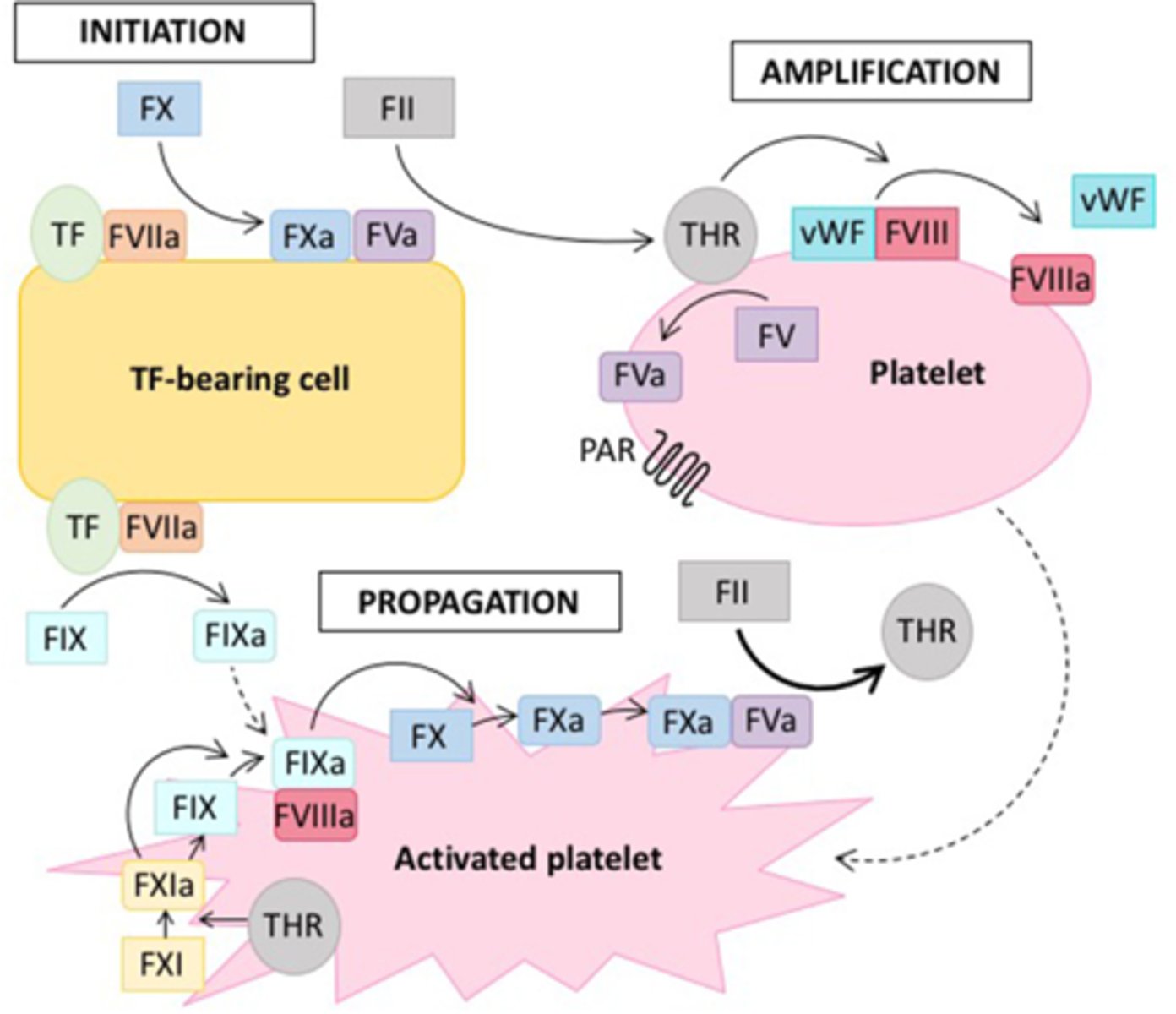
Describe the processes occurring during the amplification phase of the cellular model of coagulation.
Occurs on the surface of platelets
1. Thrombin (IIa) generated from initiation activates…
- Platelets
- Factor V, VIII, and XI
2. IX → IXa: XIa catalyzes the activation of IX, providing supplemental IXa at the platelet surface
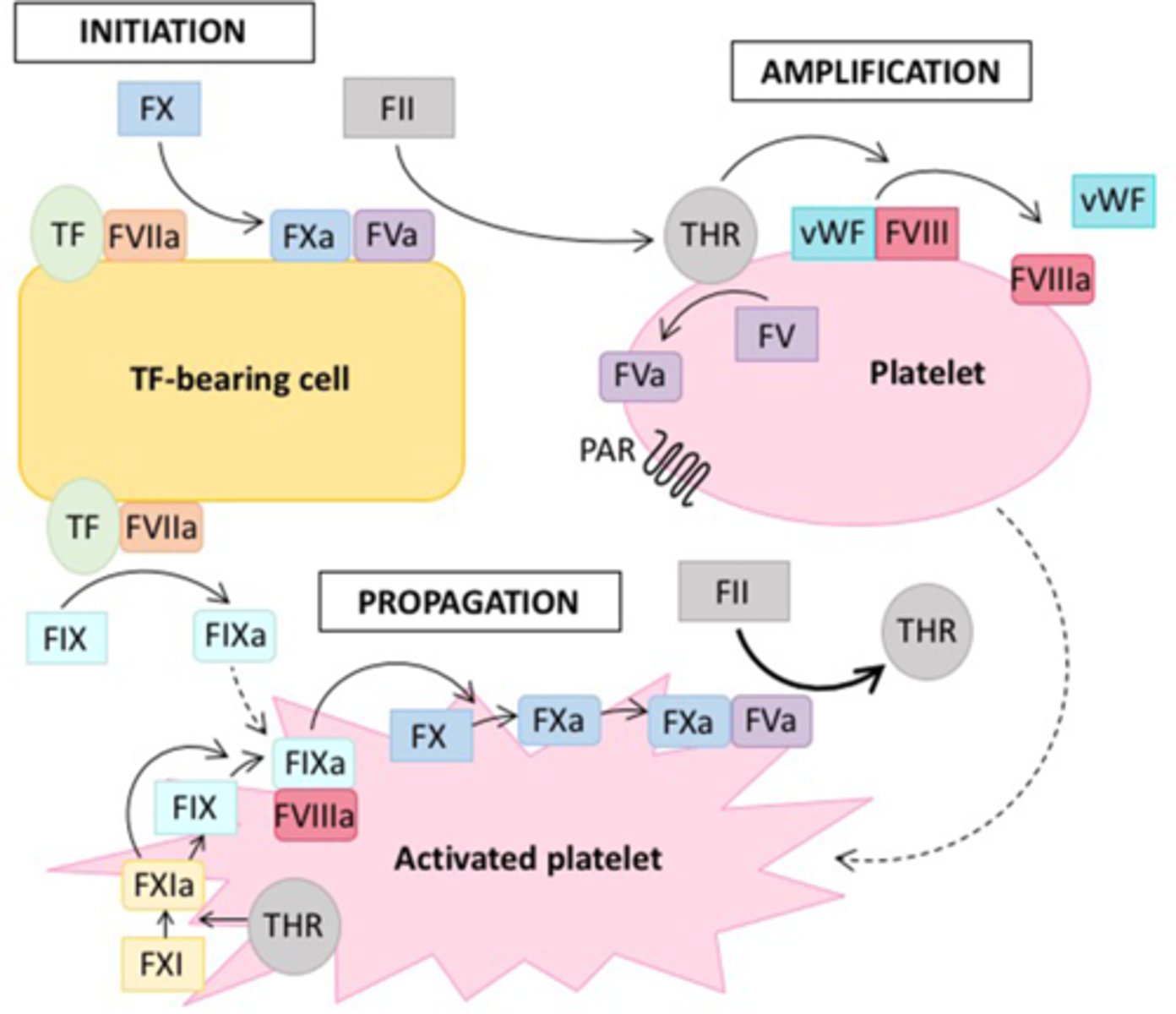
Describe the processes occurring during the propagation phase of the cellular model of coagulation.
Occurs on the surface of activated platelets
1. Activated platelets recruit other circulating platelets to the site of vessel injury via release of…
- Adenosine diphosphate (ADP)
- Serotonin (5-HT)
- Thromboxane A2 (TXA2)
2. X → Xa: Factor VIIIa and IXa (along with Ca2+ and phospholipids) form the intrinsic tenase complex
- VIIIa-IXa-Ca2+-PL complex activates Factor X
3. II → IIa: Xa and Va (along with Ca2+ and phospholipids) form the prothrombinase complex
- Xa-Va-Ca2+-PL complex catalyzes conversion of prothrombin (II) to thrombin (IIa)
- As more platelets are recruited, there is a surge of thrombin production
4. I → Ia: Thrombin converts soluble fibrinogen (I) to insoluble fibrin (Ia), which stabilizes the platelet plug
How is hemostasis controlled by thrombin?
Pro-coagulant activities:
- Activates platelets
- Activates Factors V, VIII, and XI
- Converts fibrinogen to fibrin
Anti-coagulant activities:
- When bound to thrombomodulin, activates Protein C
How is hemostasis controlled by tissue factor pathway inhibitor (TFPI)?
Natural anticoagulant protein
- Binds to and inhibits circulating Xa
- TFPI-Xa complex inhibits TF-VIIa complex
How is hemostasis controlled by antithrombin (AT III)?
Natural anticoagulant protein
- Inhibits Xa and thrombin (IIa)
- Also inhibits XIIa and IXa
How is hemostasis controlled by activated protein C (aPC) and protein S?
Natural anticoagulant proteins
- Inactivate Va and VIIIa (thereby inhibiting the function of prothrombinase and intrinsic tenase complexes)
- Prevent coagulation reactions from spreading to healthy, uninjured vessel walls
How is hemostasis controlled by heparan sulfate?
- Heparin-like compound secreted by endothelial cells
- Exponentially accelerates AT III activity
How is hemostasis controlled by thrombomodulin?
