Materials Midterm 2
1/75
Earn XP
Description and Tags
diffusion, diffusion and ion conductors, phase diagrams I II, nano materials, ceramics, composites, electrical properties
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
diffusion net motion direction and speed
from high to low concentration, happens quickly in liquids and gases
interdiffusion
in an alloy, atoms tend to migrate from regions of high concentration to regions of low concentration
diffusion is driven by ____ but atoms must move via _____.
concentration gradients, specific mechanisms
two types of diffusion mechanisms in solids
vacancy diffusion and interstitial diffusion
vacancy diffusion rate depends on
number of vacancies, activation energy to exchange
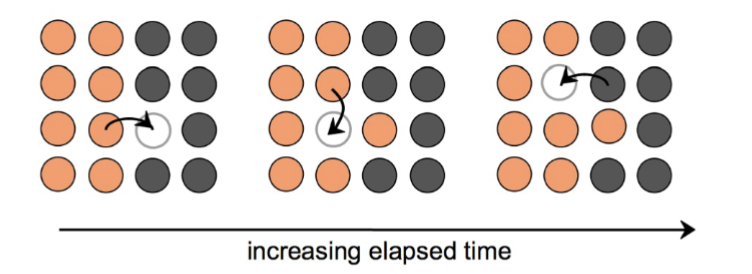
interstitial diffusion
smaller atoms can diffuse between atoms
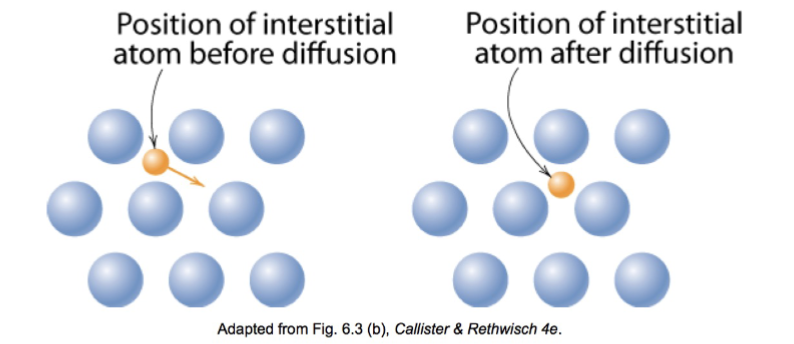
vacancy and interstitial diffusion comparison chart

case hardening
diffuse carbon atoms into host iron at the surface, iron (steel) becomes harder
semiconductor processing
deposit phosphorous on silicon surface, heat it, doped semiconductor regions
rate of diffusion: diffusion coefficient ____ with increasing T
increases
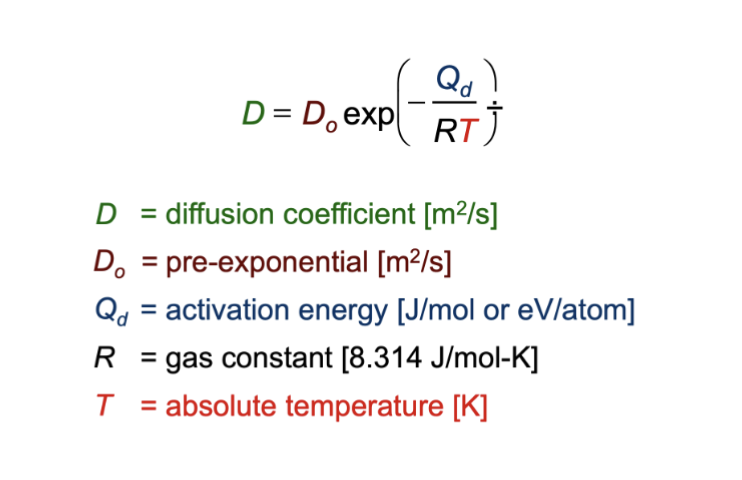
diffusion coefficient equation
D = Do exp ( -Qd / R*T )
activation energy
___ (Q) is required to squeeze atoms past one another during diffusion. generally, more energy is required for a substitutional atom than for an interstitial atoms.
