amino acid metabolism
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
amino acid catabolism accounts for
10-15% of human energy production
nitrogen cannot be stored in a usable form because
NH4+ is toxic
nitrogen that is lost as a result of protein and nucleic acid degradation must be
replenished through diet
three reasons why proteins are constantly degraded
to store nutrients in the form of proteins and break them down in times of metabolic need (significant in muscles)
to eliminate the accumulation of abnormal proteins
short half-lives of all enzymes (seconds, to days, to weeks, to months)
nitrogen balance
the daily intake of nitrogen (from proteins) equals the amount of nitrogen excreted (in waste)
ATP-independent process of amino acid degradation
degrades proteins in lysosomes
lysosomes contain acidic proteases that non-selectively digest protein particles
ATP-dependent process of amino acid degradation
ubiquitin-proteasome pathway
degrades proteins containing ubiquitin polymers in the proteasome
uses three different protease activities located in the central core of proteasome
ubiquitin
regulatory protein with seven lysine residues which serve as linking sites between ubiquitin monomers
the C-terminal glycine of ubiquitin serves as the attachment site to targeted proteins or to other ubiquitin monomers
at least four ubiquitin subunits linked between glycine and lysine tags a protein for proteasomal degradation
enzymes involved in ubiquitination
E1- attach ubiquitin to E2 enzymes
E2- attach ubiquitin to target proteins
E3- facilitate ubiquitination of target proteins by forming a complex with E2 enzymes and target proteins (ubiquitin ligases)
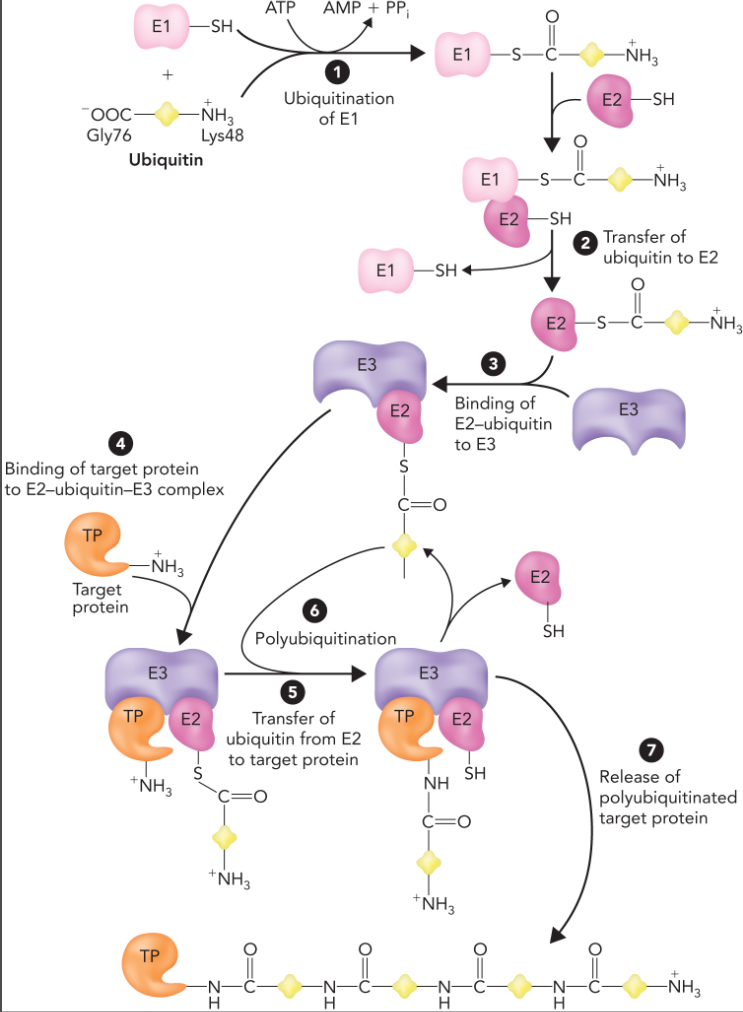
attachment of ubiquitin to a target protein
ubiquitin is activated through an ATP dependent reaction which links ubiquitin to E1
ubiquitin is then transferred to E2 which releases E1 and leads to the formation of an E2-E3 complex
ubiquitination of target protein initiates the polyubiquitination process which links at least four ubiquitin subunits together through a series of gly76-lys48 linkages
gastrin
a small peptide hormone that gets released when food enters the stomach; it triggers the release of gastric juices containing HCl and the secretion of pepsinogen (zymogen of pepsin)
the increasing acidity in the stomach
denatures dietary proteins, allowing for greater chances of peptide bond hydrolysis, and activates pepsin by autocatalytic cleavage of pepsinogen (maximally active at a pH value of around 2)
proteases
pepsin— cleaves long polypeptide chains into a mixture of smaller peptides in the stomach (low pH)
enteropeptidase— a protease that specifically activates several proteolytic zymogens released from the pancreas
trypsin and chymotrypsin— cut proteins and larger peptides into smaller peptides in the small intestine
aminopeptidase and carboxypeptidases A and B— degrade peptides into amino acids in the small intestine