Natural anticoagulant protein
- Binds to thrombin (IIa) to change its function from pro-coagulant to anti-coagulant
- Thrombomodulin-IIa complex activates Protein C (PC → aPC)
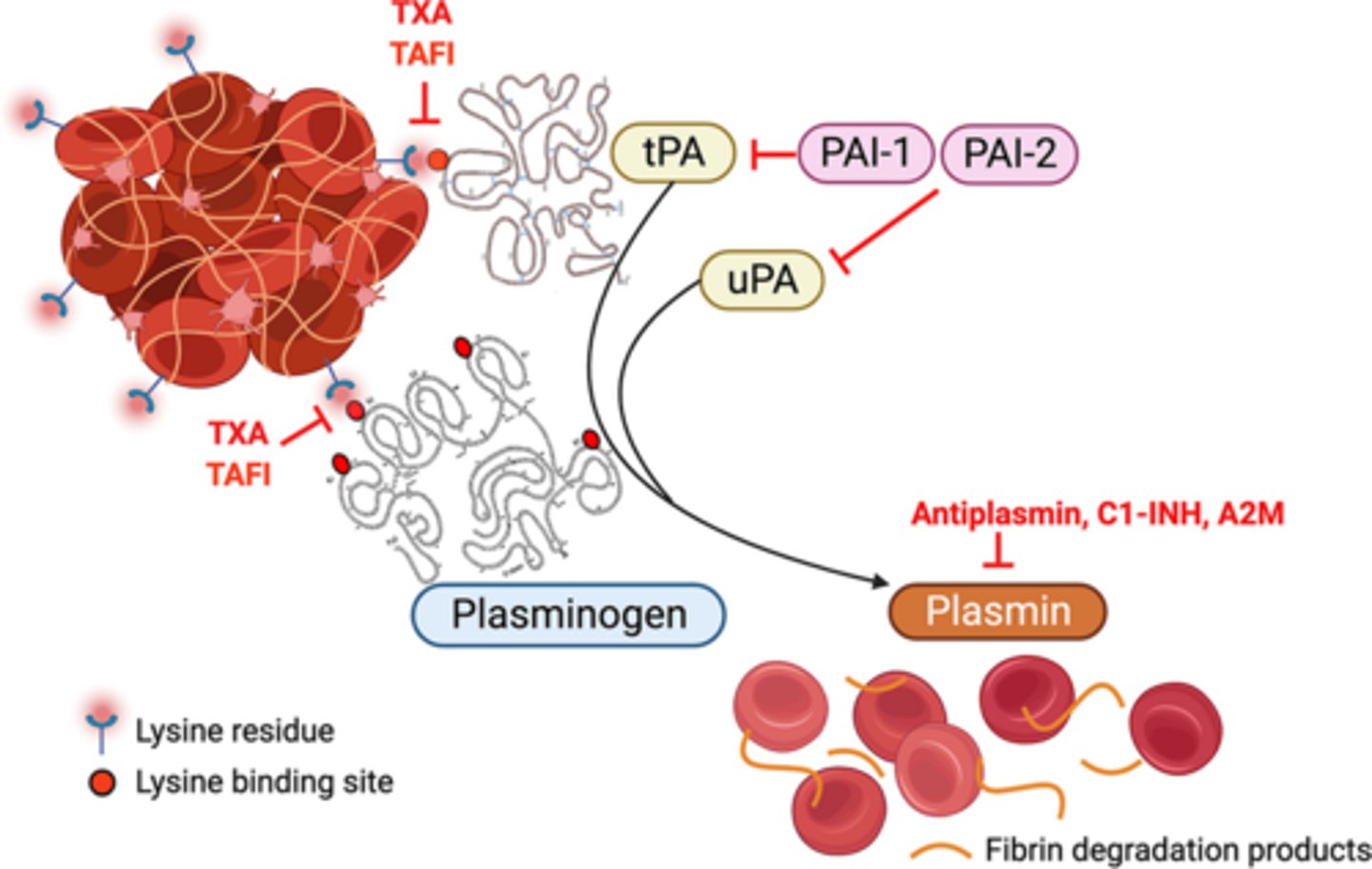
Describe the process of fibrinolysis.
1. Activation of plasminogen to plasmin
- Plasminogen – inactive plasma enzyme in the clot
- Plasmin – enzyme that catalyzes the breakdown of fibrin clots into soluble degradation products
2. Enzymatic degradation of fibrin by plasmin
How is fibrinolysis controlled?
Enhanced by:
- Tissue plasminogen activator (tPA) – enhances conversion of plasminogen to plasmin
- Thrombin (minor effects)
Inhibited by:
- α2-antiplasmin (α2-AP) – inhibits plasmin
- Plasminogen activator inhibitor-1 (PAI-1) – inhibits tPA
What role does normal endothelium play in the control of blood flow and coagulation?
Exhibits anticoagulant/antithrombotic and fibrinolytic properties (i.e., prevents clot formation), promoting production of…
- Prostacyclin (PGI2): Inhibits platelet activation
- Nitric oxide (NO): Inhibits platelet adhesion and aggregation
- Thrombomodulin: Binds thrombin (IIa), resulting in Protein C activation (and thereby inactivation of Va and VIIIa)
- Heparan sulfate proteoglycans: Enhance antithrombin activity (thereby inhibiting thrombin and Factor Xa)
- Plasminogen activators (e.g., tPA, uPA): Activate fibrin breakdown
What role does abnormal endothelium play in the control of blood flow and coagulation?
Exhibits procoagulant/prothrombotic and anti-fibrinolytic properties (i.e., enhances clot formation)
Damage to the endothelium (e.g., mechanical/shear stress, vascular injury, exposure to noxious particles, inflammation), results in…
- Decreased NO/PGI2 and increased vWF (promotes platelet adhesion and aggregation)
- Exposure of TF (triggers extrinsic pathway of coagulation)
- Increased release of fibrinolysis inhibitors (e.g., PAI-1)
Describe the dysregulated coagulation events in diabetes and obesity.
Diabetes and obesity are prothrombotic states
- Associated with hypercoagulability
- Impaired fibrinolysis (via increased PAI-1 levels) reduces breakdown of fibrin clots
How is D-dimer measurement used as a diagnostic method for DVT?
D-dimer is formed as a fibrin degradation product when fibrin is broken down by plasmin
- Sensitive marker of clot formation
- Serum concentrations of D-dimer are significantly elevated in patients with acute thrombosis
Positive D-dimer test is not conclusive evidence of DVT on its own
- May also be elevated due to recent surgery/trauma, pregnancy, advanced age, or cancer
- However, a negative test can be useful to rule out DVT
What are examples of heparin/heparin derivatives?
Unfractionated heparin (UFH)
- Also called high molecular weight heparin (HMWH)
Low molecular weight heparin (LMWH)
- Enoxaparin (Lovenox)
- Dalteparin
Fondaparinux
How do pharmacokinetic properties (e.g., half-life, bioavailability) compare among heparin/its derivatives?
