Biochem Exam 2 Questions
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Which of the following forms of DNA is left-handed?
a. Z-form
b. C-form
c. A-form
d. B-form
e. single stranded
a. Z-form
Which of the following vitamin/cofactor participates in one or two electron transfer reactions:
a. Pyridoxal phosphate
b. Biotin
c. FAD
d. NAD
e. Thiamine
c. FAD
When glucose levels are high, glucose transport into red blood cells exhibits saturation kinetics. When glucose concentrations are low, transport of glucose across the membrane depends upon the sodium ion concentration. What of the following explain these observations?
a. passive diffusion at high [glucose]; primary active transport at low [glucose]
b. passive diffusion at high [glucose]; secondary active transport at low [glucose]
c. facilitated diffusion at high [glucose]; primary active transport at low [glucose]
d. facilitated diffusion at high [glucose]; secondary active transport at low [glucose]
e. facilitated diffusion at both low and high [glucose]
d. facilitated diffusion at high [glucose]; secondary active transport at low [glucose]
Which of the following best describes an example of transverse asymmetry in membranes?
a. proteins phase separate and cluster into patches on red blood cell membranes
b. lipids phase separate and cluster to form lipid rafts on red blood cell membranes
c. cholesterol is evenly distributed on the inner and outer leaflet of red blood cell membranes
d. membranes “melt” when thermal energy converts lipids from anti to gauche conformations
e. phosphatidylserine is found exclusively on the inner leaflet of red blood cell membranes
e. phosphatidylserine is found exclusively on the inner leaflet of red blood cell membranes
Which of the following statements about the fluid-mosaic model is NOT true?
a. mosaic refers to the distribution of proteins in the membrane
