Group 7
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
what colour and state is fluorine at room temperature
pale yellow gas
what colour and state is chlorine at room temperature
green gas
what colour and state is bromine at room temperature
orange/ brown liquid
what colour and state is iodine at room temperature
grey/black solid
what colour vapour does iodine very easily form
purple
what happens to the melting and boiling points going down group 7
they increase → volatility decreases down group
what type of molecules are halogens
diatomic
why does mpt decrease down the group
atomic radius increases
more electron shells
outermost electrons are further away from positive nucleus so is less strongly attracted to it
what is the trend in bond enthalpies down group 7 between atoms of group 7 diatomic molecules, and what is the exception to this
e.g. energy needed to break covalent bond between Br-Br vs energy needed to break I-I
higher bond enthalpy = stronger covalent bonds
bond enthalpies decrease down group 7 = bonds strengths decrease going down the group
exception = fluorine = smaller bond enthalpy than chlorine and bromine
lone pairs on fluorine get so close to each other in a fluorine molecule that they cause repulsion which decreases the bond strength
what is the trend in electronegativites down group 7
electronegativity decreases down group
larger atoms / atomic radius increases / more shells → outermost electron is further away from nucleus
weaker attraction between nucleus and shared pair of electrons
what type of structures are halogens with what forces between the diatomic molecules
simple molecular structure
weak VDWs
when a halogen reacts with a metal, what sort of agent is it?