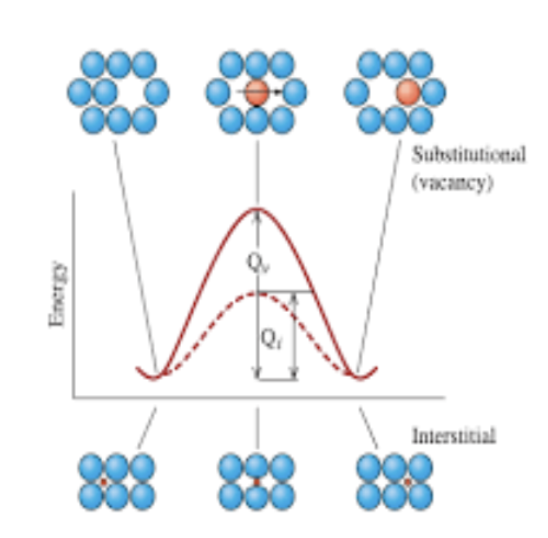
example of different diffusion coefficients in silicon chart
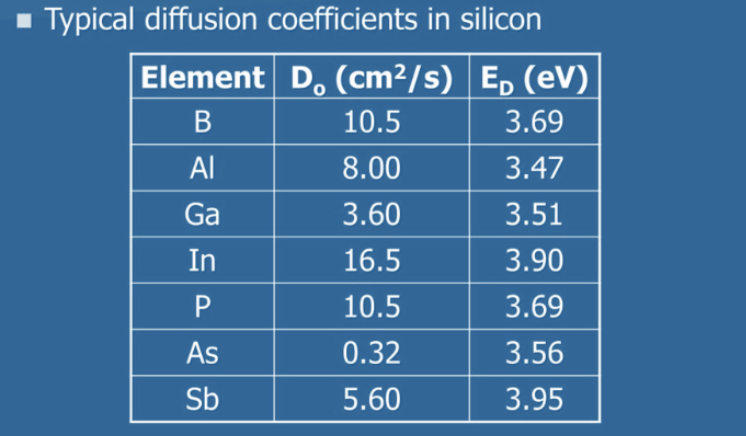
same material, different atoms, orders of magnitude difference
diffusion coeffcient: example
@ 300 deg C the diffusion coefficient and activiation energy for Cu in Si are:
D(300 deg C) = 7.8E-11 m²/s
Qd = 41.5 kJ/mol
What is D @ 250 deg C?
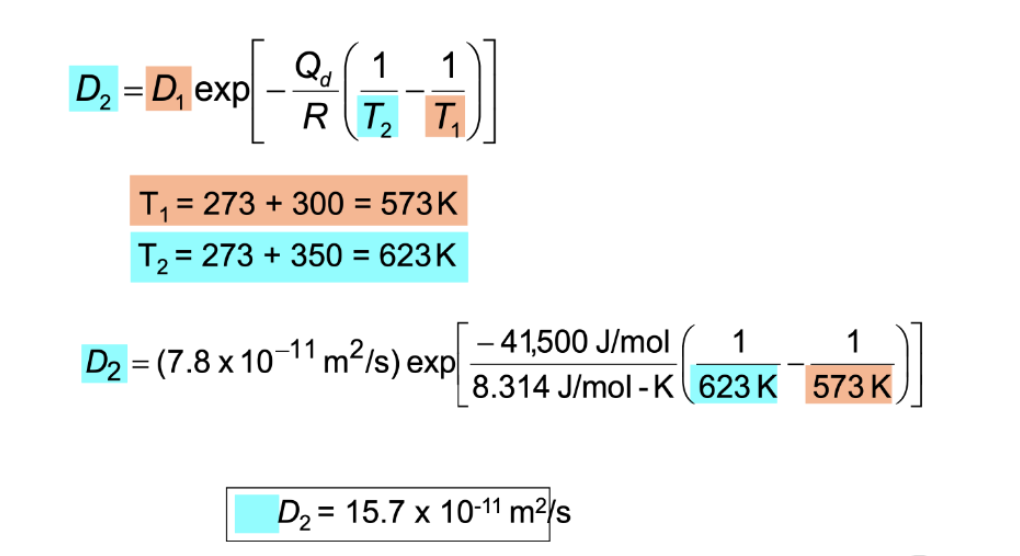
D = 15.7E-11 m²/2
rate of diffusion (fick’s first law)
J = -D (dc/dx)
J is flux, D is diffusion coefficient, dc/dx is concentration gradient
flux during diffusion
the number of atoms passing through a plane of unit area per unit time
fick’s second law
dC/dt = D * d²C/dx² (partials not normal d)
diffusion factors chart (rate)

____ controls materials processing and performance
diffusion (heat treatment, doping, batteries, alloys)
ion conductor
material that allows ions (charged atoms or molecules) to move through it, enabling the transport of electrical charge without the flow of electrons
how does ion conduction work?
