Organtic Chemistry 2
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
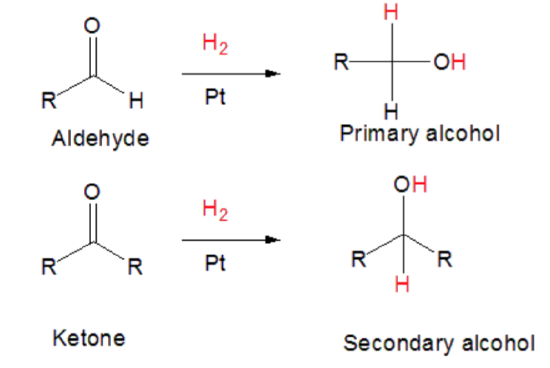
Hydrogenation of a Ketone / Aldehyde
With a metal catalyst, an aldehyde or ketone looses O-C bond and gains an H.
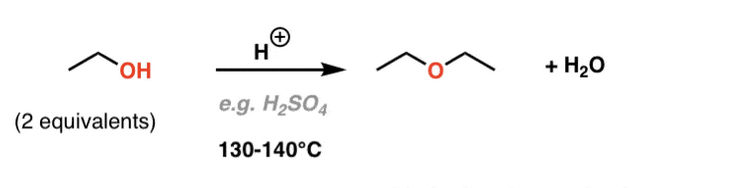
Alcohol dehydration to an ether
2 alcohols with catalytic H+ and heat produce an ether and H2O
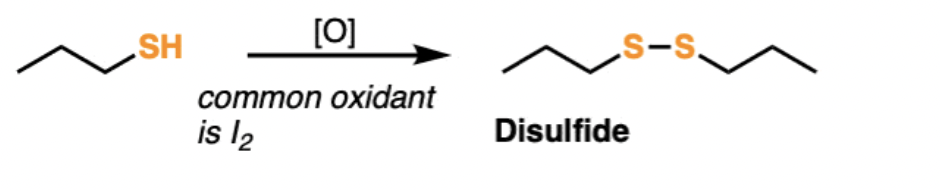
Oxidization of a Thiol
2 thiols loose their hydrogens and bond together with the addition of a catalytic acid to forma disulfide
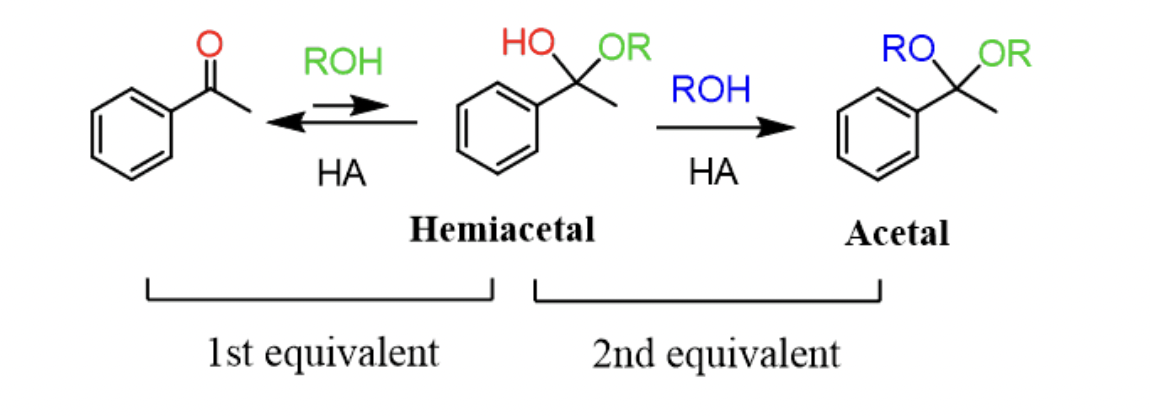
Hemiacetals and Acetals
Aldehyde condensation reaction with alcohol (and catalytic acid) to form hemiacetal and then again to form acetal. Involves 1 R group, and the chains C-O-C-O-H and C-O-C-O-C
Hemiketals and ketals
Ketone condensation reaction with alcohol (and catalytic acid) to form hemiketal and then again to form ketal. Involves 2 R groupa, and the chains C-O-C-O-H and C-O-C-O-C
