Lecture 3-4 -- Chronic Inflammation and Healing
1/27
Earn XP
Description and Tags
These flashcards cover key concepts regarding inflammation, healing processes, and outcomes as discussed in the lecture on Inflammation and Healing.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What are the four possible outcomes of acute inflammation?
Complete resolution
Progression to chronic inflammation
Healing by fibrosis
Abscess formation
What happens if acute inflammation completely resolves?
Return to normal vascular permeability
Drainage of fluid and proteins into lymphatics
Drainage of macrophages (pinocytosis)
Macrophages remove remaining fluid and small particles
Phagocytosis of degenerated neutrophils
Dead or dying neutrophils are cleared by macrophages
Phagocytosis of necrotic debris
Macrophages digest damaged tissue and cellular debris.
Removal of macrophages
Macrophages either leave via lymphatics or undergo apoptosis once cleanup is complete
What factors are likely to result in chronic inflammation?
Persistent infections
Mycobacteria, certain fungi and parasites
Prolonged irritation
Foreign material, exogenous and endogenous toxins
Cellular immune response: Autoimmunity/ Viral infections
Which inflammatory cells are typically seen in chronic inflammation?
Macrophages
Lymphocytes
Plasma cells (Specialised B cells)
Mast cells (especially in parasitic infections)
Eosinophils (especially in parasitic infections)
Sometimes neutrophils (in chronic suppurative 化膿 inflammation)
How do macrophages accumulate in chronic inflammation?
Continued recruitment of monocytes from circulating blood
Once one macrophage is activated, it induces the endothelium to express the right receptors to recruit more monocytes out of the circulating pool
Local proliferation of macrophages in tissues
Immobilisation/retention of macrophages at the site of inflammation
Cytokines are released to keep those macrophages in place
What are the main roles of macrophages as inflammation progresses?
Phagocytosis
Antigen presentation (via MHC II)
Macrophages phagocytose bacteria (Extracellular pathogen) → Peptide fragments are produced via lysozyme → Peptide then binds to MHC II molecules → CD4+ helper T cells recognise the complex
Secretory functions
Pro-inflammatory (Produce IL-1, TNF)
Pro-coagulatory factors
Immune regulatory products
Enzymes
Wound healing / tissue repair
Regulation of monocyte and granulocyte pools (via growth factors)
What types of cells determine whether tissue can regenerate during repair?
Labile cells
Stable cells
Permanent cells
Give examples of labile cells and their characteristics.
Examples of labile cells:
Epidermis, GI tract epithelial cells, Haematopoietic cells in bone marrow
Characteristics (= Continuously dividing cells)
High capacity for regeneration
Have a large population of stem cells
Give examples of stable cells and their characteristics.
Examples:
Hepatocytes in the liver
Characteristics (= Quiescent cells)
Very low level of replication
Can proliferate when stimulated by injury
BUT not as good as labile cells
Give examples of permanent cells and their characteristics.
Examples:
Neurons in the brain
Cardiac muscle cells
Skeletal muscle cells
Characteristics (= Non-dividing cells):
Cannot undergo mitosis
Describe the process of tissue repair after injury.
Persistent stimulus / injury → Activates macrophages and lymphocytes
Release of growth factors such as:
TGF-β (Transforming Growth Factor-β)
EGF (Epidermal Growth Factor)
FGF (Fibroblast Growth Factor)
VEGF (Vascular Endothelial Growth Factor)
Growth factor stimulates epithelial cells, endothelial cells, parenchymal cells and fibroblasts proliferation
Extracellular matrix deposition
VEGF increases vascular permeability → Fibrinogen leaks into the injured tissue → Fibrinogen is converted to fibrin → Fibrin forms a temporary extracellular matrix (ECM) scaffold that allows fibroblasts to migrate into the injury site
Granulation tissue formation
Fibroblasts produce collagen
New blood vessels form (angiogenesis).
This creates immature granulation tissue
Tissue remodelling and maturation
As inflammation subsides:Inflammatory cells decrease
Collagen fibers re-organise (align parallel to the tissue surface)
Blood vessels become more organised (perpendicular to the greatest plane of tension)
Epithelialization completes
± Scaring
Tissue may return to normal structure or
Scar formation (= fibrosis) may occur
How does the extracellular matrix (ECM) contribute to tissue remodeling?
The ECM provides structural support for cells and helps regulate cellular functions, contributing to the strength and stability during and after tissue healing.
What mediates the production of collagen by fibroblasts during tissue repair?