ions move through medium, move occurs due to electric fields, concentration gradients or temperature, charge is carried by ions not free electrons
ion conduction is driven by ___ , produces ____
concentration gradient and electric field, electrical current
ionic versus electronic conductivity comparison

__ crystals do not conduct ions
perfect, defects are essential (schottky defects (paired vacancies, frenkel defects (vacancy + interstitial))
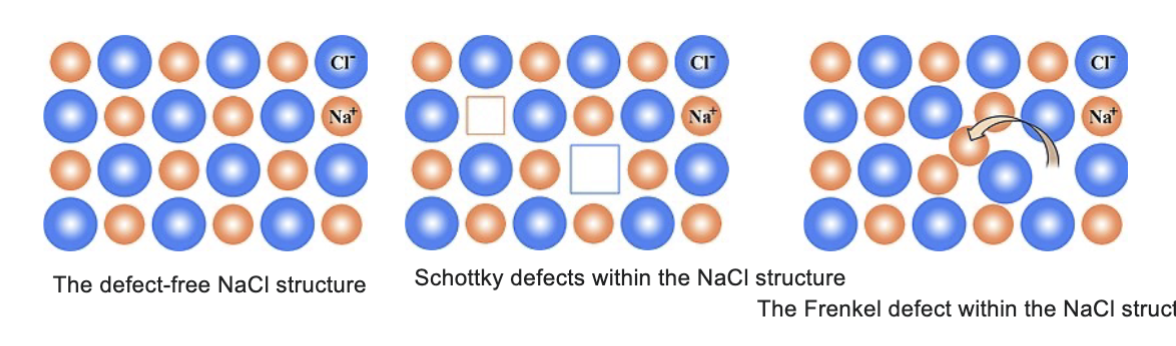
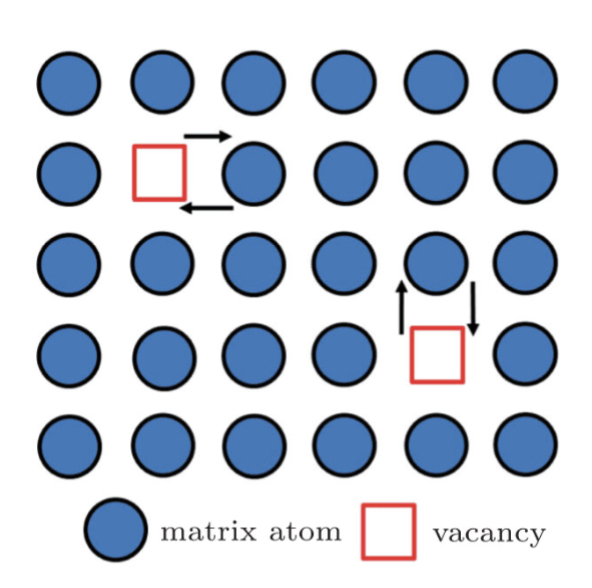
vacancy migraton
ion hopes into neighboring vacancy (requires lattice distortion, energy barrier determines mobility)
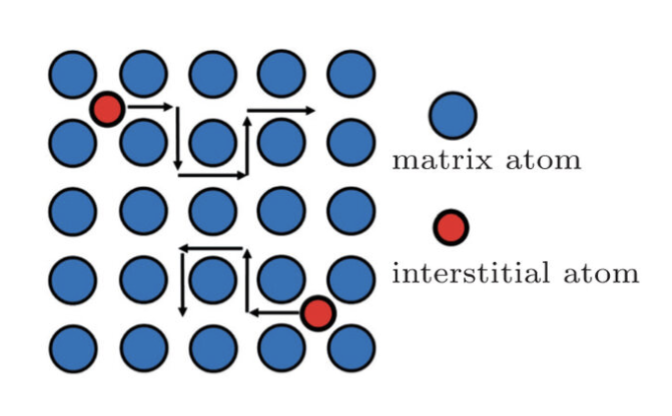
interstitial mechanims
direct interstitial hopping (often faster for small ions)
