VSPER Shapes
0.0(0)
Card Sorting
1/19
Earn XP
Description and Tags
note: flashcards terms are formatted in the order of [hybridization] [# of bonding directions] [# of lone pairs] [bond angle] and definitions are the molecule geometry
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
20 Terms
1
New cards
VSEPR theory
valence shell electron pair repulsion theory
2
New cards
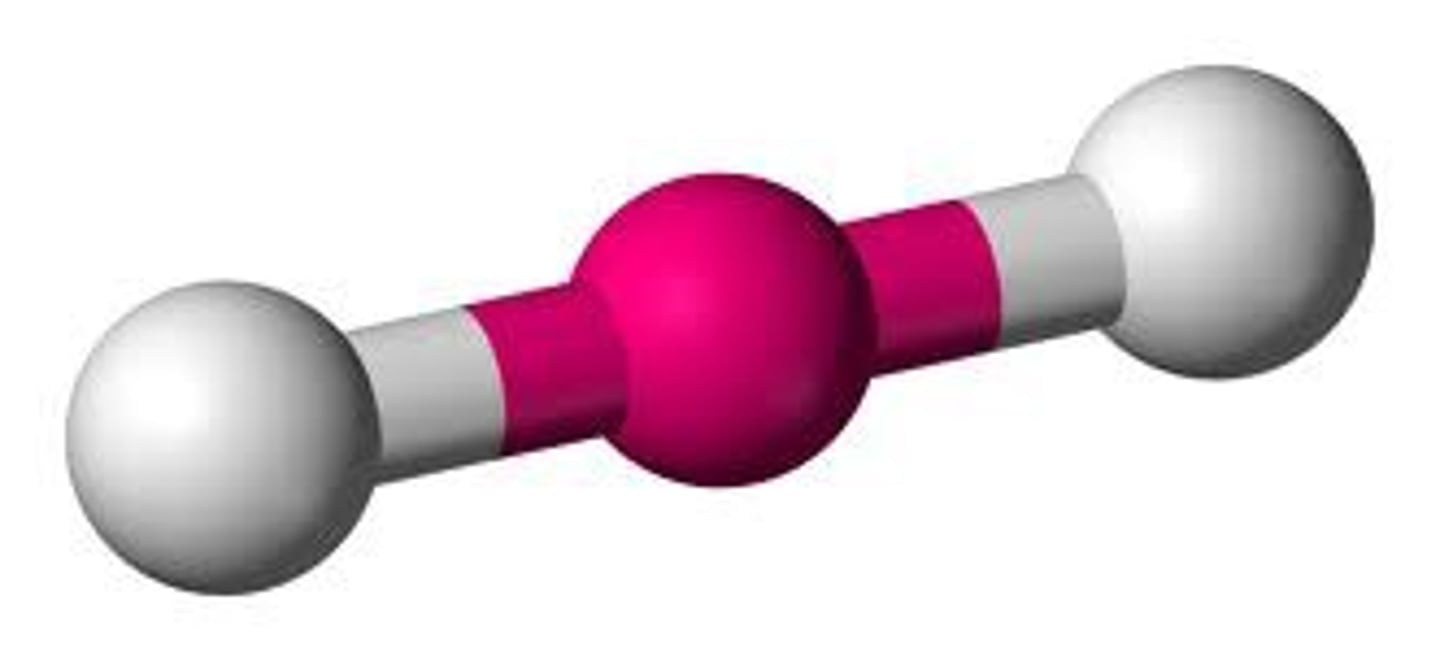
sp 2 0 180
linear
3
New cards
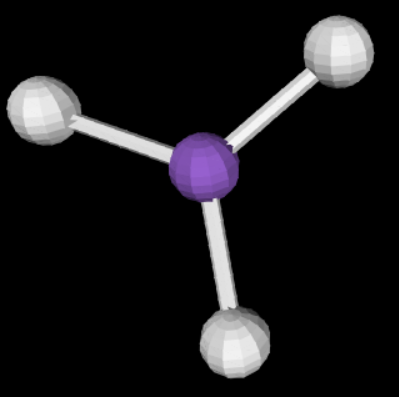
sp2 3 0 120
trigonal planar
4
New cards
sp2 2 1 <120
bent

5
