IA #4
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What happens when antigens and antibodies combine? What do they make? 3 inactivate 1 trigger
they create antigen-antibody complexes
inactivates by neutralization (masks dangerous parts of bacterial exotoxins/viruses so pathogen can’t infect), agglutination (Ab bind to RBC to make cell-count Ag), and precipitation (Ag get larger and get out)
triggers complement activation
all enhance phagocytosis, trigger enhance phagocytosis/inflammation, trigger leads to cell lysis
What are the 4 jobs of Ab?
neutralization
agglutination
precipitation
complement activation
what 2 things are needed for lymphocyte activation? Why does it need the steps?
1) needs to bind to Ag
2) costimulation- additional chemical signal needed for activation usually an antigen
needed to make sure immune system isn’t activated by accident
What do helper t-cells produce? what is its job?
IL-2- costimulates B-cells, T helper and cytotoxic cells, activates NK cells
Where is IL-1 produces? What is its job?
produced by APC to state cycle and goes to CD4 cells to costimulate
Where to b and t lymphocyte precursors originate? where do they move to mature further? what happens during their maturation
precursors originate in bone marrow
precursors of T-cells migrate in blood to thymus and mature there, precursors of B-cells mature in red bone marrow
while maturing they become immunocompetent and self-tolerant
where do lymphocytes travel after they are immunocompetent?
still naive (not exposed to antigen), seed/colonize the secondary lymphoid organs (lymph nodes, spleen, etc) and blood/lymph where likely to encounter antigens
What are the two types of humoral immunity? What are the subtypes of these?
active- making memory cells against Ag, either naturally acquired (infection/contact w/ pathogen) or artificially acquired (vaccine (exposed to either dead piece or contained piece so don’t get sick), dead/attenuated pathogen, can sometimes cause reaction but doesn’t mean have it)
passive- no memory cells made/intervention, naturally acquired (Ab passed from mother to fetus through placenta/milk, not exposed so kids are sick more, Ab dont last forever) or artificially acquired (injection of exogenous Ab, when really sick so don’t have time for body to respond so have to give Ab)
What happens during the primary response to an antigen? How long does this take?
1) Ag binds to receptor on B-lymphocyte
2) these B-lymphocytes then proliferate and form clones called activated B cells
3) 2 activated B cells make 3 plasma/effector B cells, which secretes antibody molecules, and 1 memory b-cell primed to respond to same Ag
takes 5 days to make antibodies, peaks @ 10 days, @ 28 days Ab drop to 0
What happens during the secondary response to an antigen? How is this response different?
response is faster/stronger because made memory cells so no lag time bc already immunocompetent so Ab [ ] is higher/produced quicker because Ag-lymphocyte bonding receptor is stronger
1) memory cells make plasma cells which secrete Ab molecules (increases Ab [ ]) and more memory b-cells
What does the variability of thickness (other characteristics/structure) mean for the function? How many Ag can bind to the Ab monomer?