ionic conductivity equation
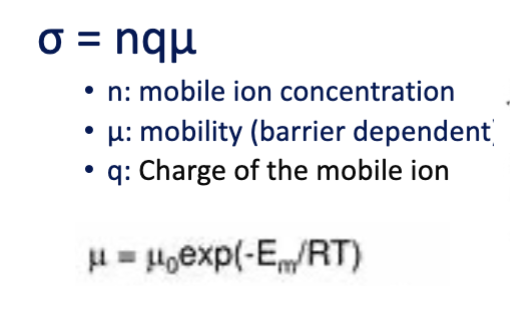
sigma = n x mu x q
structural features of good conductors
open frameworks, connected pathways, low activation barriers
types of ion conductors
liquid electrolytes, polymer electrolytes, solid-state eelctrolytes
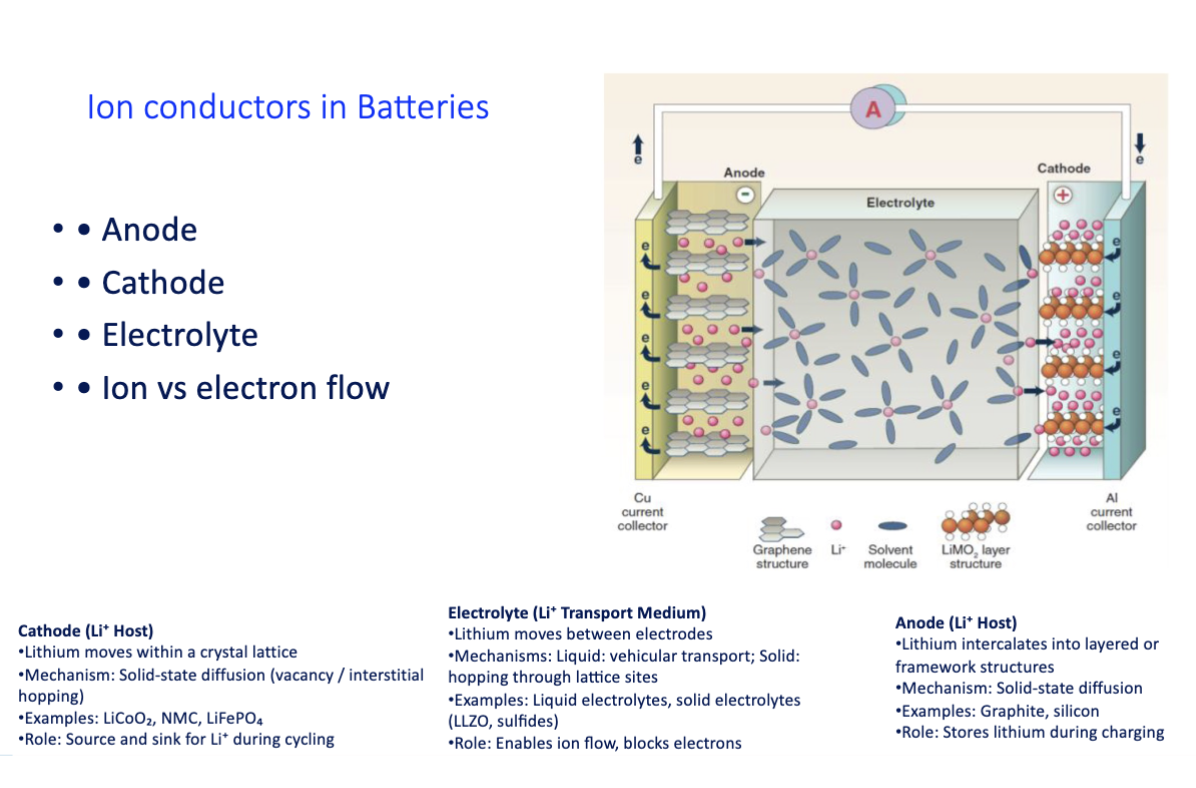
ion conductors in batteries
phase
homogeneous in chemical composition, physics properties, uniform crystal structure, separated by boundaries
phase diagram
plots the equilibrium phase as a function of composition, temperature and pressure
equilibrium phase depends on:
temperature pressure and composition
gibbs free energy
the stable phase is the one with the lowest gibbs free energy
equilibrium and free energy graphically