New cards
sp3 4 0 109.5
tetrahedral
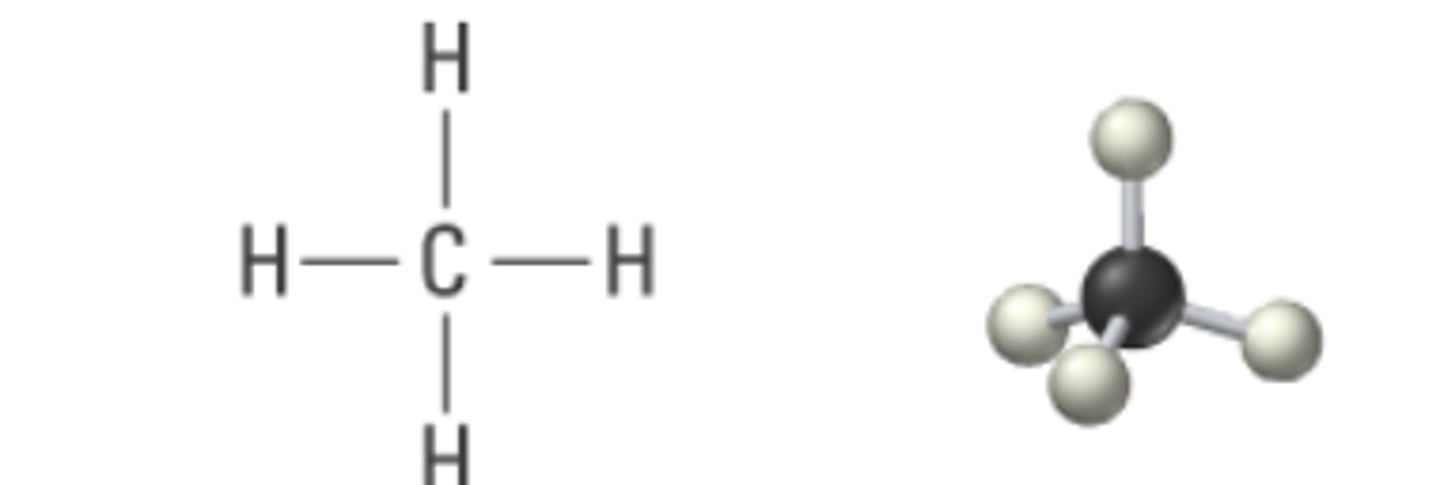
6
New cards
sp3 3 1 <109.5
trigonal pyramidal

7
New cards
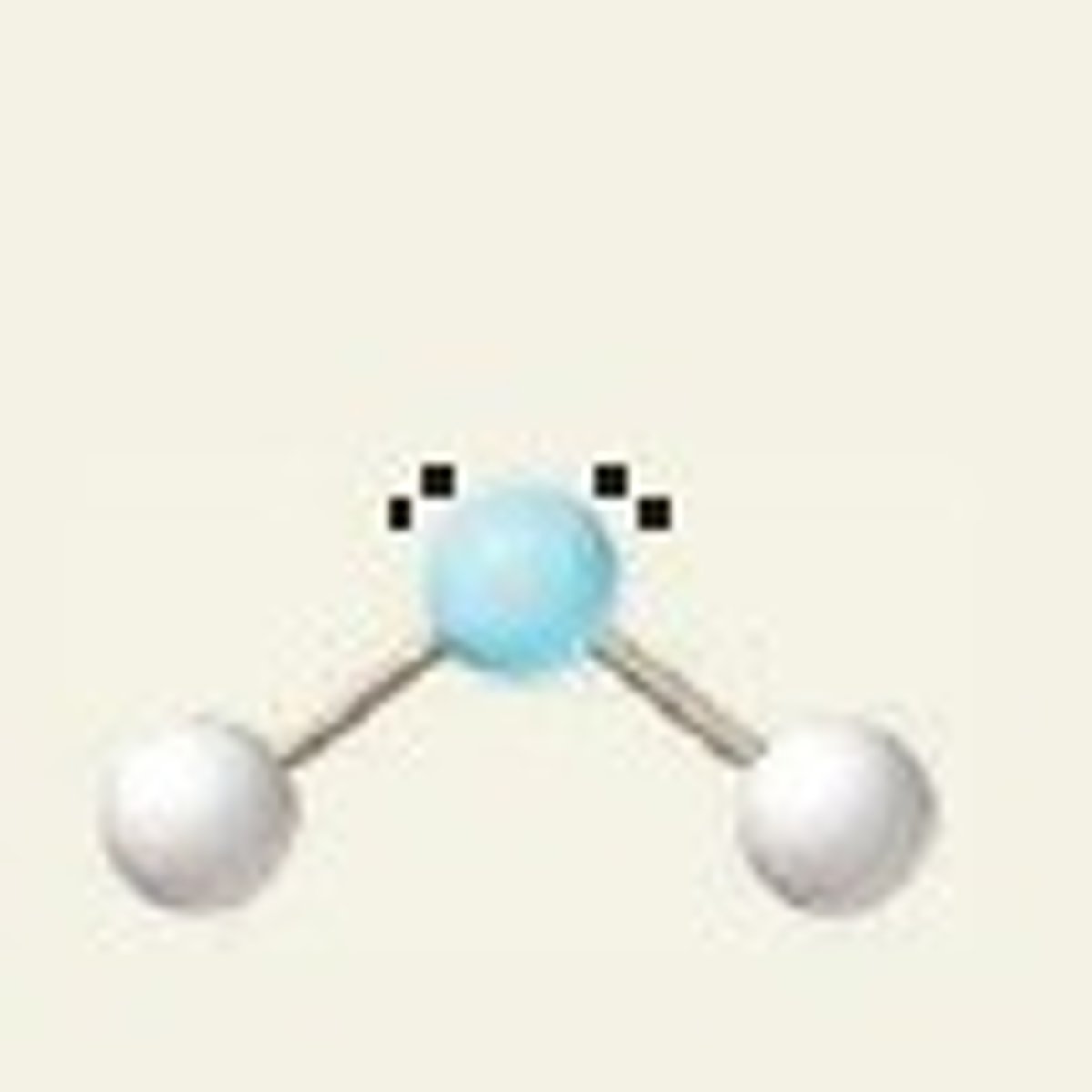
sp3 2 2 <109.5
bent
8
New cards
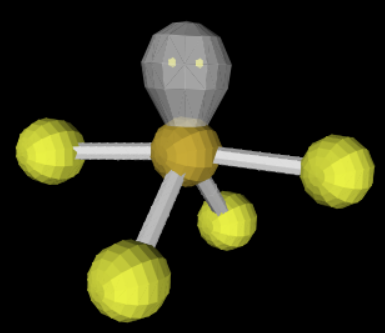
sp3d 5 0 120/90
trigonal bipyramidal