secretin
a hormone secreted into the blood in response to low pH in the small intestine; stimulates the pancreas to secrete bicarb into the small intestine to bring pH back to neutral conditions (which allows for enteropeptidase activity)
cholecystokinin
a hormone secreted into the blood in response to the arrival of peptides in the duodenum; stimulates the secretion of pancreatic proteases trypsinogen, chymotrypsinogen, and procarboxypeptidases A and B
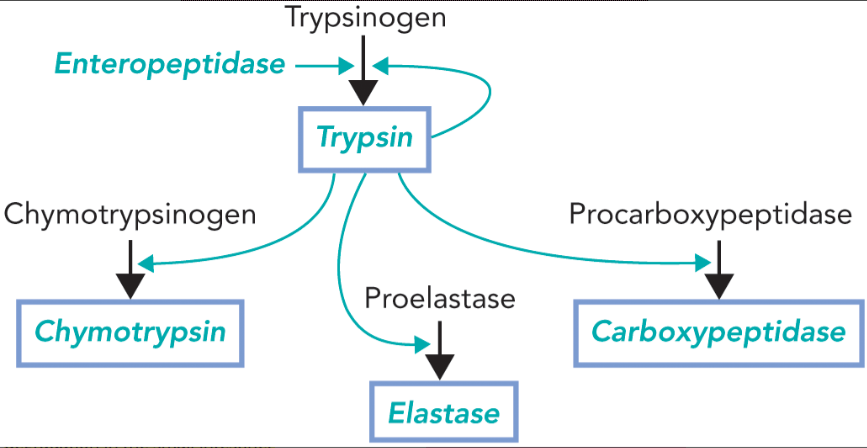
proteolytic cascade
enteropeptidase cleaves trypsinogen resulting in the active trypsin
trypsin cleaves and activates more trypsinogen as well as other zymogens
pancreatic trypsin inhibitor protects the pancreas against
self digestion (pancreatitis)
free amino acids are transported into
epithelial cells lining the small intestine, get exported to the blood, and travel to the liver
amino acids which play a key role in the transport and distribution of other amino acids
alanine, glutamate, glutamine, and aspartate
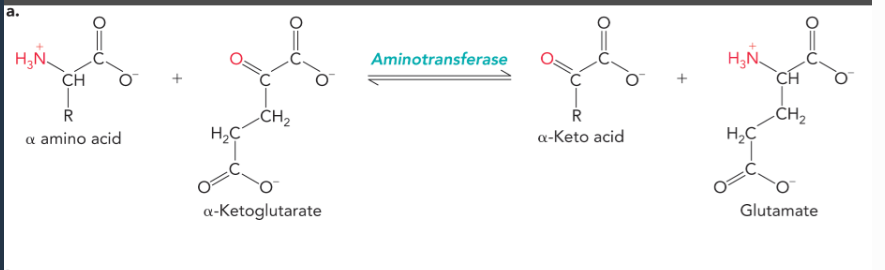
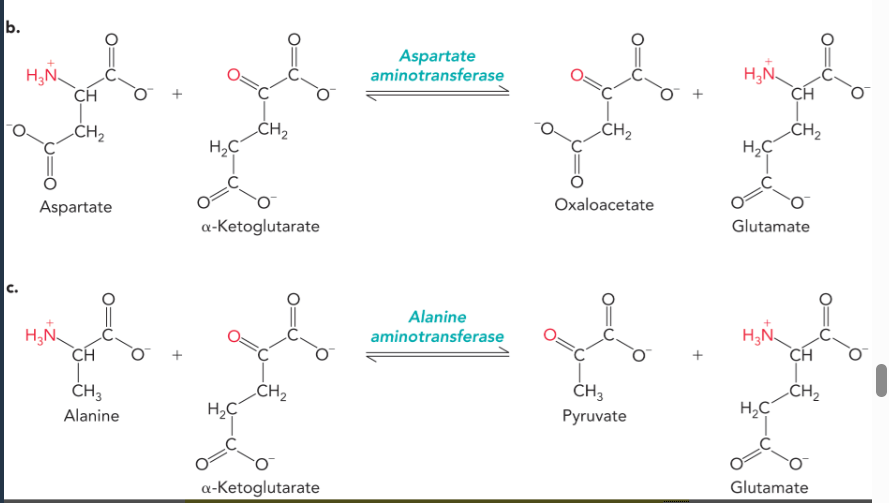
glutamate provides nitrogen for amino acid biosynthesis through the action of
aminotransferase enzymes, which transfer the alpha amino group from an amino acid to alpha-ketoglutarate which yields glutamate and an alpha-keto acid analog of the amino acid (reversible reactions)
pyridoxal phosphate (PLP)