UFH:
- Short half-life (~1-2 hours)
- Lower bioavailability (30%)
LMWH:
- Longer half-life (~4-6 hours)
- Higher bioavailability (90%)
Fondaparinux:
- Longest half-life (~17-21 hours)
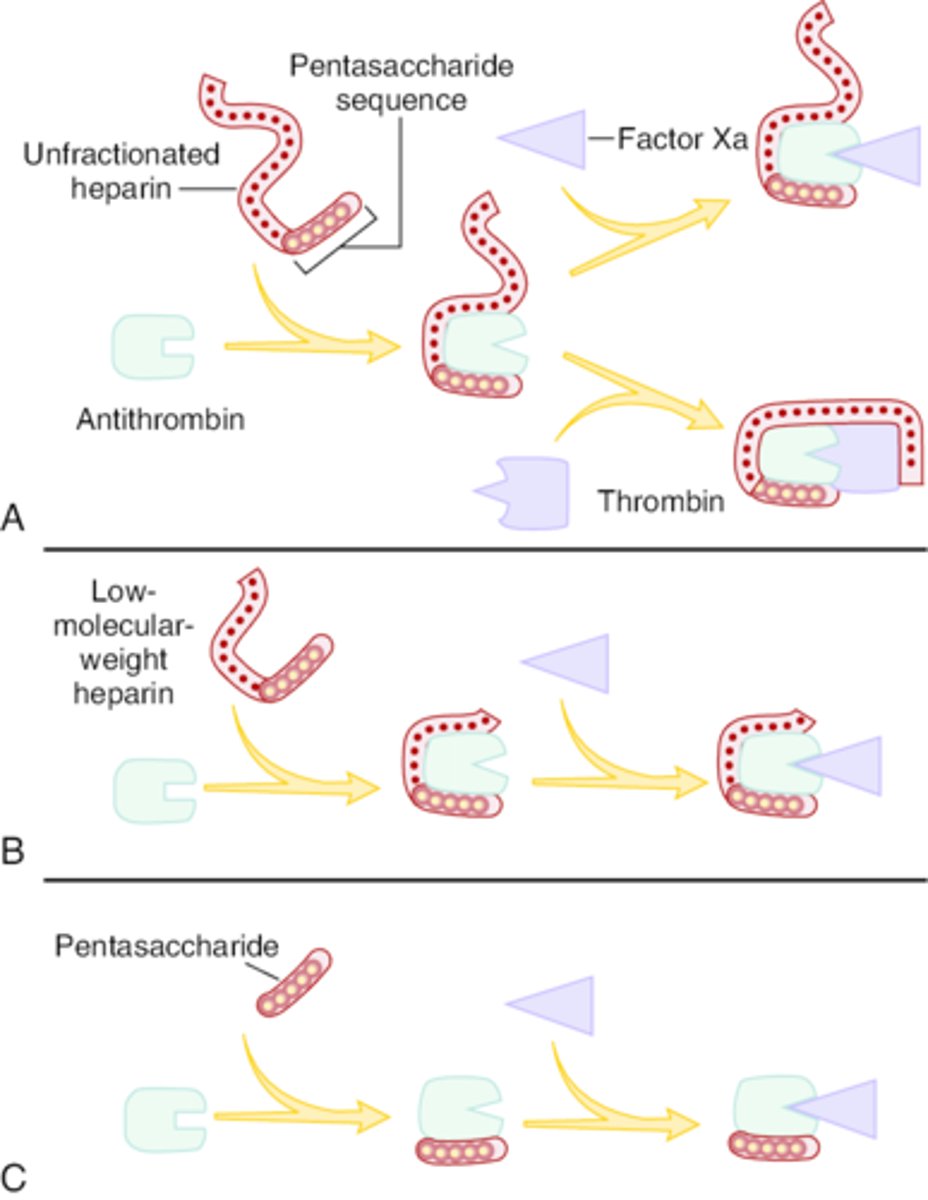
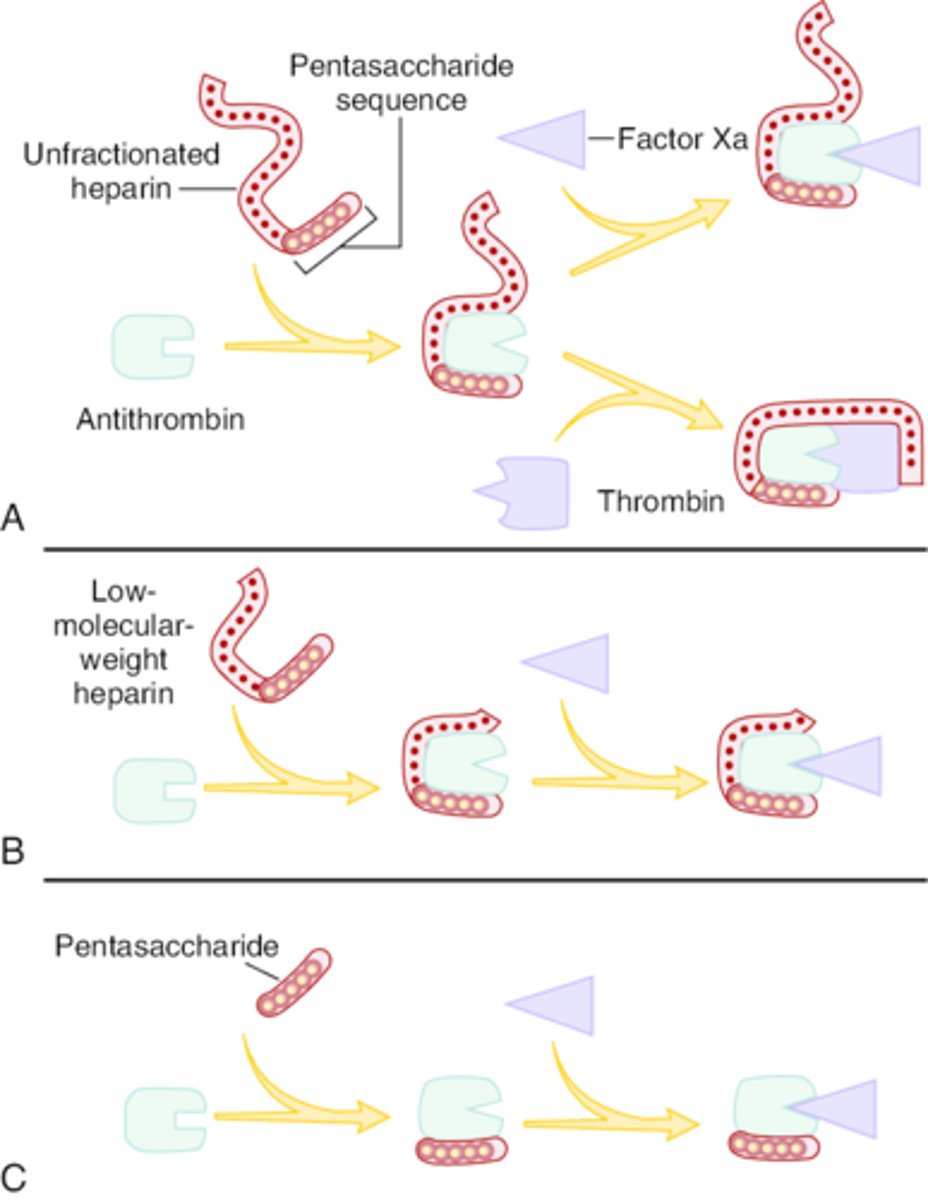
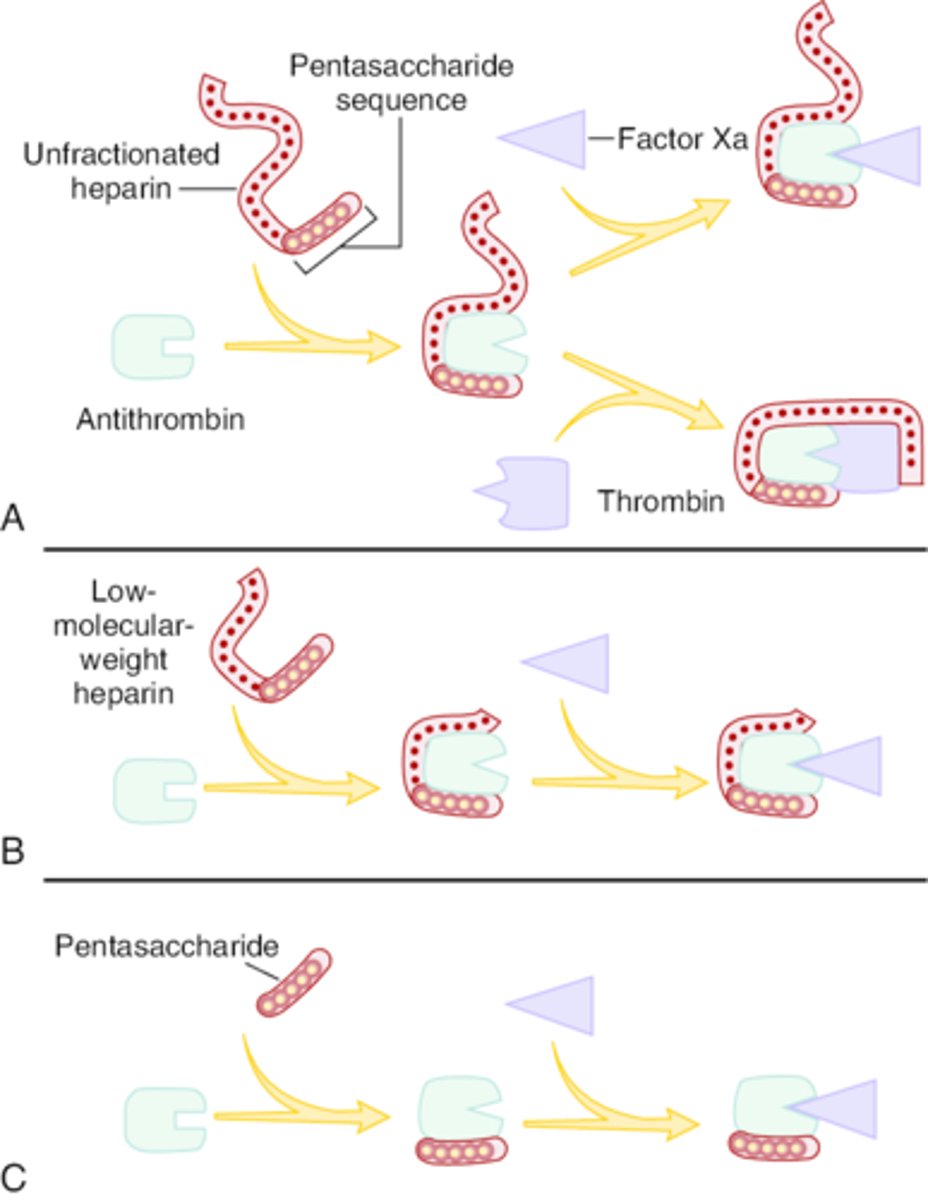
What is the MOA for UFH? Describe its relative effects on Factors Xa and IIa.
Acts on the intrinsic pathway via binding to ATIII
- Binding induces conformational change in ATIII, increasing its affinity for clotting factors
- AT-UFH complex primarily inhibits Xa and thrombin (IIa) at a 1:1 ratio
- Also inhibits XIIa and IXa (less sensitive)
What adverse effects may occur with UFH? Describe mitigation strategies.