equilibrium (stable) state exists at the lowest point on free energy diagram, unstable state is on a slope and transforms rapidly into metastable/stable phases, metastable state is local minimum and transforms slowly
phase boundary
set of conditions where two phases have equal free energy
unary phase diagrams axes
typically temperature and pressure
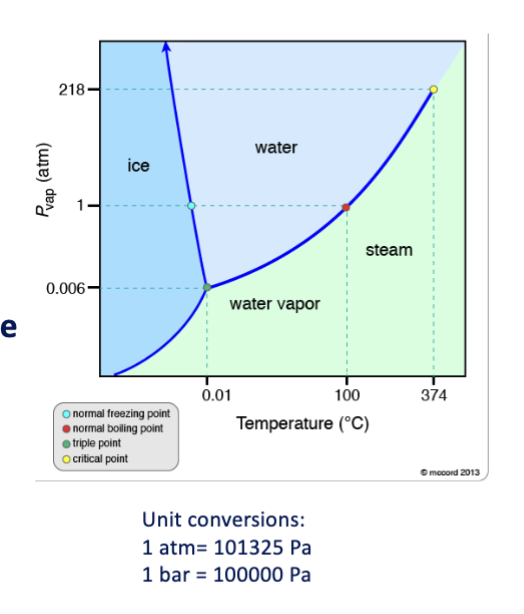
triple point
three phases, equal G
critical point
disappearance of liquid gas phase distinction
solubility limit
maximum amount of solute that can added before forming a new phase
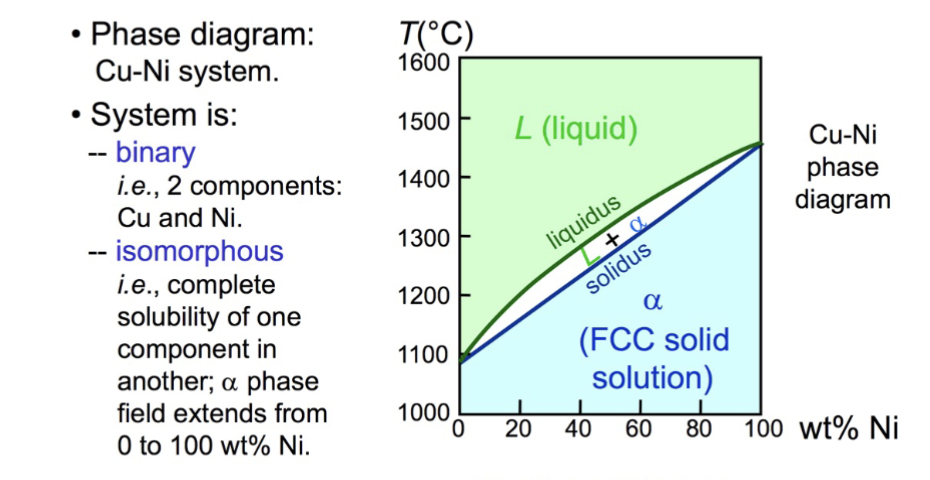
binary isomorphous phase diagram
exhibit complete liquid and solid solubility, contain two single-phase regions
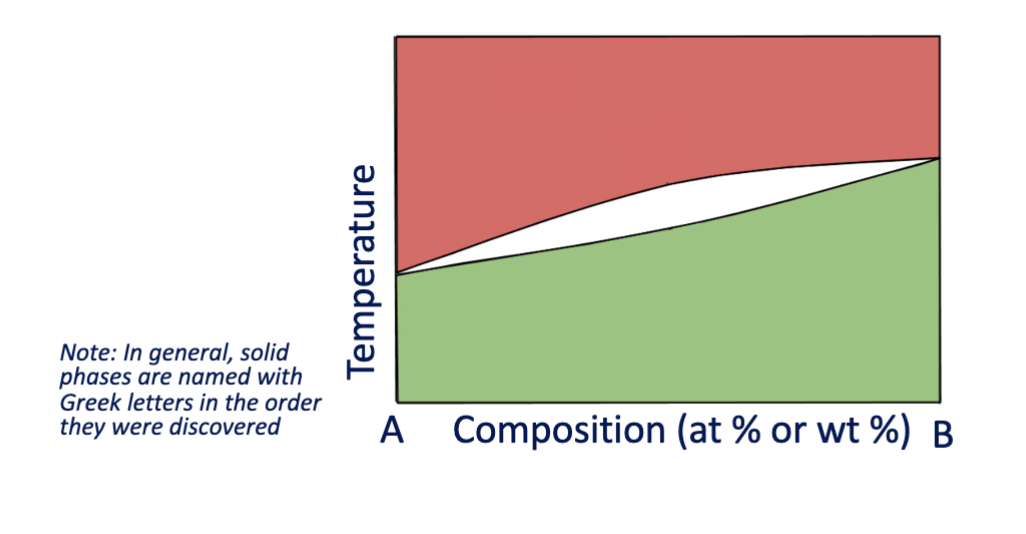
binary eutectic phase diagrams