a coenzyme used as a prosthetic group by all aminotransferases; first accepts amino group of amino acid and releases corresponding alpha-keto acid, then transfers amino group to alpha-ketoglutarate or oxaloacetate
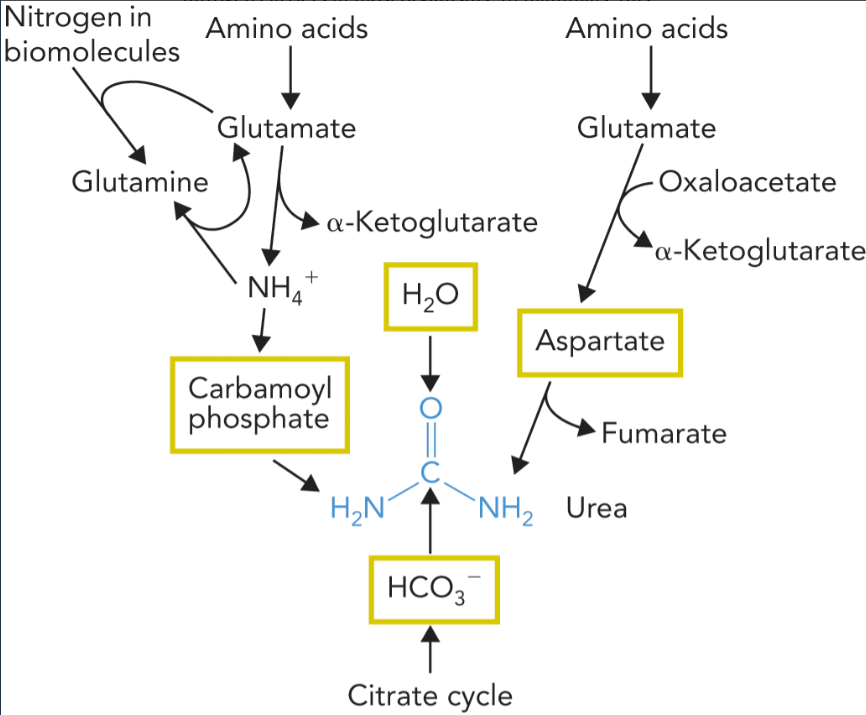
urea is derived from
nitrogen
the ammonium released when glutamate or glutamine is deaminated
aspartate which is formed when oxaloacetate is transaminated by aspartate aminotransferase
carbon
bicarb from citrate cycle
oxygen
from water produced by citrate cycle
why are certain aminotransferases used as an indicator of liver health
buildup of aspartate aminotransferases and alanine aminotransferases mean that the liver is not functioning properly
nitrogen assimilation
the processes used by plants and prokaryotes to incorporate nitrogen (usually as ammonium) into organic compounds
the primary nitrogen carriers in the cell are the amino acids
glutamate and glutamine
when animals eat plants, the glutamate and glutamine they ingest provide
the nitrogen needed to synthesize a variety of biomolecules
glutamine is the primary source of amino groups for the biosynthesis of
nucleotide bases, carbamoyl phosphate, and the side chains of tryptophan and histidine
ammonia assimilation
the incorporation of ammonium into glutamate and glutamine
glutamine synthetase (found in all organisms)
glutamate synthase (found in plants, bacteria, and some insects)
glutamate dehydrogenase (found in all organisms)