means different/specific Ag can bind
heavy chain is longer and thicker
1 Ab binds to 2 Ag
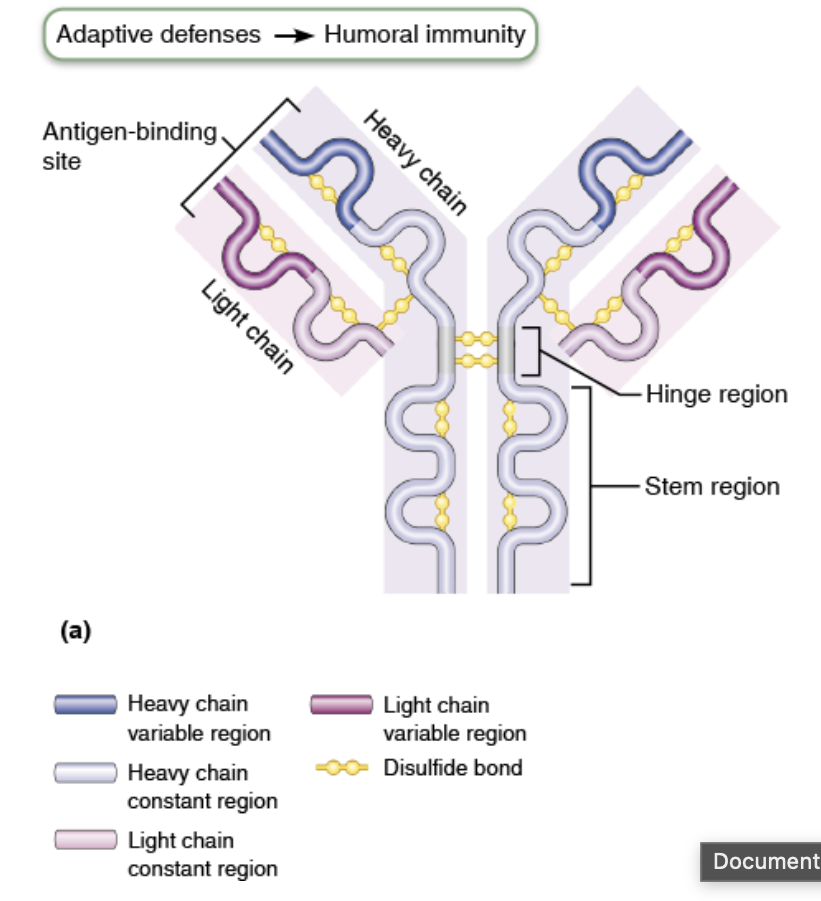
What is the function of an IgM (pentameter)? How is it secreted? What does it activate? What forms does it exist as? What is the function of that second form?
function of pentamer= readily activates complement, indicates current infection by pathogen eliciting IgM formation, potent agglutinating agent because Ag binding sites, B cell antigen receptor
secreted by plasma cells during the primary response and circulates in blood plasma
exists as a monomer and pentamer (5 united monomers)
function of monomer= Ag receptor on B cell surface which makes it immunocompetent
Where is IgA (dimer) found? What is its function? Does it exist in a monomer?
in body secretions (ex. saliva, sweat, intestinal juice, milk)
helps stop pathogens from attaching to epi cell surfaces
monomers exist in limited amounts in plasma
Where is IgD (monomer) found? What is its function?
found on B cell surface which functions as a B cell antigen receptor like IgM
What is the most abundant antibody? How much does it account for?
IgG- makes up 75-85% of circulating antibodies
What is the function of a IgG monomer?
main antibody in both secondary and late primary responses, readily activates complement, protects against bacteria, viruses, and toxins in blood/lymph, crosses placenta and confers passive immunity from mother to fetus
What end binds on a IgE monomer? To what does it bind? What does it trigger? What is it secreted by? When would levels rise?
stem end binds to mast cells/basophils, Ag binding to its receptor end triggers cells to release histamine and other chemicals that mediate inflammation/allergic reaction
secreted by plasma cells in skin, mucosa of GI and respiratory tracts, and tonsils w/ only traces actually in the plasma but more in GI and respiratory tract mostly
levels rise during severe allergic attacks or chronic parasitic infections in GI tract
How do APC bind to T helper cells
1) MCH II on APC presents exogenous (originates outside cell) Ag
2) T-cell receptor on inactive T-helper cell finds/binds which to Ag
3) T-helper cell turns into effector cells (mature T-helper cells) or memory cells
4) T-helper cells have surface protein that binds to MHC II and anchors it called CD4 protein
What types of cells are APCs (Antigen presenting cell) ?