Cytokines, IL-1, IL-4, IL-13 and TNF produced by activated macrophages and lymphocytes
P.S. From immunology lecture, with IL-4 and IL-13 cytokines, macrophages change to phenotype II, which produce RELM-alpha → Help with deposition of ECM and wound healing
What mediates the process of tissue remodeling?
MMPs (Matrix Metalloproteinases) and their inhibitors TIMPs (Tissue Inhibitors of Metalloproteinases) produced by fibroblasts, neutrophils and macrophages etc.
P.S. MMPs break down the ECM; TIMPs prevent excessive break down of ECM
What is the difference between healing by first intention and healing by second intention?
Healing by first intention:
Occurs in wounds with opposed edges (e.g., surgical wounds)
Minimal disruption of basement membrane
Only small inflammatory response in dermis e.g. presence of neutrophil and little formation of new blood vessels
Healing by second intention:
Occurs in wounds with large tissue defects where edges cannot be closed
More intense inflammatory reaction (e.g., ulcers, abscesses, infarction)
Formation of larger amount of granulation tissue to fill the tissue defect → Epithelium may not regenerate to how it was initially → Cause puckering 起皺 of overlying epidermis → Larger scar formation
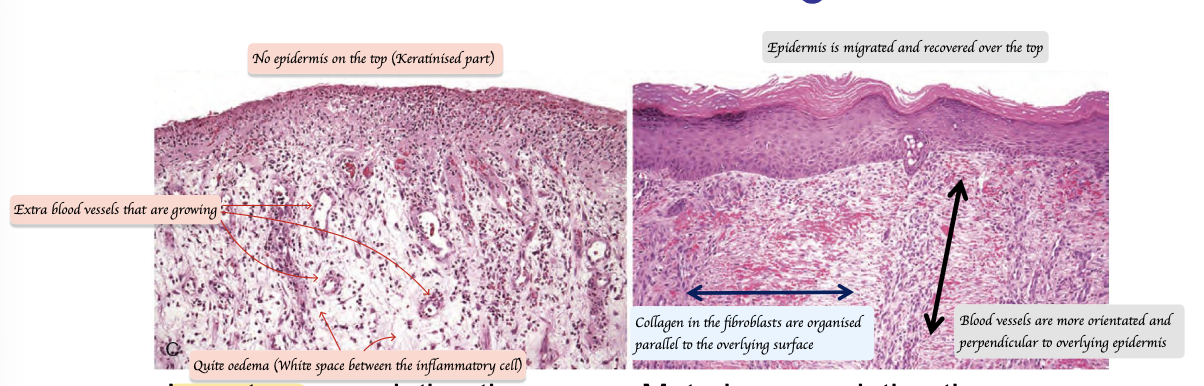
What is the difference between immature granulation tissue and maturing granulation tissue?
Immature granulation tissue:
Many inflammatory cells
No epidermis (Keratinised) on the top layer
Little collagen
Oedema (White space between the inflammatory cells)
Newly blood vessels
Maturing granulation tissue:
Fewer inflammatory cells
Epithelialisation completed
More organised collagen (Parallel to overlying surface)
More organised blood vessels (Perpendicular to the tissue surface)
What systemic factors influence wound healing?
Nutrition
Poor nutrition slows wound healing
Hormones
Insulin
In diabetes mellitus, insulin deficiency or resistance leads to delayed wound healing
Steroids
High levels of endogenous steroids (e.g., adrenal gland disorders) or steroid medication suppress inflammation and delay healing
What local factors influence wound healing?
Bacterial infection
Mechanical factors
Movement disrupts healing
Example: Immobilising a fractured limb helps healing
Foreign material
Size and type of tissue injury
Location of the wound
Poor perfusion
What are abnormalities in wound healing?
Deficient granulation tissue and scar formation
Wound dehiscence = Wound breakdown
Ulceration
What are abscesses?
Inflammation buried in a confined space, tissue or organ, caused by necrosis
What are the layers of an abscess?
Inner layer:
Purulent exudate (pus) with liquefaction
Middle layer:
Immature, cell-rich granulation tissue
Red in colour (Contains lots of blood vessels)
Outer layer:
Mature, cell-poor, fiber-rich granulation tissue
Whiter (As the inflammation subsides, collagen dominants and blood vessel decreases)
What are the possible causes of abscesses?
Bacteria with low hyaluronidase
Spread slowly but induce a strong local inflammatory reaction (stasis, thrombosis).