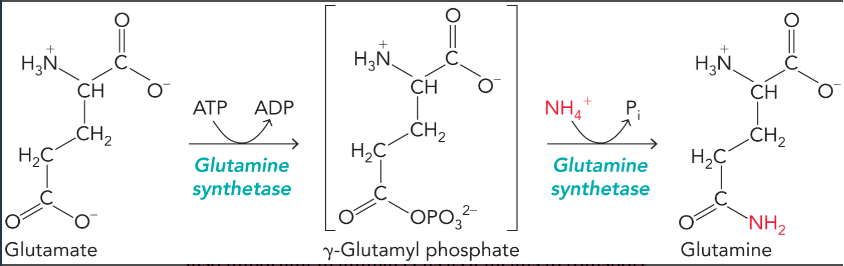
glutamine synthetase
converts glutamate to glutamine using ammonium (requires ATP)
primary entry point for ammonium into biomolecules
allows ammonium transport from peripheral tissues to liver to be excreted as urea
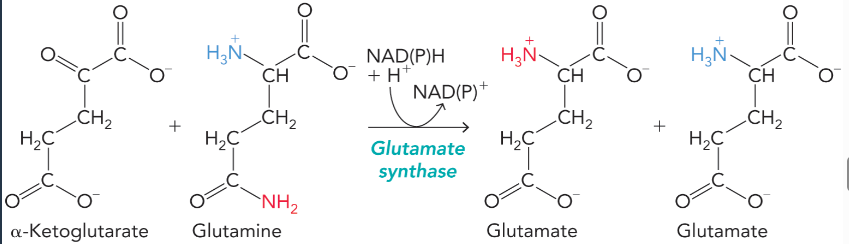
glutamate synthase
transfers the amide nitrogen from glutamine to alpha-ketoglutarate to form two molecules of glutamate
NAD(P)H is oxidized
animals depend on plants for this
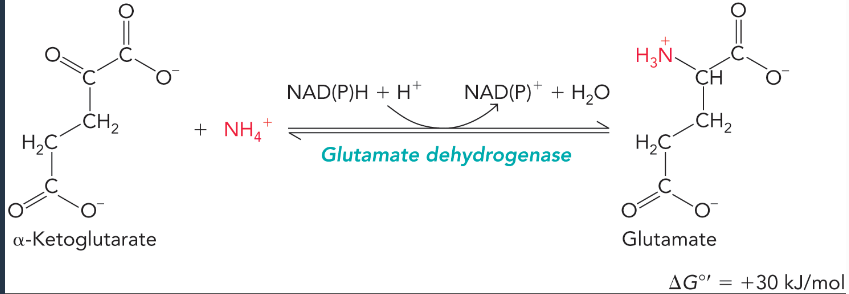
glutamate dehydrogenase
interconverts alpha-ketoglutarate and glutamate in the presence of high ammonium
most often generates ammonium for carbamoyl phosphate synthesis by doing the more favorable reverse reaction
cells cannot store
amino acids that accumulate as a result of protein degradation
must be recycled for protein synthesis or deaminated in order to reuse their carbon skeleton for other metabolic pathways (glycolysis, the citrate cycle, gluconeogenesis, fatty acid synthesis, ketogenesis)
deamination generates ammonium which is used to synthesize other nitrogen containing biomolecules or excreted as urea
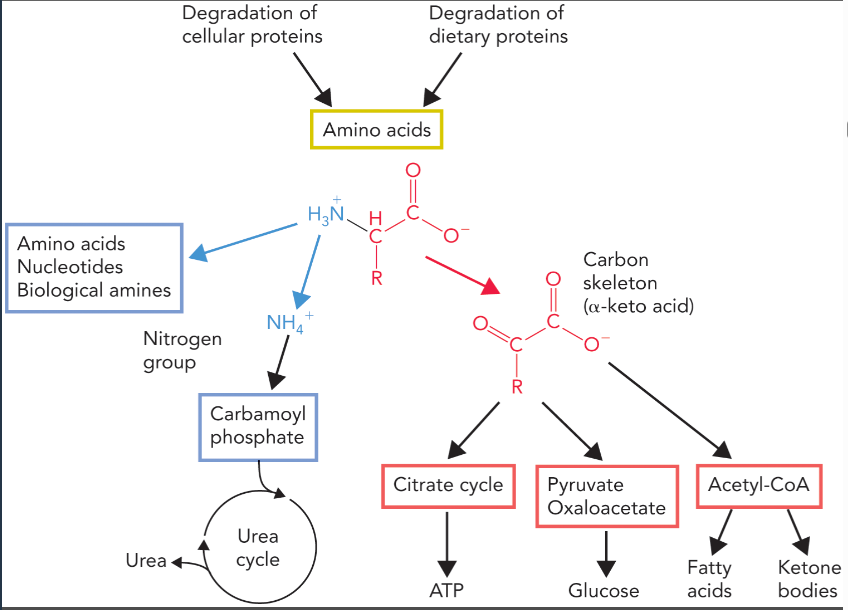
amino acids transported to the liver have three sources
digestion of dietary proteins
glutamine, which comes from glutamate and ammonium through glutamine synthase (in peripheral tissues)
alanine which is formed by alanine aminotransferase (removes excess nitrogen from skeletal muscle)