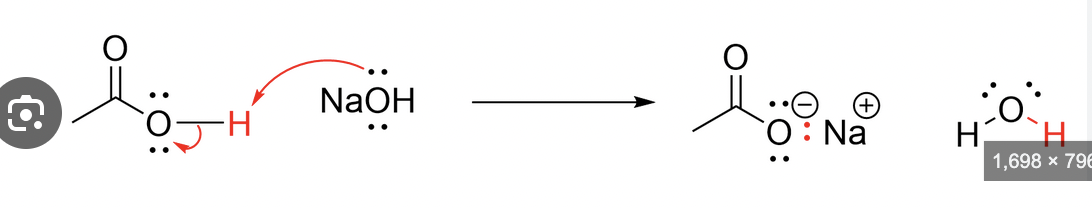
Carboxylic acid-base reaction
Carboxylic acid plus a stong base (KOH or NaOH) looses a hydrogen from OH, and becomes negative with a floating positive Na, plus water
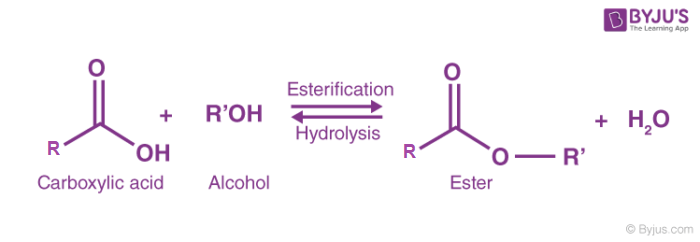
Esterification Reaction
Condensation & dehydration reaction between a carboxylic acid and an alcohol to produce and ester and water with the addition of catalytic acid and heat
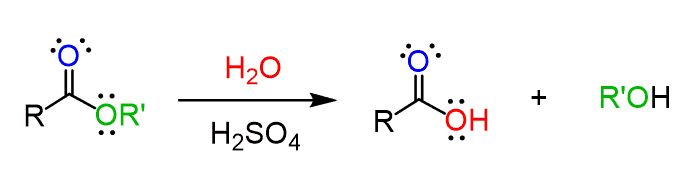
Acid Catalyzed Hydrolysis of an Ester
Water breaks an ester into a carboxylic acid and an alcohol with the addition of heat and catalytic H+
Base Catalyzed Hydrolysis of an Ester
“Soponification” A base separates an ester into a carboxylate salt (O- and a floating Na+) and an alcohol

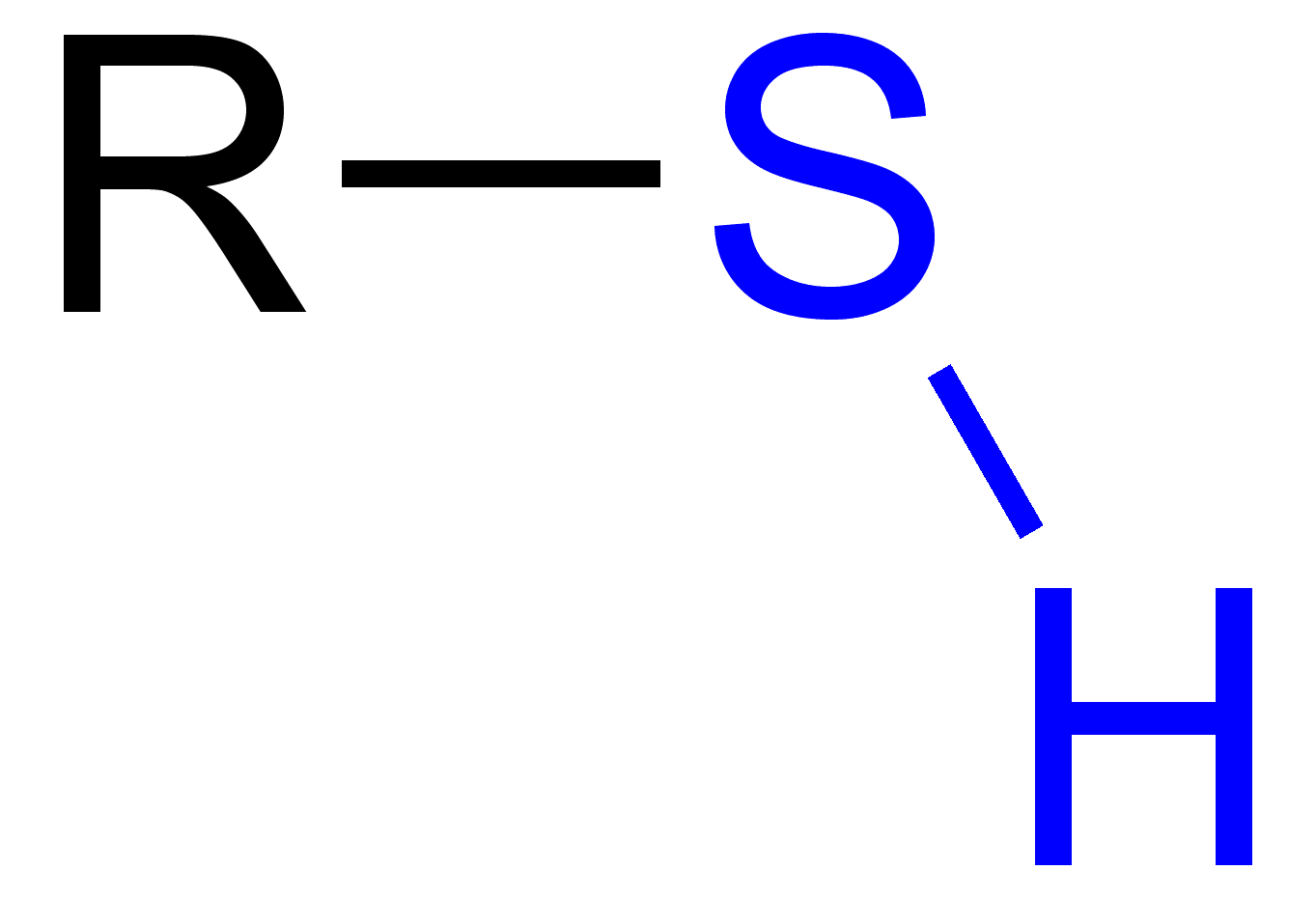
Thiol
SH sulfur hydrogen flanked by hydrocarbon