b. fluid refers to the hydrocarbon tails of the lipids in the membrane
c. the phospholipid bilayer is the organizational feature
d. proteins and lipids are free to move laterally in the membrane
e. peripheral proteins interact exclusively with cholesterol
e. peripheral proteins interact exclusively with cholesterol
An uncompetitive, reversible enzyme inhibitor is expected to:
a. associate with both E and ES, alter the value of Km
b. associate with both E and ES, and the value of kcat
c. associate with ES, and leave the value of Vmax unchanged
d. associate with ES, and alter the values of Km
e. associate with ES and alter the overall Keq of the reaction
d. associate with ES, and alter the values of Km
Which of the following conditions would be expected to cause a decrease in the rate of a chemical reaction that is susceptible the specific base catalysis:
a. increasing pH
b. increasing buffer concentration
c. decreasing pH
d. decreasing buffer concentration
e. increasing ionic strength
c. decreasing pH
In the catalytic triad common to many serine proteases, _______ increases the basicity of ______, thus allowing deprotonation of ________ to serve as a nucleophile:
a. S-H-D
b. H-S-D
c. E-H-S
d. D-H-S
e. E-H-C
d. D-H-S
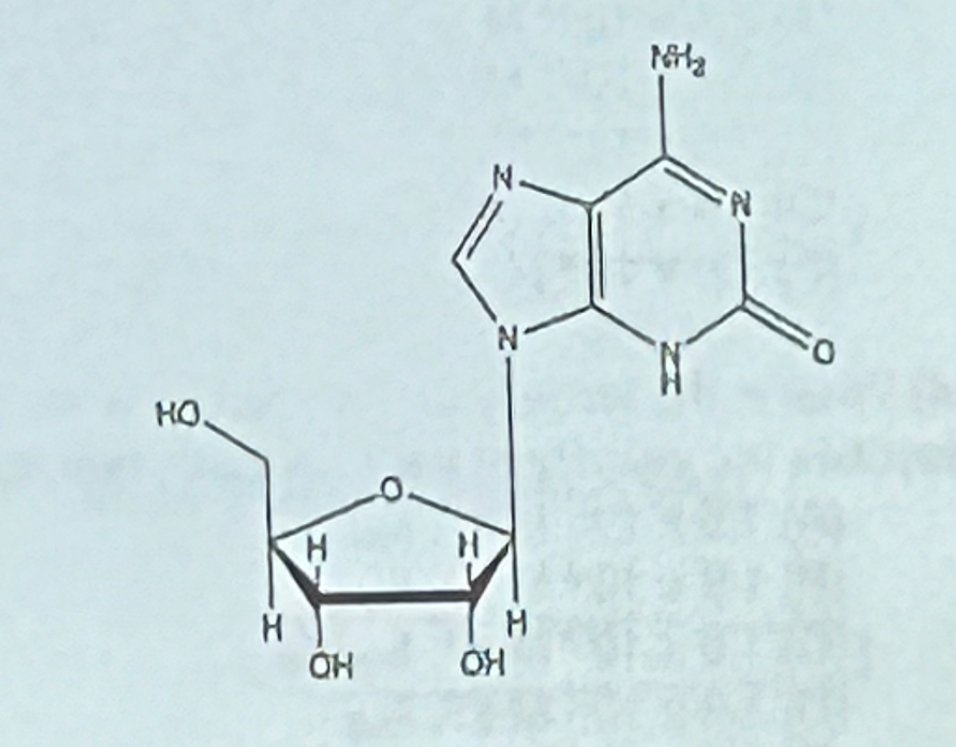
What structure is shown at the right?
a. adenine
b. guanine
c. adenosine
d. guanosine
e. none of the above
e. none of the above
The removal of 2,3-bisphosphoglycerate from hemoglobin will:
a. increase the affinity of hemoglobin for O2
b. decrease the affinity of hemoglobin for O2
c. have no effect on the affinity of hemoglobin for O2
d. produce the same effect as lowering the pH
e. produce the same effect as increasing CO2 concentrations
a. increase the affinity of hemoglobin for O2
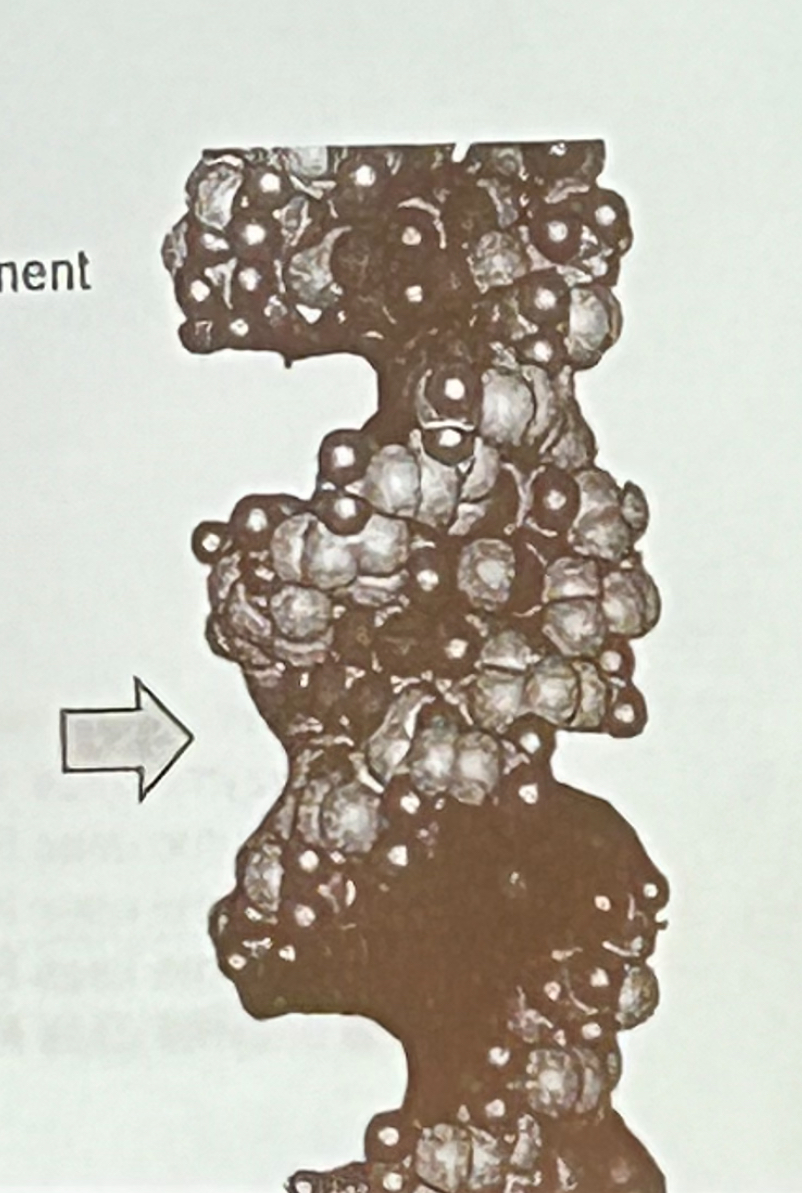
The arrow point to the image at the right depicts which component of the DNA helix?