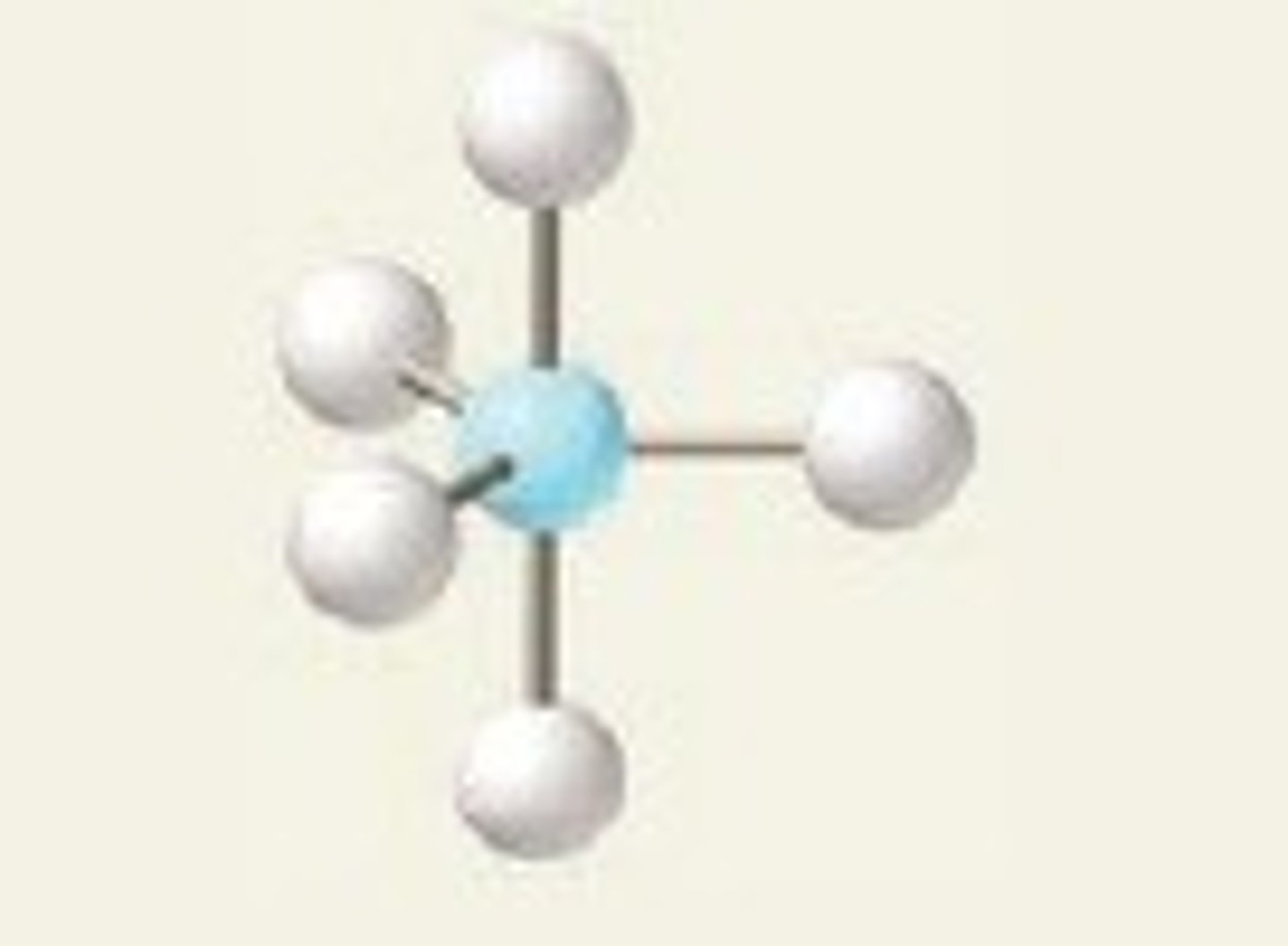
9
New cards
sp3d 4 1 <120/<90
seesaw
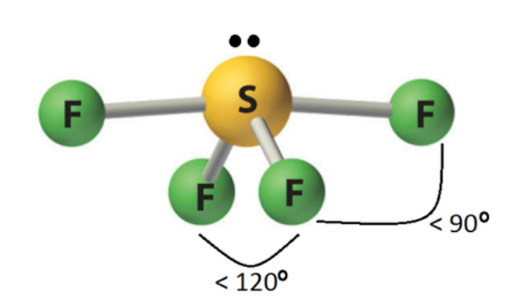
10
New cards
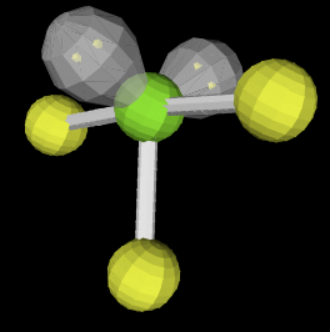
sp3d 3 2 <90
T shaped
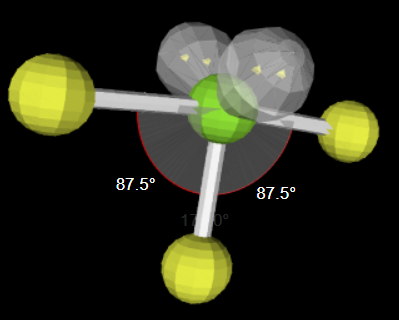
11
New cards
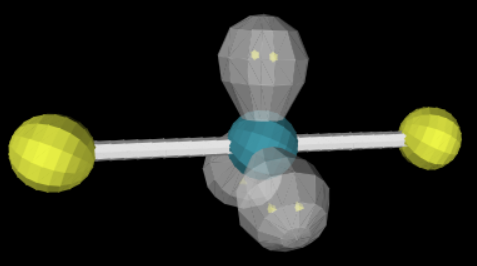