b-cells, dendritic cells (most powerful with both MHC I and II), and other phagocytic cells
How do APC bind to T-cytotoxic cells
1) MHC I on APC presents exogenous (originates inside cell) Ag to T-cytotoxic receptor on inactive T-c cell
2) CD8 protein binds to MHC I to anchor
3) turns into effector or memory cells
How do Cytotoxic T cells attack infected and cancerous cells
1) Tc identifies foreign Ag on MHC I proteins and binds to target cell
2) Tc releases perforin and granzyme molecules through exocytosis
3) perforin molecules insert into target cell membrane, polymerize, and form transmembrane pours leading to cell lysis
4) granzymes activate enzymes that trigger apoptosis
What does the MHC I cell tell the immune system? To what cells does this happen?
tell immune system if cell is infected or not
in all body cells do this process for both foreign and self antigens- if competent won’t take the foreign antigen but if not body will fight
What is HIV? What does HIV target on a cellular level? What is AIDS?
HIV- human immunodeficiency virus, targets T-helper cells so can’t costimulate because no IL2 is being produced so its immunocompromised
AIDS- acquired immunodeficiency syndrome, no everyone with HIV develops AIDS especially with new drugs so it doesn’t spread to others
neither are autoimmune diseases bc healthy body cells aren’t attacked by immune system
What happens in the sensitization stage of an allergic reaction?
1) allergen invades body
2) plasma cells produce a lot of IgE antibodies
3) IgE antibodies attach to mast cells in body tissues and circulating basophils which triggers inflammation and leaky capillaries cause the mast cells have granules w/ histamine
What happens in the subsequent response of allergic reactions?
1) more of same Ag invades body
2) Ag combines w/ IgE attached to mast cells/basophils which triggers degranulation and releases histamines and other chemicals
3) histamine causes blood vessels to dilate/become leaky → edema, stimulates lots of mucus, causes smooth muscles to contract, asthma can happen is respiratory system is site of Ag entry
What are the 4 processes of respiration?
1) pulmonary respiration aka breathing
2) external respiration- O2 diffuses from lungs to blood, CO2 diffuses from blood to lungs
3) transport of resp gases- using blood O2 is transported from lungs to tissue cells, CO2 is transported from tissue cells to lungs and out of the body
4) internal respiration- O2 diffuses from blood to tissue cells, CO2 diffuses from tissue cells to blood through capillaries
What four things help trap debris, heat up air, and moisten the air using mucous membranes?
cilia on mucosa, nasal conchae, nasal meatuses, and vestibular fold aka false vocal cord
Why is it advantageous for the hyaline cartilage of the trachea to be a horseshoe shape?
the trachealis allows for some give if someone is stalling something bigger
During cellular respiration how much CO2 comes out of one glucose?
6 CO2
order of structures from nose to lungs
nasopharynx → oropharynx → larynogopharynx → larynx → trachea → branches to 2 primary bronchi
what is the larynx made of? what is its function?
cartilaginous, acts as a sound chamber for reverberation w/ vocal cords
what is the function of the tracheal cartilage?
helps keep airway open and protects it so air can get to the lungs
Why are mens thyroid cartilage and laryngeal prominence aka Adam’s apple bigger?
they are a target for testosterone
what is the function of the cricothyroid and cricotracheal ligament?
provide movement to get more or less air to lungs
What is pleurisy?
inflammation in pleural cavity, loses lubrication in layers → friction, sometimes more fluid → harder to breathe, might drain the fluid and/or give antibiotics
What is the function of the smooth muscle on the terminal bronchioles?
contracts to narrow airway during inflammatory responses (ex allergies, asthma, etc.) or can get bigger if need more air (ex exercise), meds can relax these like albuterol
direction of air flow after trachea
trachea → left main/primary bronchus → lobar/secondary bronchus → segmental/tertiary bronchus → 1 mm → < 1mm → terminal bronchiole (end of conducting zone) → respiratory bronchiole (start of respiratory zone, have alveoli for gas exchange) → alveolar ducts surrounded by alveolar sacs w/ individual alveoli
Alveoli
very thin, simple squamous epithelium, single, flat layer where O2 and CO2 diffuses