a. minor groove
b. major groove
c. phosphodiester backbone
d. helical twist
e. propeller twist
a. minor groove
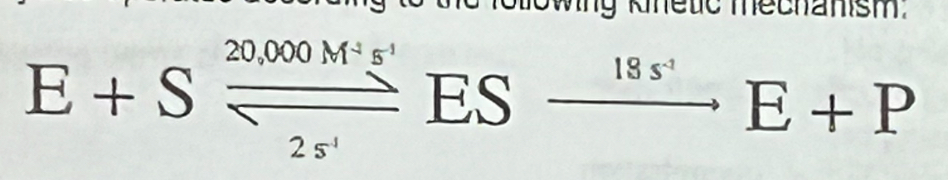
The spontaneous decarboxylation of lipoic acid occurs with a half-life of 90 million years, whereas lipoic acid decarboxylase operates according to the following kinetic mechanism:
What is the dissociation constant for the ground state enzyme-substrate complex?
a. 0.001M
b. 0.0001 M
c. 0.001 M^-1
d. 0.0001 M^-1
e. 2 s^-1
b. 0.0001 M
What is the dissociation constant for the enzyme-transition state complex?
a. 2.4 × 10^-16 M
b. 1.4 × 10^-20 M^-1
c. 1.6 × 10^11 M
d. 1.4 × 10^-20 M
e. 1.0 × 10^4 M
d. 1.4 × 10^-20 M
What is the velocity of the reaction when [E] = 10^-8 M and [S] = 10^-5 M; and which rate constant describes the velocity of the enzymatic reaction under these specific conditions?
a. 1.6 × 10^-9 M s^-1; kcat
b. 1.6 × 10^-9 M s^-1; k1
c. 1.8 × 10^-9 M s^-1; kcat/Km