sp3d 2 3 180
linear
12
New cards
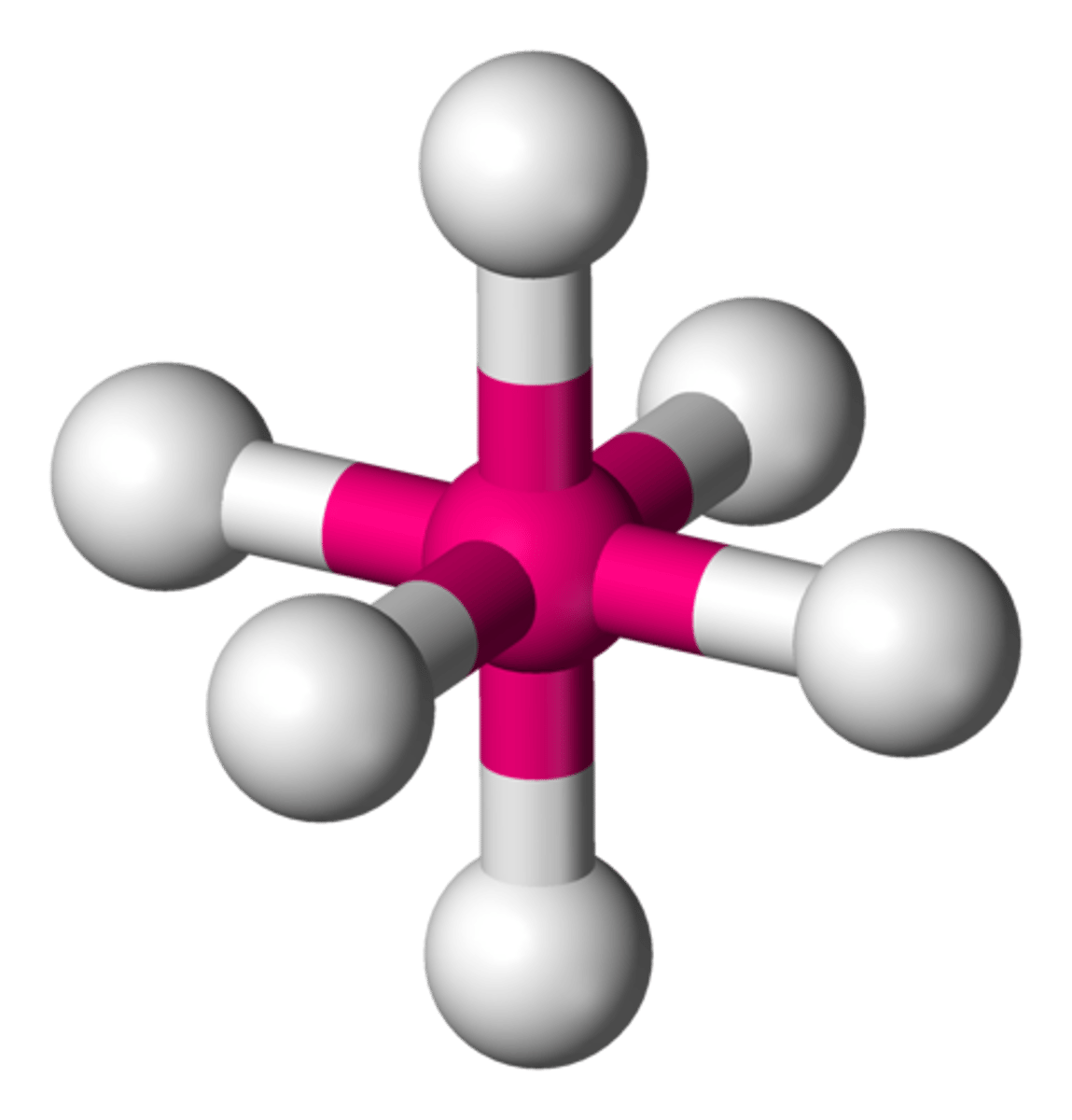
sp3d2 6 0 90
octahedral
13
New cards
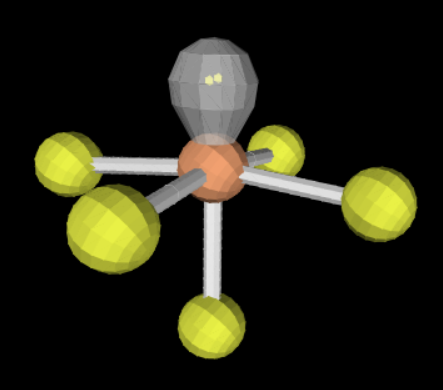
sp3d2 5 1 <90
square pyramidal
14
New cards
sp3d2 4 2 90
square planar
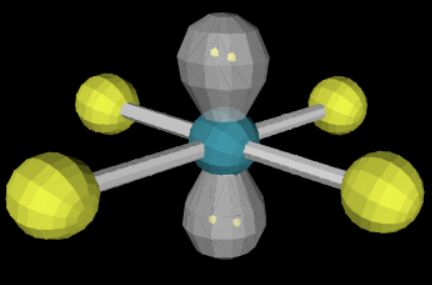