eutectic: a system in which a single composition melts and solidifies at a lower temperature than the individual components of any other mixture
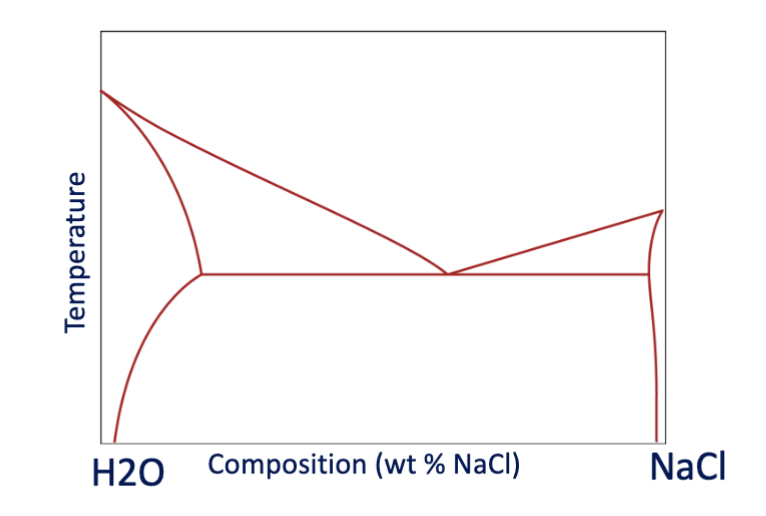
why is salt more effective than sugar for keeping streets from icing over?
the minimum freezing temperature for sugar and ice is around 7 degrees C warmer than ice and salt. you need a large % of sugar before there is a noticeable decrease in freezing temperature, salt begins decreasing the freezing temperature linearly.
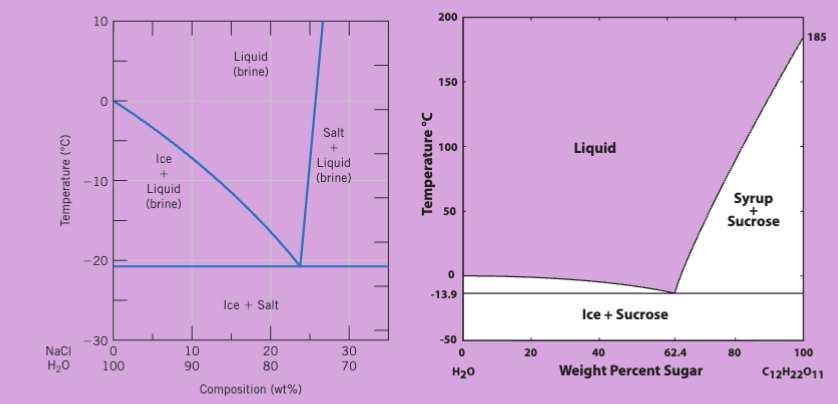
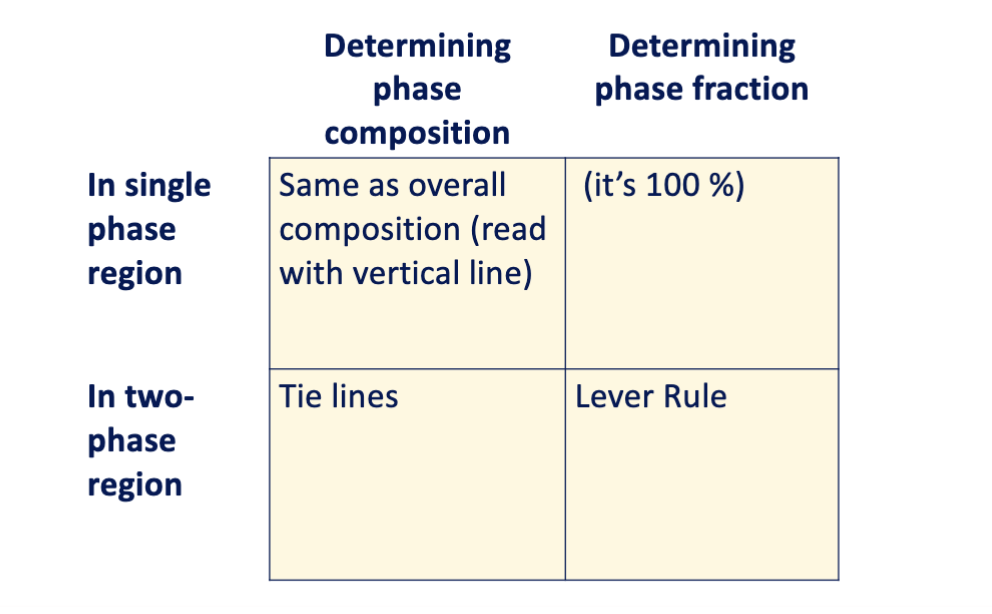
determine phase composition and fraction table
why do phase transformations matter?
properties depend on microstructure, microstructure depends on how phases form, phase diagrams only tell eq phases