d. 1.8 × 10^-7 M s^-1; kcat
e. 1.8 × 10^-7 M s^-1; kcat/Km
c. 1.8 × 10^-9 M s^-1; kcat/Km
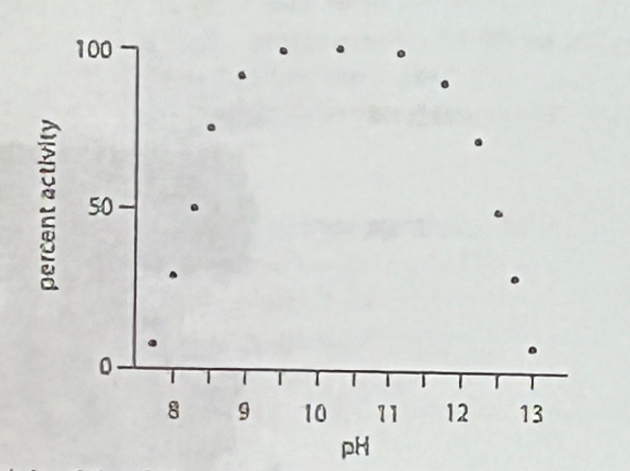
Which of the following statements is consistent with this data:
a. the enzyme uses Y as a general acid during catalysis
b. the enzyme uses R as a general base and C as a general acid during catalysis
c. the enzyme uses K as a general acid and H as a general base during catalysis
d. the enzyme uses R as a general acid and C as a general base during catalysis
e. the enzyme uses K as a general base and H as a general acid during catalysis
d. the enzyme uses R as a general acid and C as a general base during catalysis
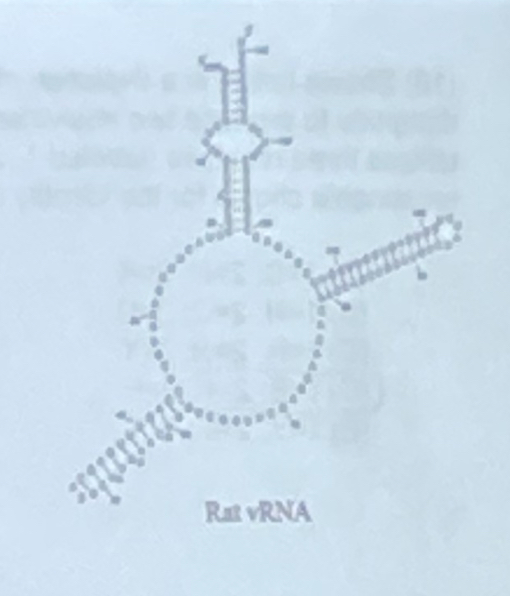
The structure at the right represents what level of RNA structure:
a. primary
b. secondary
c. tertiary
d. quaternary
e. phosphodiester backbone
b. secondary
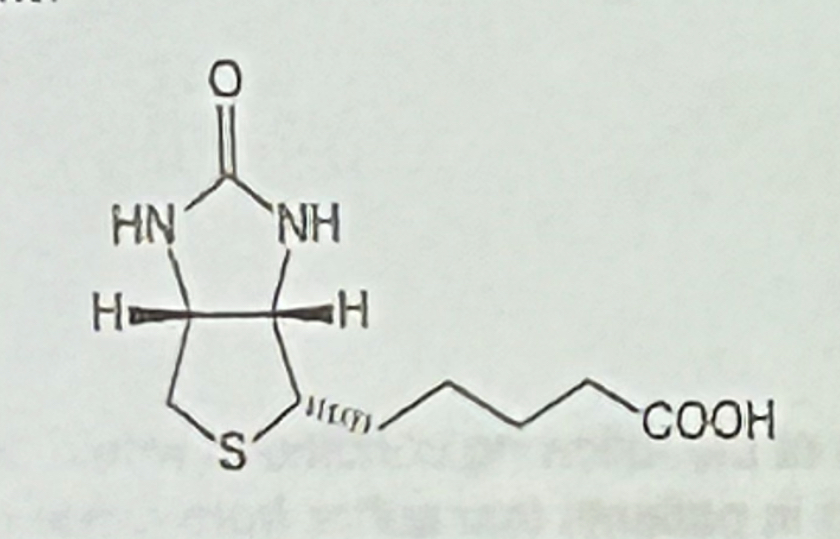
Which description fits the vitamin shown to the rights?