Tissue necrosis caused by:
Bacterial toxins → damage blood vessels
Reduced perfusion → hypoxia → necrosis
Neutrophil emigration
Neutrophils phagocytose bacteria → release enzymes → tissue necrosis
What are ulcers?
Full thickness defect in epithelium, surface of a tissue or organ due to sloughing of inflammatory necrotic tissue
What is granulomatous inflammation?
Occurs when the injurious agent cannot phagocytosed and degraded easily
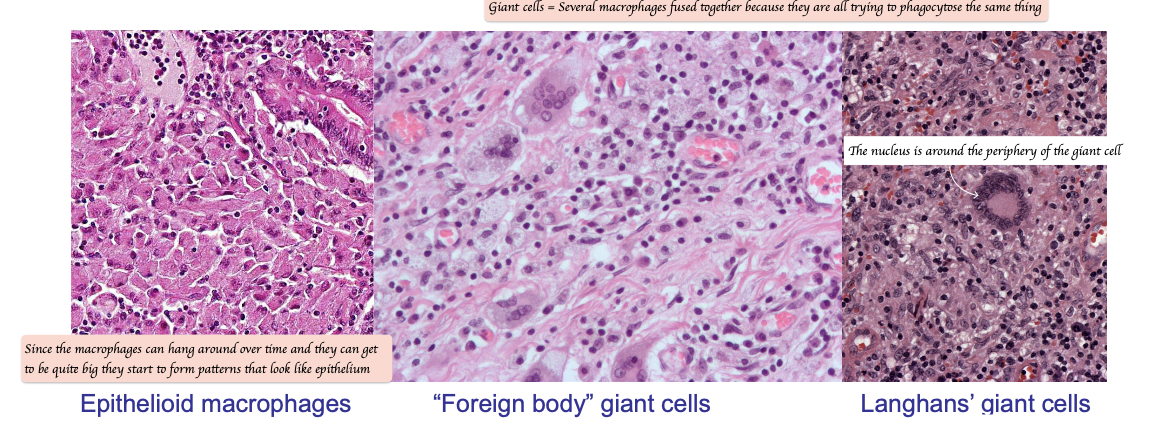
What type of cells dominate in granulomatous inflammation?
Macrophages
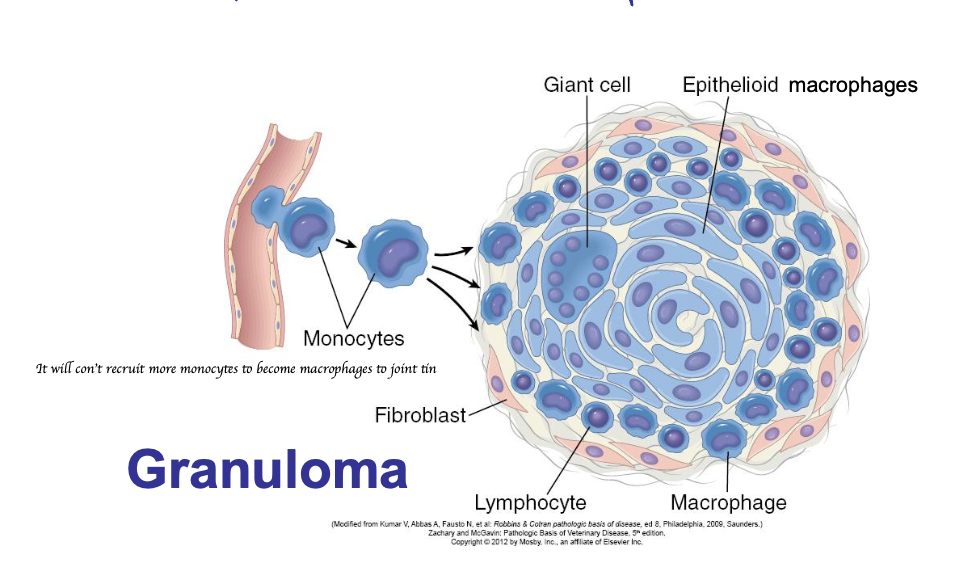
What are giant cells?
Giant cells = Multi-nucleated cells formed by epithelioid macrophages fusing together
Foreign body giant cells
Langhans’ giant cells
What is granuloma?
Focal area of granulomatous inflammation
What is the typical structure of a granuloma?
Rim of fibroblasts and connective tissue
Periphery: Lymphocytes and plasma cells
Centre: Macrophages and epithelioid cells