15
New cards
sp3d2 3 3 90
T-shaped
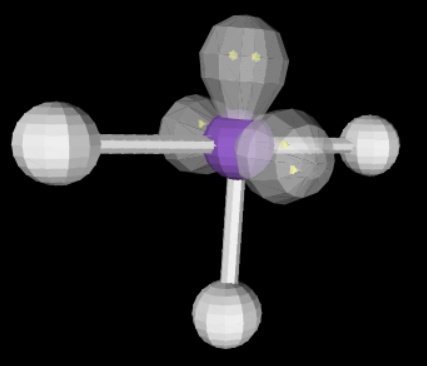
16
New cards
sp3d2 2 4 180
linear
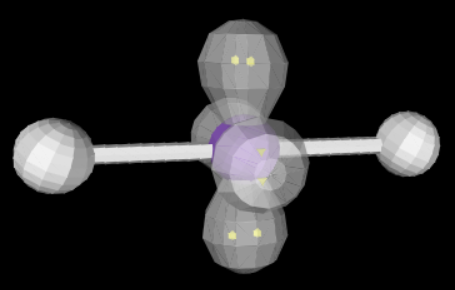
17
New cards
linear
sp
18
New cards
trigonal planar
sp2
19
New cards
trigonal bipyramidal
sp3d
20
New cards
octahedral
sp3d2