a. acts as an acyl group carrier
b. readily forms a carbanion
c. acts as a carboxyl group carrier
d. forms a Schiff base
e. participates in redox reactions
c. acts as a carboxyl group carrier
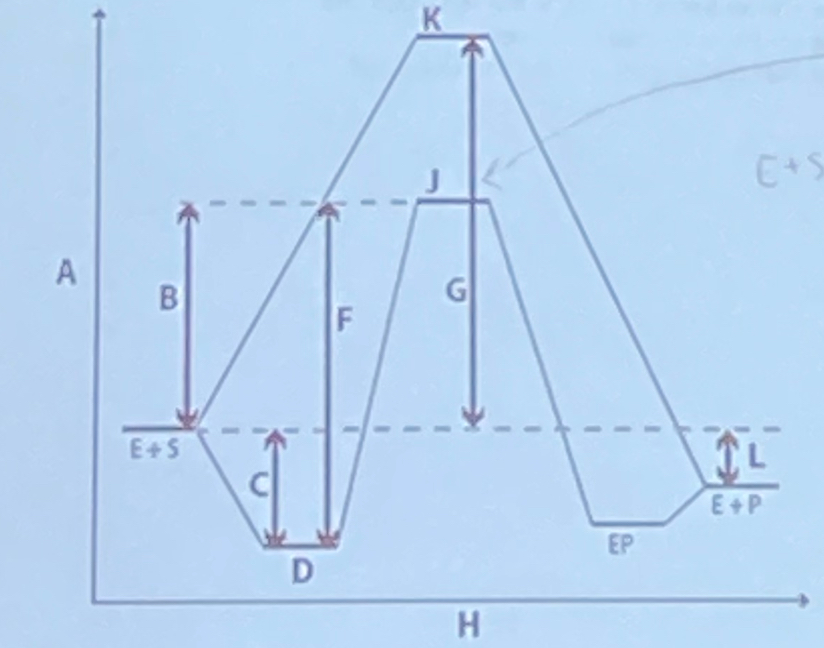
In the reaction coordinate diagram shown below, the arrow labeled B matches which description?
a. a process with a rate constant of kcat
b. ΔG‡ at sub-saturating substrate concentrations
c. ΔG‡ at saturating substrate concentrations
d. a process with a rate constant of knon
e. -RTln(Keq)
b. ΔG‡ at sub-saturating substrate concentrations
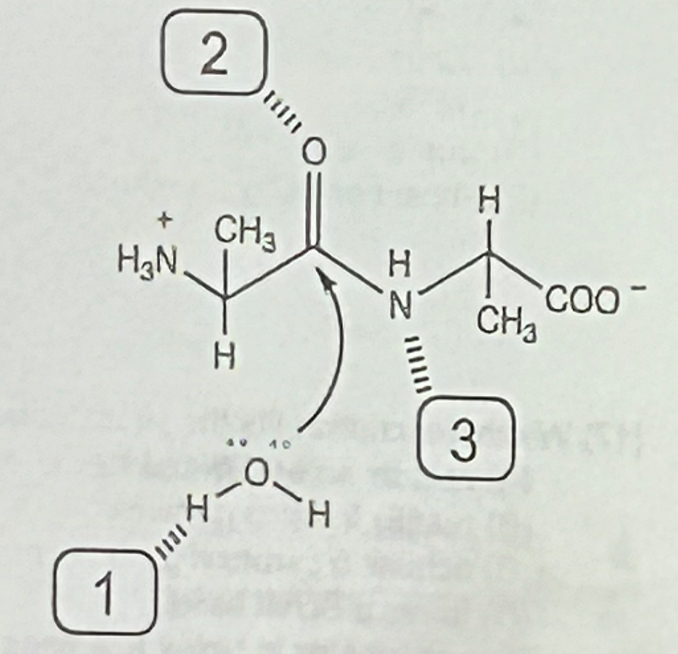
Shown below is a depiction of the active site of a protease that catalyzes the hydrolysis of a dipeptide to produce two equivalents of alanine. The enzyme has maximal activity at pH = 7.0 and utilizes three residues (labeled 1,2 and 3) to catalyze the reaction. Which of the following is a reasonable choice for the identity of the three residues?
a. 1=C, 2=H, 3=K
b. 1=H, 2=C, 3=D
c. 1=R, 2=N, 3=Y
d. 1=E, 2=K, 3=R
e. 1=Y, 2=E, 3=C
d. 1=E, 2=K, 3=R
Which of the following conditions would be expected to STIMULATE the polymerization of hemoglobin in patients that suffer from sickle cell anemia?
a. a decrease in the concentration of 2,3-bisphosphoglycerate in the red blood cell
b. an increase in the acidity of the red blood cell
c. a decrease in the concentration of CO2 in the red blood cell
d. a decrease in the acidity of the red blood cell
e. an increase in the Na+ concentration in the red blood cell
b. an increase in the acidity of the red blood cell