oxidising agent
accepts an electron from metal atom to become an ion with a 1- charge
what happens to the oxidising power of the halogens as you go down the group
decreases→ halogens get less reactive
halogens ability to accept an electron decreases
define electronegativity
how strongly an atom attracts the electrons in a covalent bond towards itself
order halogens in order of reactivity
fluroine, chlorine, bromine, iodine
what happens to the reducing power of halide ions as you move down the group
increases going down the group → ions are more likely to donate electrons going down group
why does reducing power of halide ions increase going down the group
halide ions become larger
outermost electron gets further away from nucleus
outermost electron experience more shielding due to more shells
weaker electrostatic force of attraction between outermost electron and positive nucleus
electron lost more easily → reducing power increases
what 2 substances can you use to test for halides
silver nitrate
ammonia
give the full equation and half equation for the reaction of chloride ion and silver nitrate
AgNO3 (aq) + NaCl (aq) → AgCl (s) + NaNO3 (aq)
Ag+ (aq) + Cl- → AgCl (s)
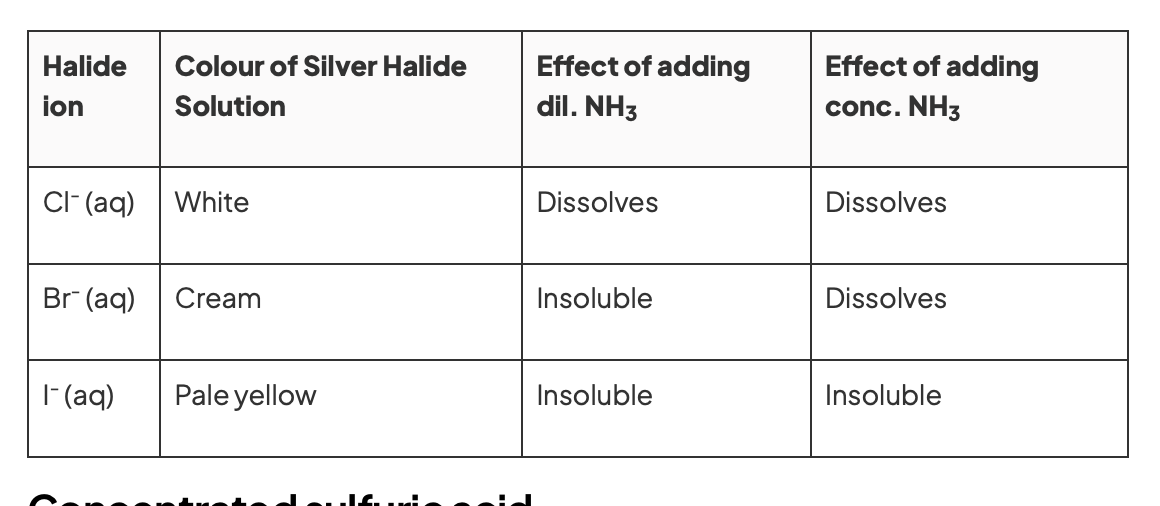
when testing for halide ions suing silver nitrate, list the colour of the precipitates formed
with chloride
with bromide
with iodide
white
cream
yellow
why do you add ammonia to the precipitates after doing a halide test with silver nitrate
as white white, cream and yellow precipitates can look very similar
describe the test of adding ammonia to the AgX precipitates and the results
dilute followed by concentrated ammonia is added to silver halide solution