__ predict eq phases
phase diagrams
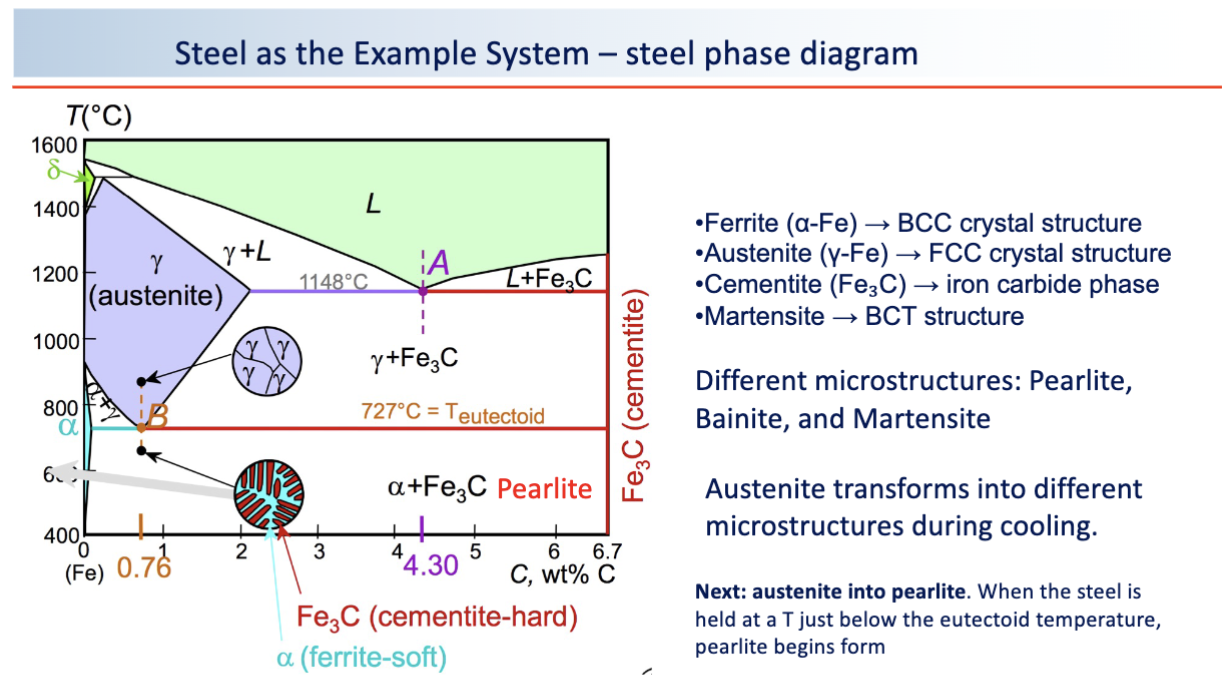
steel phase diagram
TTT diagram
time temperature transformation digram, describes how fast a phase transformation occurs at a constant temerpature
TTT diagrams predict and help:
predict when transformations start and finish, help determine resulting microstructures
austenite transformation in steel summary
high T yields pearlite, intermediate T yields bainite, rapid quenching yields martensite
microstructure control properties: pearlite, bainite, martensite
p: moderate strength, good ductility
b: strong and tough
m: very hard but brittle
nanomaterials and light
strong and unusual interactions with light
what happens when materials are smaller than light?