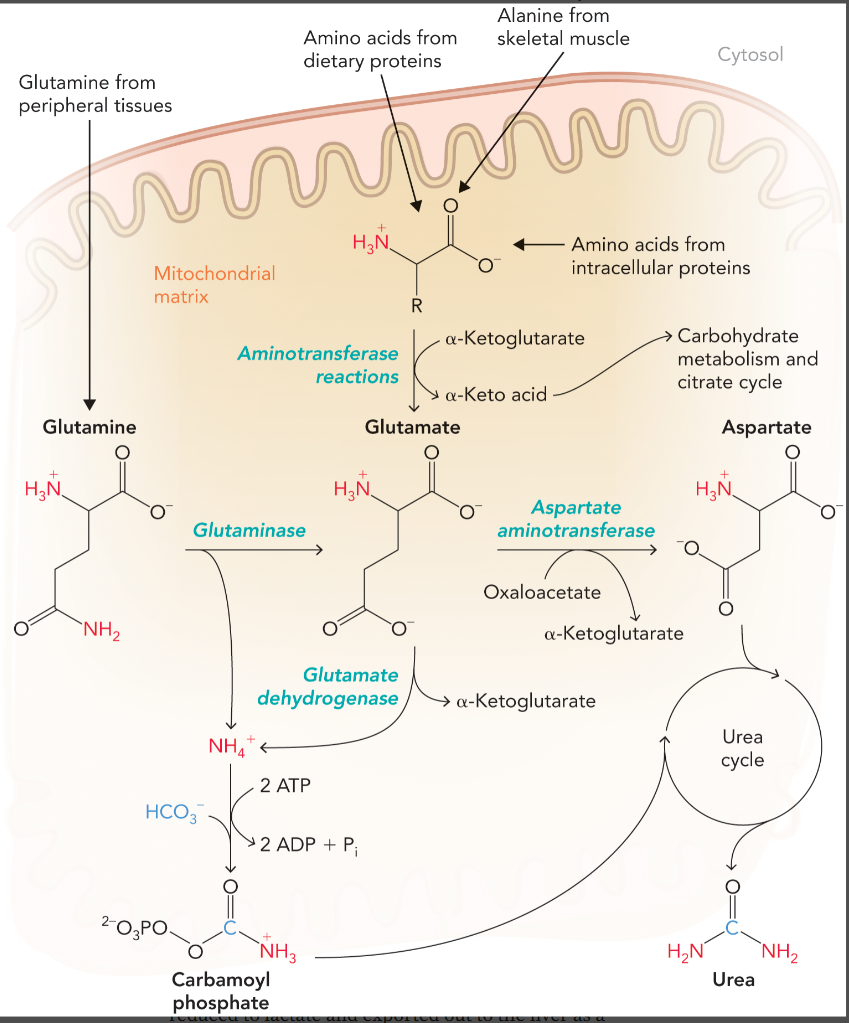
how ammonium enters the urea cycle in the liver
carbamoyl phosphate, the ammonium can come from either glutamine or glutamate
aspartate which is converted from glutamate through aspartate aminotransferase
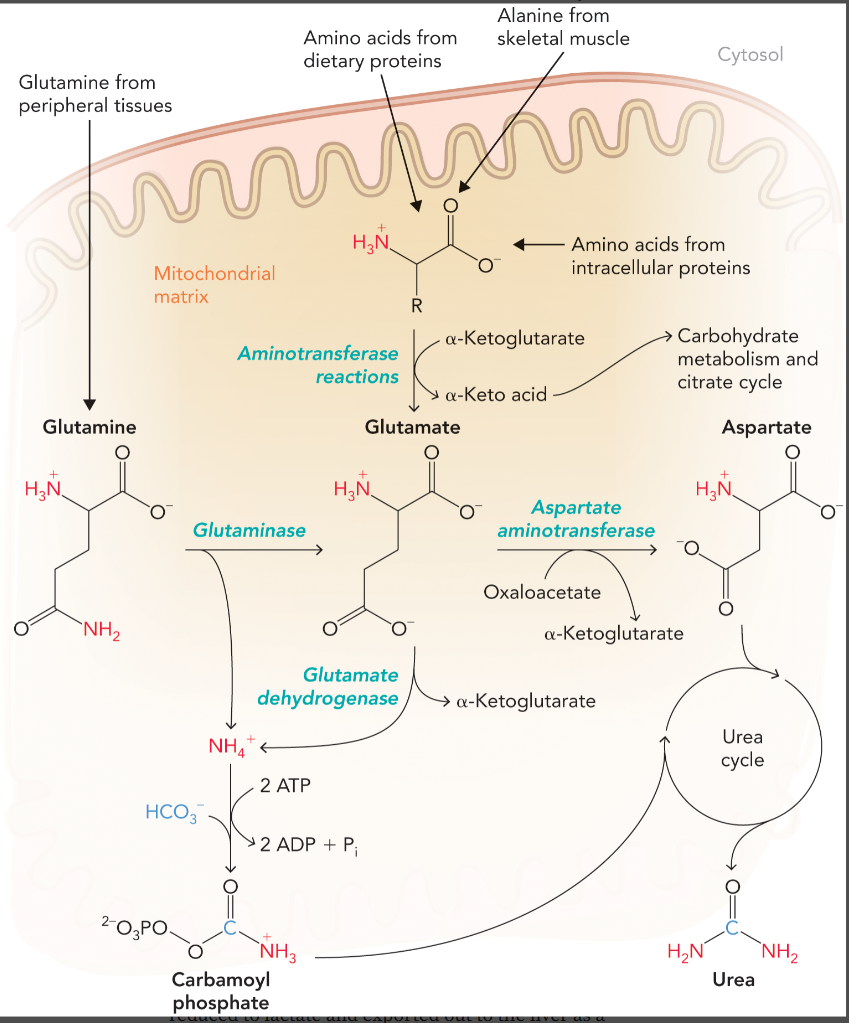
alanine-glucose cycle
removes excess nitrogen from amino acid catabolism during exercise
links nitrogen metabolizing reactions in muscle and liver cells with alanine as the nitrogen carrier
pyruvate is converted to alanine with alanine aminotransferase (uses glutamate)
alanine is exported to the blood and taken in by the liver to regenerate glutamate and pyruvate by the reverse reaction (deaminated)
glutamate is metabolized by glutamate dehydrogenase to release ammonium for urea synthesis
pyruvate is used to synthesize glucose via gluconeogenesis and exported back to the muscles
net reaction of the urea cycle

in humans, urea is synthesized in the liver and transported
through the blood to the kidneys where it is concentrated and excreted in urine
reactions of the urea cycle that take place in the mitochondria
mitochondria— carbamoyl phosphate synthetase I and ornithine transcarbamoylase
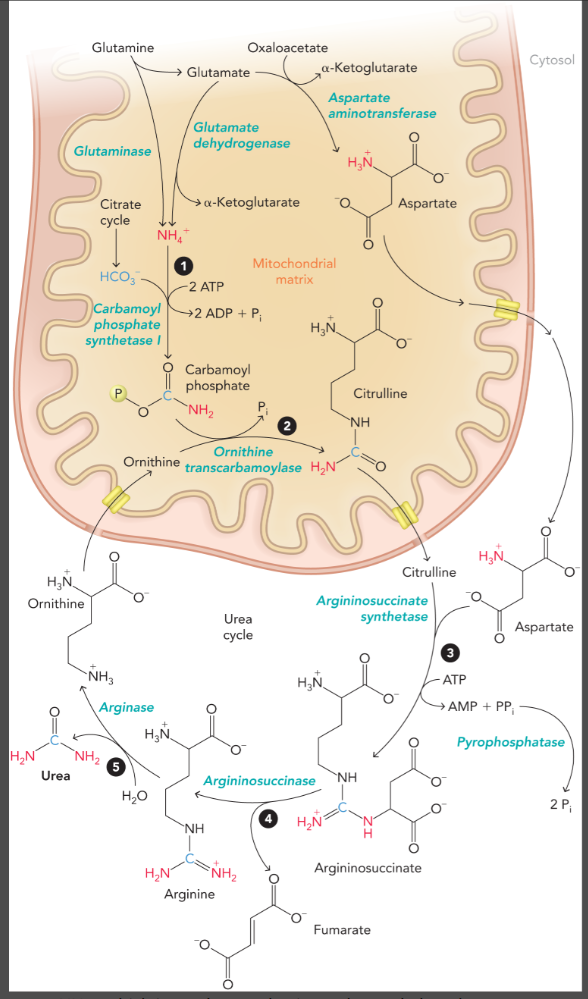
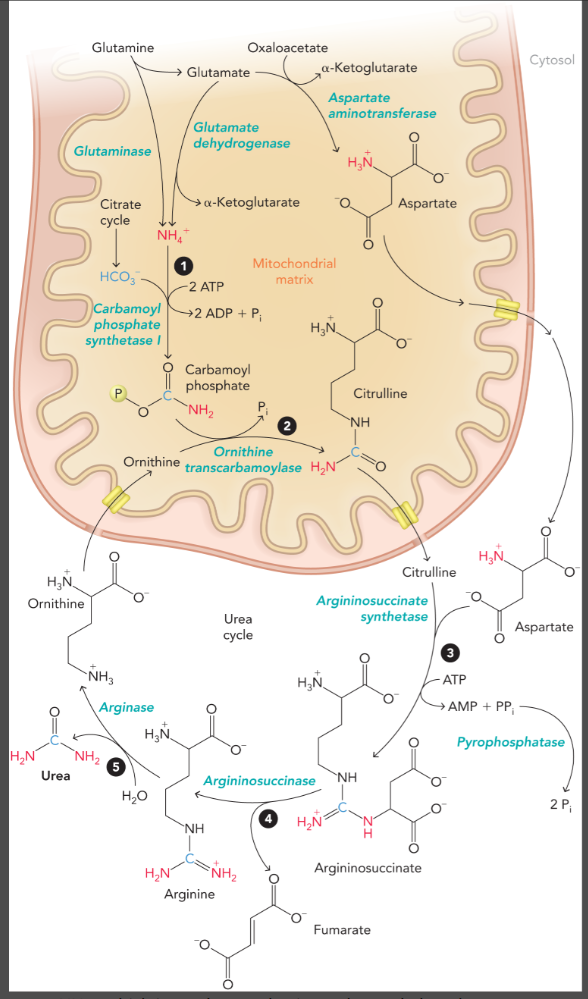
steps of the urea cycle
carbamoyl phosphate (first nitrogen atom) is formed in the mitochondrial matrix by ATP-dependent carbamoyl phosphate synthetase I