precipitate redissolves in dilute ammonia to form colourless solution→ halide = chloride
precipitate doesn’t redissolve in dilute ammonia but does in concentrated → halide = bromide
precipitate doesn’t redissolve in either dilute or concentrated ammonia → halide = iodide
reaction of halide ions with silver nitrate and ammonia solutions
learn this table:
okay
when testing for halides using silver nitrate, why do you have to add nitric acid (aq) first to the solution being tested
removes any other ions that could give a precipitate with silver nitrate
e.g. carbonate ions
CO32- + 2H+ → CO2 + H2O
give the chemical equation for when AgCl reacts with dilute ammonia
AgCl + 2NH3 → (Ag(NH3)2)+ → Cl-
give the chemical equation for when AgBr reacts with concentrated ammonia
AgBr + 2NH3 → (Ag(NH3)2)+ → Br-
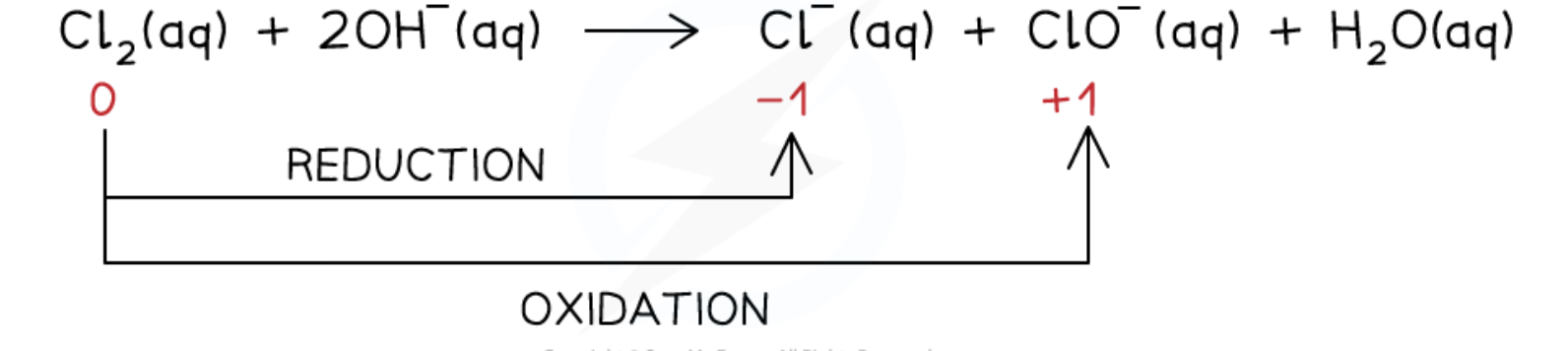
what is a disproportionation reaction
a reaction in which the same species is both oxidised and reduced
give 2 examples of a disproportionation reaction
chlorine with dilute alkali
- chlorine gets oxidised and reduced at same timechlorine with water
give the equation for the disproportionation reaction of chlorine in cold alkali
chemical
ionic
Cl2 (aq) + 2NaOH (aq) → NaCl (aq) + NaClO (aq) + H2O (l)
Cl2 (aq) + 2OH- (aq) → Cl- (aq) + ClO- (aq) + H2O (aq)
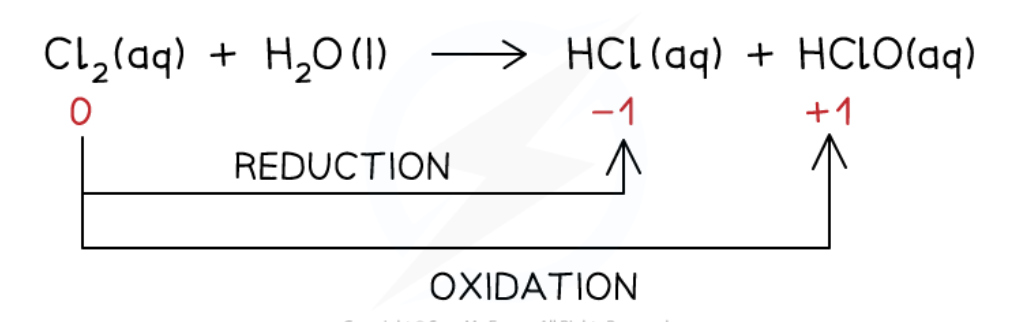
increase in ox. no. from 0 to +1 in ClO- (aq) = oxidation
decrease in ox. no from 0 to -1 in Cl- (aq) = reduction
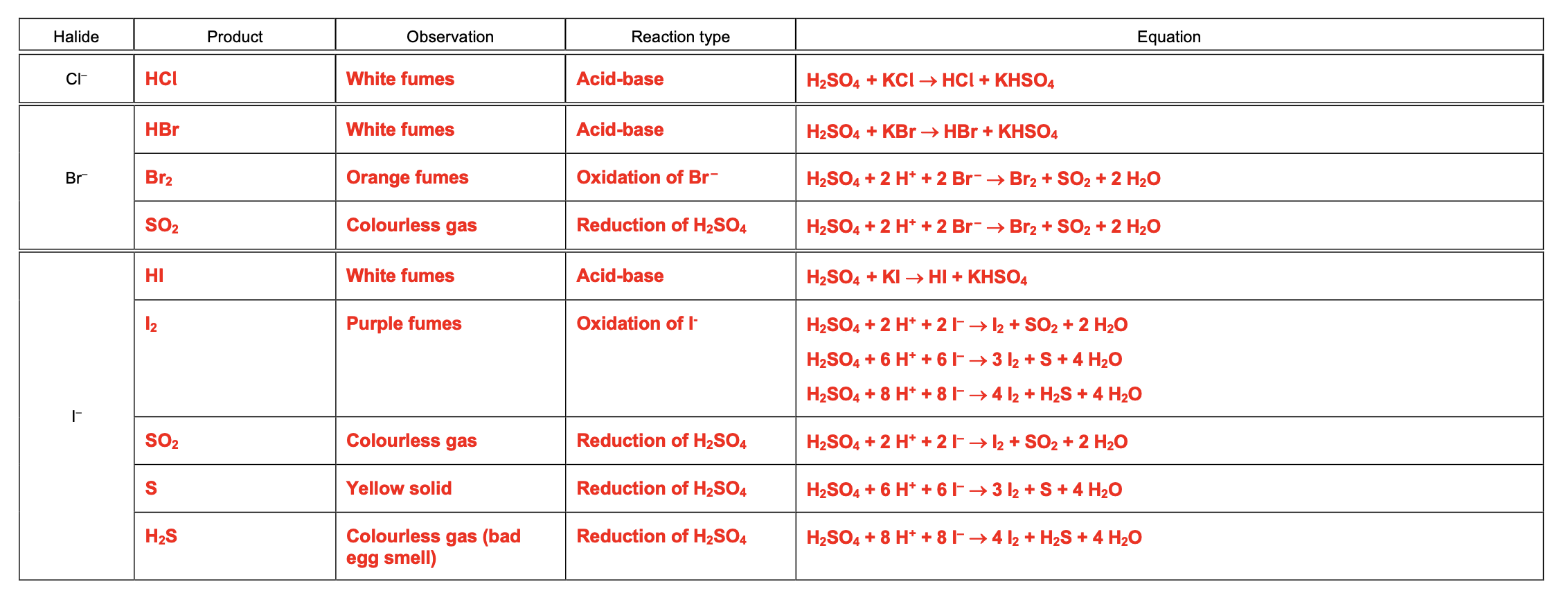
learn this table for the reaction of halides with concentrated sulphuric acid
okay
what can chlorine be used for
clean water and make it drinkable
give the equation for the disproportionation reaction of chlorine and water
Cl2(aq) + H2O(l) → HCl(aq) + HClO(aq)