Bleeding (very common)
- May need to administer reversal agent if severe
Osteoporosis
- Replace UFH with LMWH
HIT
- Replace UFH with DIT
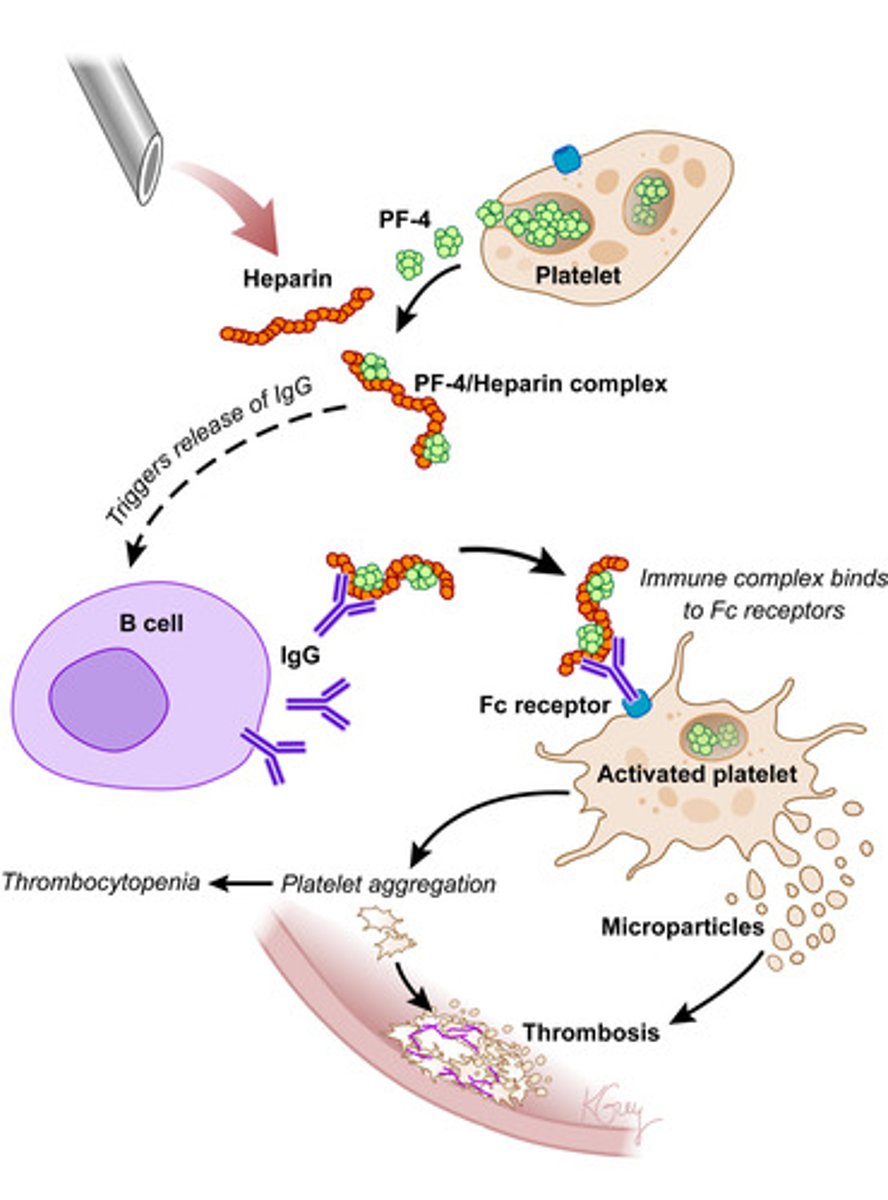
What is heparin-induced thrombocytopenia (HIT)?
Immune-mediated reaction to heparin
- Antibodies form against heparin-platelet factor 4 (PF4) complex
- Antibodies bind to Fc receptor on platelet, inducing activation and aggregation
- Results in decreased platelet count (i.e., thrombocytopenia) and hypercoagulable state (i.e., thrombosis)
How does the incidence of HIT compare among heparin/its derivatives?
UFH: Highest incidence
- If HIT occurs, replace UFH with DIT
LMWH: Lower incidence than UFH
- Less interaction with PF4
- Fewer heparin-dependent IgG antibodies
Fondaparinux: No risk (does not cross react with HIT antibodies)
What reversal agent is used for UFH?
Protamine sulfate
- Greater affinity for heparin than heparin’s affinity for ATIII
- Rapid onset (neutralizes effects of heparin in 5 minutes)
- Should only be used for a prolonged aPTT due to risk of ADRs
- Excess protamine should be avoided (functions as an anticoagulant at high concentrations)
What is the MOA for LMWH? Describe its relative effects on Factors Xa and IIa.
Acts on the intrinsic pathway via binding to ATIII
- Binding induces conformational change, increasing its affinity for clotting factors
- AT-LMWH complex inhibits Xa and thrombin (IIa) at a 3:1 ratio
What reversal agent is used for LMWH?
Protamine (only achieves partial reversal)
What is the MOA for fondaparinux? Describe its relative effects on Factors Xa and IIa.
- Synthetic pentasaccharide that binds to AT
- Selectively inhibits Xa only
What reversal agent is used for fondaparinux?
N/A (no reversal agent available)
How does monitoring compare among heparin/its derivatives?
UFH:
- Activated partial thromboplastin time (aPTT), OR
- AntiXa assay
LMWH:
- Unnecessary in majority of patients (may be needed in patients with renal insufficiency, obesity, or bleeding risk)
- If needed: AntiXa assay used (LMWH have minimal effect on aPTT)
Fondaparinux:
- Rarely required
- If needed: AntiXa assay used
What are examples of parenteral direct thrombin inhibitors (DTIs)? Describe their MOA and indication.