citrulline is formed from carbamoyl phosphate and ornithine by ornithine transcarbamoylase
citrulline is exported to the cytosol to form argininosuccinate from aspartate (second nitrogen atom) which is catalyzed by argininosuccinate synthetase and pyrophosphatase
argininosuccinate is cleaved by argininosuccinase to yield fumarate and arginine (arginine contains both nitrogen atoms)
arginase converts arginine to urea and ornithine to complete the cycle
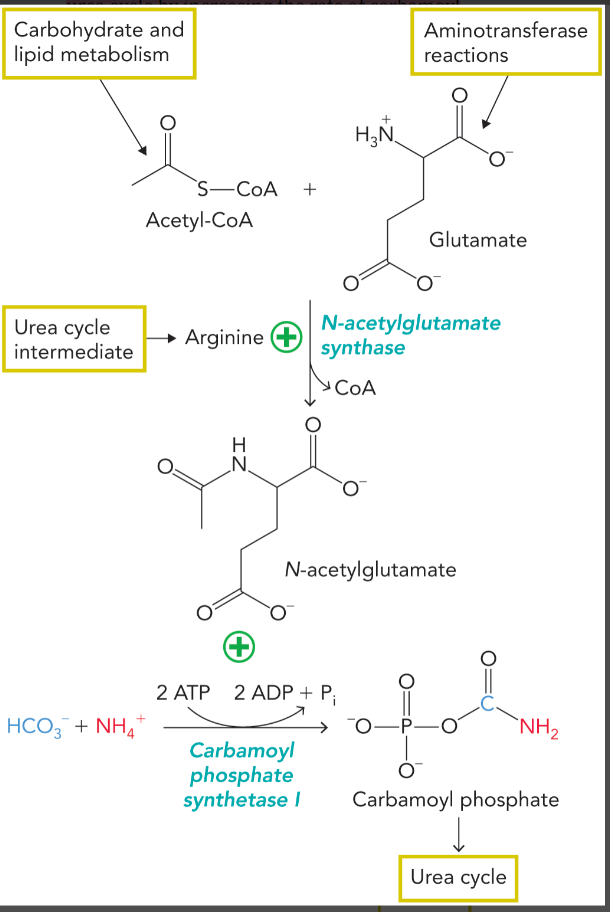
urea cycle regulation
carbamoyl phosphate synthetase I is positively allosterically regulated by N-acetylglutamate which is formed from glutamate and acetyl-CoA
formation of N-acetylglutamate is positively regulated by arginine
glutamate and arginine stimulate flux through the urea cycle by increasing the rate of carbamoyl phosphate synthesis
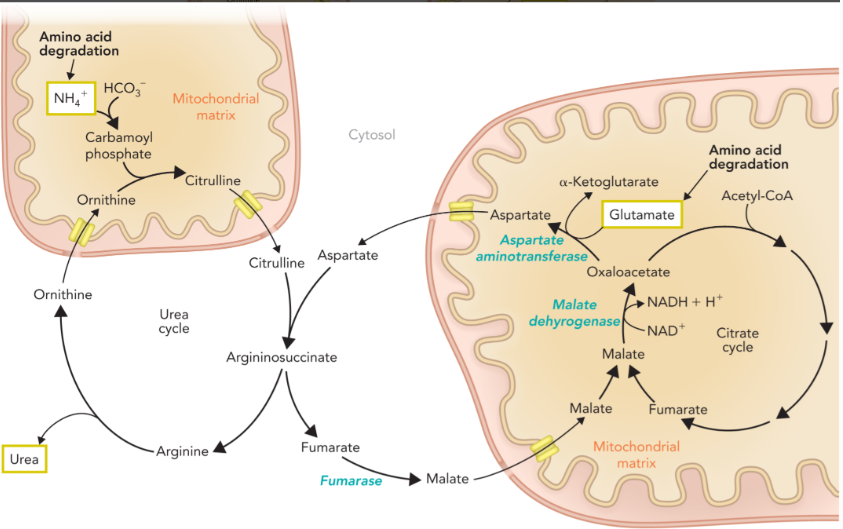
the krebs bicycle/ the aspartate-argininosuccinate shunt
fumarate is converted to malate in the cytosol by an isozyme of fumarase
malate is transported into the mitochondrial matrix through the malate-aspartate shuttle and converted into oxaloacetate though malate dehydrogenase
oxaloacetate is used as substrate with glutamate in the aspartate aminotransferase reaction to generate aspartate which is transported back to the cytosol
aspartate is then used to form argininosuccinate
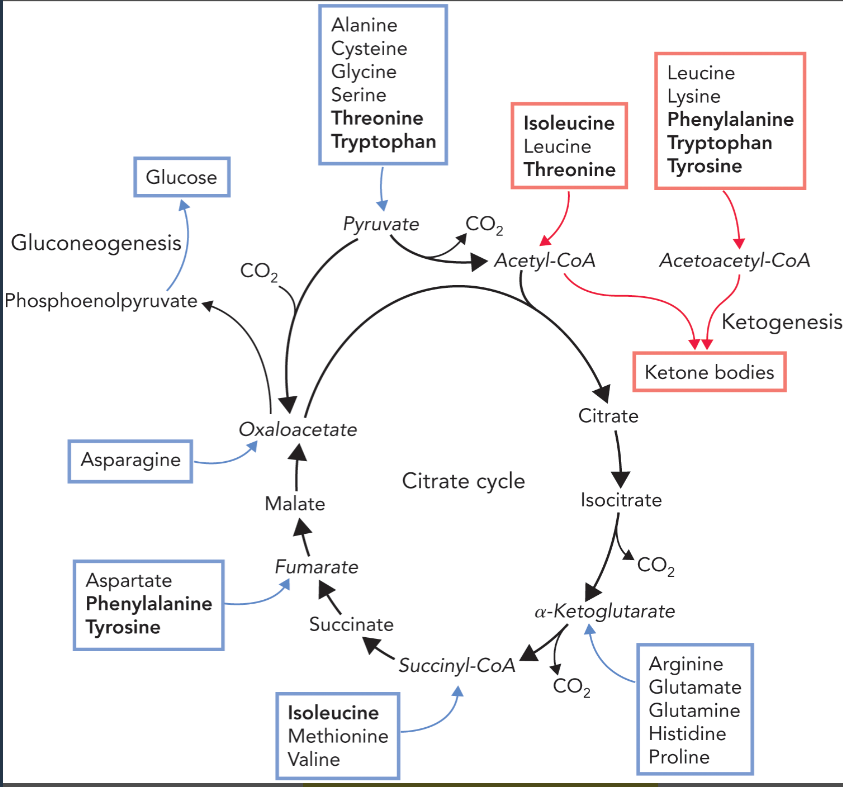
glucogenic amino acids (formed from amino acid catabolism)
