Organic Chemistry chp. 14 - Aromatic Compounds
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Benzene nomenclature
All are attached to benzene
“Halogen” benzene - halogen
phenol - alcohol
Toluene - CH3
Aniline - NH2
Benzenesulfonic acid - SO3H
Benzoic acid - carboxylic acid
Acetophenone - ketone
Anisole - ether
Disubstituted benzene names
Ortho - 1,2
Meta - 1,3
para - 1,4
Xylene
disubstituted benzene with two methyls
phenyl group naming trend
If the attached chain is straight than use “prefix” benzene
If attached chain is branched use phenyl + chain name
Benzyl vs phenyl reactivity
Benzyl groups are much more reactive and susceptible to substitution rxns
Phenyls are more inert
Non reactive benzene rxns
Br2 - no halogenation
KMnO4 - no oxidation
H3O+ - no hydration
H2/Ni - slow addition at high temp
Halogenation of benzene outcome
Leads to a substitution rather than an addition.
1 Br is added not 2
Best way to depict a benzene ring
circle in the center indicating delocalization of electrons around entire ring
Kekule Benzene depiction
cyclohexane with three double bonds. This depiction has problems such as the fact that isomers cannot be created and it would have a higher heat of hydrogenation compared to the actual benzene structure.
Stability of benzene explained
Benzene has a much lower heat of hydrogenation than the Kekule depiction due to resonance stability that gives each DB less heat of hydrogenation compared to DBs in cyclohexene
Why is benzene an example of perfection in nature
All bonds lengths and angles are equal
Bond lengths: 1.09 b/w C+H 1.39 b/w C+C
Angle: 120
Frost circle rules
cyclic structure is surrounded by circle
Must be a C touching the bottom
A line must be drawn across the middle
Bonding pie orbitals are below the line and antibonding pie orbitals are on the top
Amount of Bonding pie orbitals is determined by number of C below or above center line
if center line crosses C’s than nonbonding orbitals are available
Orbital type ranked by strength
Antibonding pie: Least stable
Nonbonding pie: Middle stability
Bonding pie: Most stable
Annulene nomenclature
Cyclic structures with CnHn formula (ex. benzene C6H6)
[#C]annulene (ex. Benzene - [6]annulene)
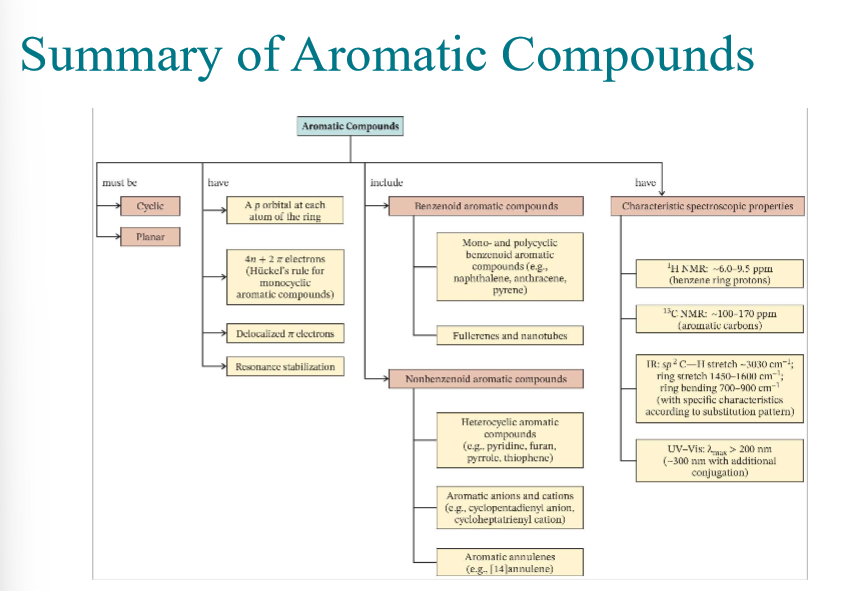
Huckels rule for aromaticity
Aromatic:
Cyclic
Planar
Fully conjugated
4n+2 resonating electrons
Anti aromatic:
cyclic
planar
Fully conjugated
4n resonating electrons
Non aromatic:
exception to rules above
Exception of [10]annulene and [8]annulene
nonaromatic because they are not planar due to steric hinderance or angle strain
Trick for antiaromatic annulenes
The ones with nonbonding pie orbitals will be antiaromatic
Numbering polyaromatic compounds
Carbons connecting rings get #a
ex. Naphthalene has 4a and 8a carbons
Can anions and cations be aromatic
yes
ex. cyclopentadiene (-) and cycloheptatriene (+)
Common Aromatic heterocycles
pyridine (6 w/ N)
pyrrole (5 w/ N)
Furan (5 w/ O)
Thiophene (5 w/ S)
two types of electrons in conjugated pie systems
coplanar
ortaganol
Are lone pairs included in aromaticity
Depends
If it is aromatic w/o than no
If it is aromatic w/ than yes
Imidazole Basicity
Accepts hydrogen at dehydrated nitrogen in order to maintain aromaticity
Furan and Thiophene lone pair electrons
One pair is coplanar and used in pie orbital to be delocalized around ring
One pair is ortaganol can be used as electron donor (Base)
Two benzene based amino acids
Phenylalanine
Tyrosine
where heterocycles are seen in bio
Tryptophan (indole)
Purines and Pyridines
NAD+
NADH and NAD+ used in bio
NAD+ is aromatic but gets hydrogenated to become NADH which not aromatic then NADH gets oxidized in the ETC and becomes NAD+ restoring aromaticity
Polycyclic benzenoid aromatic compounds to remember
Naphthalene
anthracene
pyrene
Know Summary