Agents:
- Argatroban
- Bivaluridin
MOA: Reversibly bind to and inhibit thrombin
- Blocks conversion of fibrinogen to fibrin
Indication: Prevention and treatment of HIT
What is an example (brand and generic) of an oral direct thrombin inhibitor (DTI)? Describe its MOA and pharmacokinetic properties.
Dabigatran (Pradaxa)
MOA: Orally active DTI
- Selectively and reversibly inhibits free and clot-bound thrombin (IIa)
- Prevents conversion of fibrinogen to fibrin
Pharmacokinetics:
- Formulated as a prodrug (dabigatran etexilate mesylate) to overcome poor bioavailability
- Renally eliminated (caution in patients with renal dysfunction)
What adverse effects may occur with dabigatran?
- Bleeding
- Dyspepsia
What drug interactions occur with dabigatran?
P-gp inhibitors: Decrease DOAC metabolism
- Increased bleeding risk
- Examples: Ketoconazole, amiodarone, verapamil
P-gp inducers: Increase DOAC metabolism
- Decreased efficacy, and therefore increased VTE risk
- Examples: Rifampin, phenytoin
Antiplatelet/NSAIDs
- Increased bleeding risk
What reversal agent is used for dabigatran?
Idarucizumab (Praxbind)
- Monoclonal antibody fragment
- Indicated for emergency surgery/urgent procedures or in life-threatening/uncontrolled bleeding
- Common side effect = headache
What are examples (brand and generic) of direct factor Xa inhibitors? Describe their MOA and pharmacokinetic properties.
Agents:
- Rivaroxaban (Xarelto)
- Apixaban (Eliquis)
- Edoxaban (Savaysa)
MOA: Selectively and directly inhibit free and clot-bound Factor Xa
- Blocks conversion of prothrombin (II) to thrombin (IIa)
- Do not require AT (like heparin) to exert effect
Pharmacokinetics:
- Good oral bioavailability (> 60%)
- Renally eliminated (caution in patients with renal dysfunction)
What adverse effects may occur with direct factor Xa inhibitors?
- Typically well tolerated
- Bleeding (most common)
What drug interactions occur with direct factor Xa inhibitors?
CYP3A4/P-gp inhibitors: Decrease DOAC metabolism
- Increased bleeding risk
- Examples: Ketoconazole, amiodarone, verapamil
- Note: Only rivaroxaban/apixaban metabolized by CYP
CYP3A4/P-gp inducers: Increase DOAC metabolism
- Decreased efficacy, and therefore increased VTE risk
- Examples: Rifampin, phenytoin, carbamazepine, St. John’s Wort
- Note: Only rivaroxaban/apixaban metabolized by CYP
Antiplatelets/NSAIDs
- Increased bleeding risk
What reversal agent is used for direct factor Xa inhibitors?
Andexanet alfa
- Modified recombinant Factor Xa (i.e., “decoy” Xa)
- For rivaroxaban and apixaban only
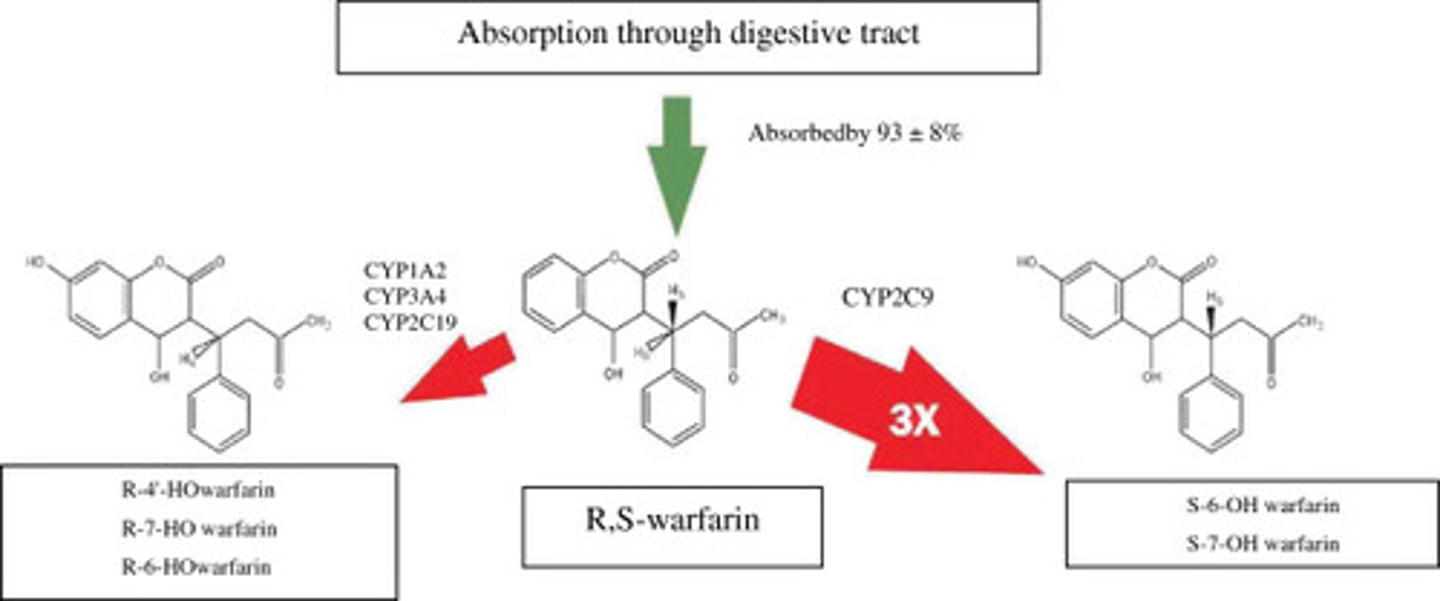
Describe the metabolism of warfarin.