amino acids whose carbon chains can be used to form glucose and glycogen via gluconeogenesis
pyruvate— ala, cys, gly, ser, thr, trp (group 1 degradation pathway)
alpha-ketoglutarate— arg, glu, gln, his, pro (group 2 degradation pathway)
succinyl-CoA— ileu, met, thr, val
fumarate— phe, tyr
oxaloacetate— asp, asn
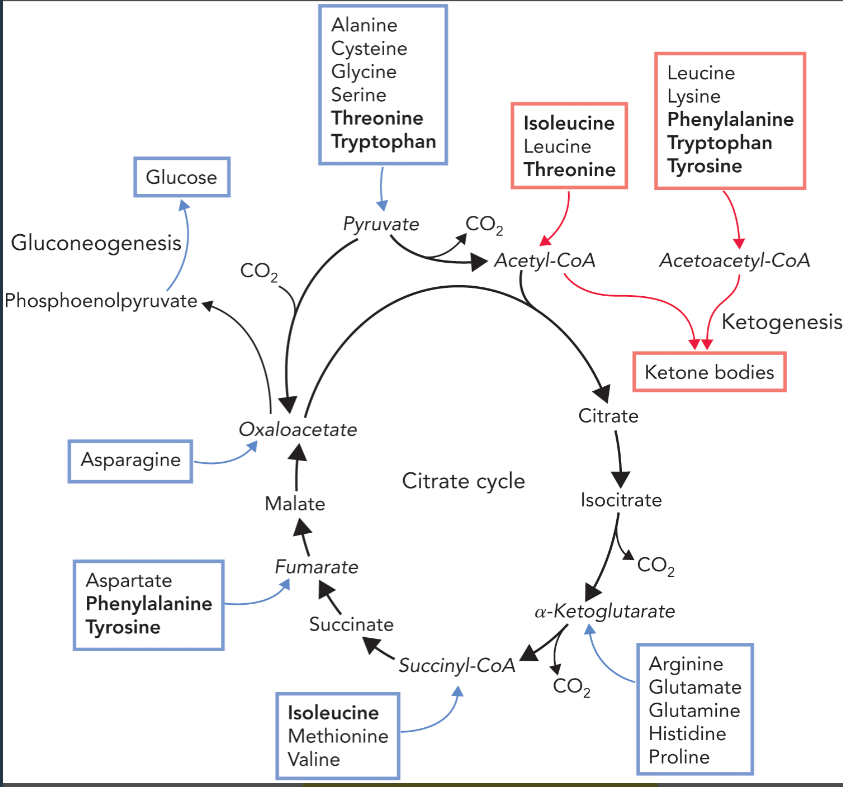
ketogenic amino acids (formed from amino acid catabolism)
amino acids whose carbon chains can be used to form ketone bodies
leu, ileu, thr, lys, phe, tyr, trp
essential amino acids
arginine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine
amino acids are derived from metabolic intermediates in
glycolysis, PPP, and the citrate cycle
tyrosine and phenylalanine come from phosphoenolpyruvate
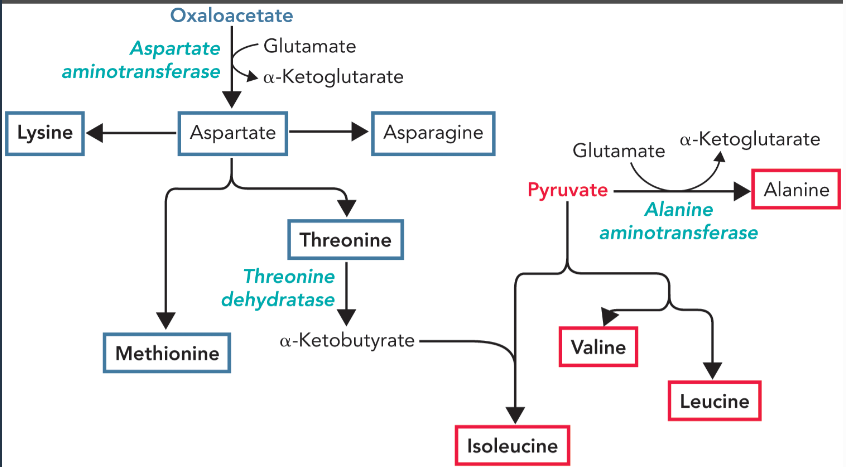
oxaloacetate derived amino acids
aspartate, asparagine, lysine, threonine, and methionine
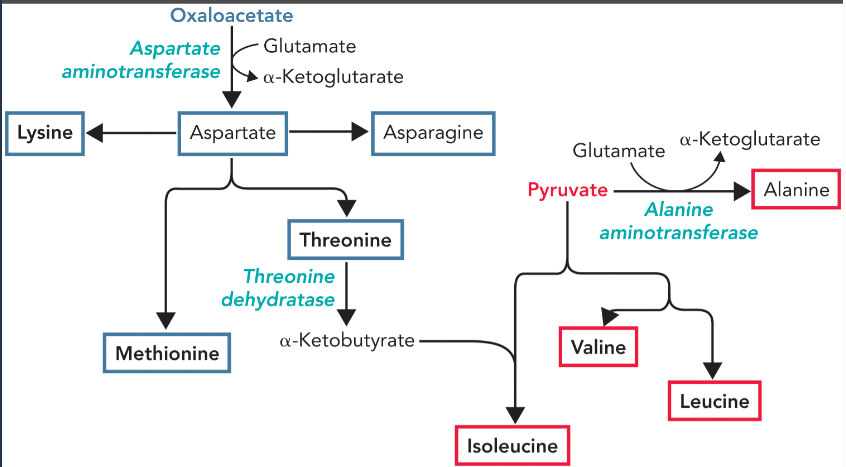
pyruvate derived amino acids
alanine, isoleucine, valine, and leucine
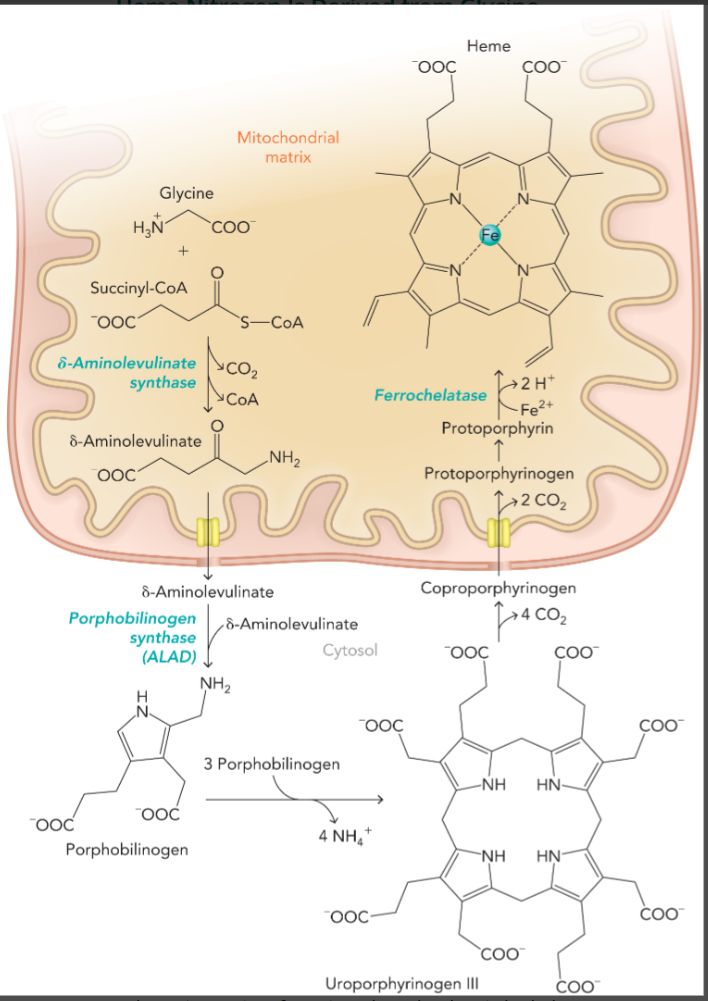
heme which is essential to cytochromes, hemoglobin, and myoglobin comes from
glycine
tyrosine is important in
metabolic signaling and neurotransmission, it is also the precursor to melanin pigments in hair and skin (mutations in tyrosinase cause albinism)
alkaptonuria and phenylketonuria
AKU causes black urine and prevents complete breakdown of tyrosine and phenylalanine
PKU causes elevated levels of phe in the body which can cause adverse neurological symptoms