Racemic mixture of two enantiomers, both of which undergo metabolism in the liver
S-warfarin (3-5x more pharmacologically active)
- Metabolized primarily by CYP2C9
- Produces inactive hydroxylated metabolites
R- warfarin
- Metabolized mainly by CYP1A2, CYP3A4, and CYP2C19
- Produces inactive hydroxylated metabolites
What is the MOA for warfarin?
Inhibits vitamin K epoxide reductase complex 1 (VKORC1) in the liver
Blocks conversion of oxidized vitamin K to reduced vitamin K
- VKORC1 recycles vitamin K epoxide back to its reduced, active form
- Reduced vitamin K is required as a cofactor for the enzyme γ-glutamyl carboxylase (GGCX)
- GGCX catalyzes the carboxylation (and activation) of certain clotting factors
Prevents carboxylation (i.e., activation) of vitamin K-dependent coagulation factors:
- Procoagulant Factors II, VII, IX, and X
- Anticoagulant proteins C and S
Results in decreased formation of active clotting factors
- No direct effect on already circulating clotting factors or previously formed thrombus
What pharmacokinetic considerations should be made with warfarin?
Onset of action: 24-72 hours
- Delayed anticoagulant effect due to long half-lives of pro-coagulant factors (especially II and X)
- As a result, warfarin requires bridging with another anticoagulant (e.g., heparin) upon initiation
Duration of action: 2-5 days
- Important consideration when scheduling surgery
Describe how the pharmacogenetics of CYP2C9 influences warfarin dosing.
CYP2C9 metabolizes the more potent S-warfarin enantiomer
Variations in CYP2C9 gene can reduce enzyme activity
- Decreases metabolism of warfarin
- Bleeding risk increased at standard doses
Common polymorphisms:
- *1 (wild-type): Rapid metabolism (standard dose)
- *2: Reduced metabolism (lower dose required)
- *3: Severely reduced metabolism (much lower dose required)
Describe how the pharmacogenetics of VKORC1 influences warfarin dosing.
VKORC1 is the target enzyme of warfarin
Variations in VKOR can affect enzyme expression
- Amount of VKOR alters how much reduced vitamin K is regenerated
- G allele = higher enzyme expression (decreased sensitivity to warfarin/requires higher dose)
- A allele = lower enzyme expression (increased sensitivity to warfarin/requires lower dose)
Common polymorphisms:
- GG: More protein (higher dose required)
- AA: Less protein (lower dose required)
Why must warfarin be monitored closely?
Warfarin has a narrow therapeutic window
- Dose adjustments are based on INR results
- INR that is too high = increased bleeding risk
- INR that is too low = increased clotting risk
Discuss different drug-drug interactions and how they influence the activity of warfarin.
CYP2C9 inhibitors: Decrease warfarin metabolism
- Increased bleeding risk
- Examples: Amiodarone, fluconazole
CYP2C9 inducers: Increase warfarin metabolism
- Decreased efficacy, and therefore increased clotting risk
- Examples: Rifampin, carbamazepine, phenobarbital, phenytoin
NSAIDs/antiplatelets/DTIs/APAP
- Increased bleeding risk
Drugs that decrease warfarin absorption
- Increased clotting risk
- Examples: Sucralfate, bile acid sequestrants (e.g., cholestyramine)
Drugs that decrease vitamin K production/absorption
- Increased bleeding risk
- Example: Broad-spectrum antibiotics
Discuss drug-food interactions that influence the activity of warfarin.
Vitamin K-rich foods: Reverse pharmacological activity of warfarin
- Examples: Leafy green vegetables (e.g., spinach, kale, Swiss chard), broccoli, black/green tea, chickpeas
- Patients should maintain consistent vitamin K intake
What adverse effects may occur with warfarin?
Bleeding (most commonly in GI tract)
Anemia
Teratogenic effects (contraindicated in pregnancy)
Warfarin-induced skin necrosis
- Caused by transient hypercoagulable state due to rapid drop in PC
- More common in patients with protein C or S deficiency
Purple toe syndrome
- Reversible, painful, blue-tinged discoloration of toes
- Appears 3-8 weeks after initiation of therapy
- Caused by cholesterol microembolization
Hepatic dysfunction
Alopecia
Hypersensitivity reactions (e.g., dermatitis)
What reversal agents are used for warfarin?
Phytonadione (vitamin K1)
- MOA: Replenishes vitamin K stores, increasing hepatic synthesis of Factors II/VII/IX/X and proteins C and S
- Administered PO or IV
- Boxed warning: Hypersensitivity reaction with IV/IM administration
Kcentra (four-factor prothrombin complex concentrate, 4F-PCC)
- MOA: Blood product that contains Factors II/VII/IX/X and proteins C and S
- Boxed warning: Arterial and venous thromboembolic complications
Fresh frozen plasma (FFP)
What is the MOA for fibrinolytic agents?
Function as plasminogen activators
- Converting plasminogen to plasmin
- Plasmin then degrades fibrin and breaks up thrombi
What are examples of fibrinolytic agents? Discuss their pharmacology.
First-generation agents (no longer available in the U.S.):
- Streptokinase
- Urokinase
Alteplase
- Recombinant tPA
- Short half-life (~5 minutes)
- High affinity for fibrin
- IV infusion dosing (requires pump)
Tenecteplase
- Modified tPA)
- Longer half-life than alteplase (~20-30 minutes)
- More fibrin-specific than alteplase
- Single bolus dosing (easier to administer)