optical properties are determined by the band structure of the material, in nano materials shape size determine optical properties
why nanomaterials are special?
large surface to volume ratio, many surface atoms, quantum confinement of electrons, interacts with light
nano and light
quantum dots: color depends on particle size, photonic crystals: nanostructure controls light
ceramics can be () or (); atomic structure contians strong covalent or ionic bonds
amorphous, crystalline
key ceramic properties
brittle, strong at high T, insulators
why are ceramics brittle?
atoms cannot slide easily, charges repel, bonds are directional
the more electronegativity difference the more ionic the bond, this produces what property of ceramics
strength
ceramic ocide crystal structure
oxygen anions much larger than metal cations, close packed oxygen in lattic (FCC) cations in the holes of oxygen lattice
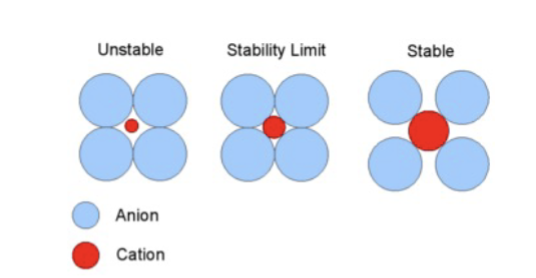
visualizing coordination
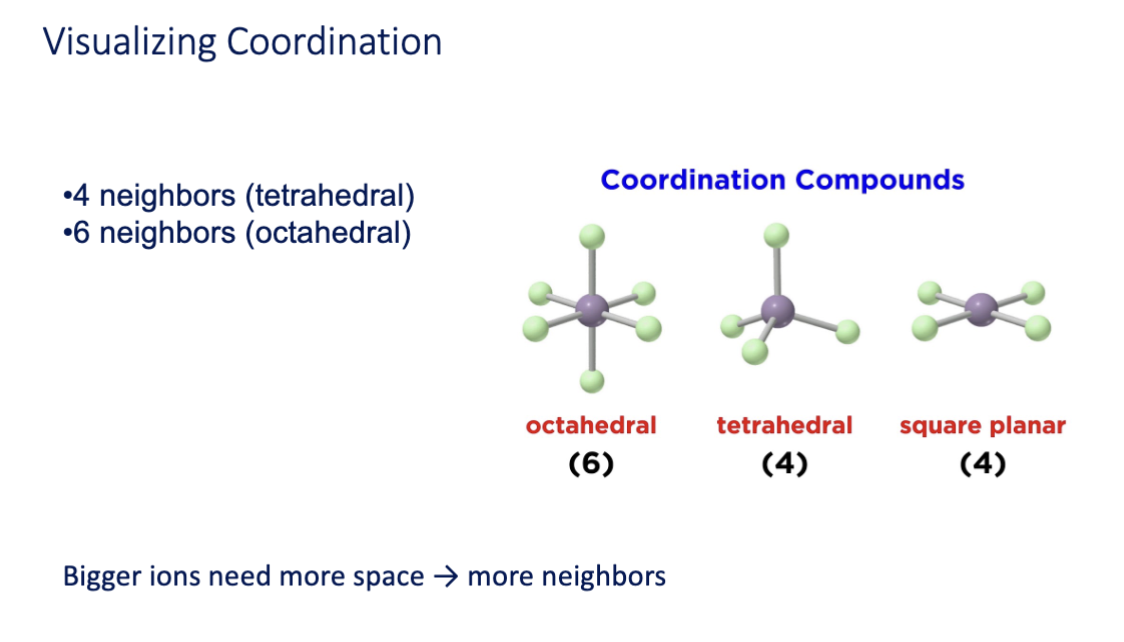
coordination number (CN)
the number of nearest enighbor ions surrounding a central ion
radius ratio formula
radius of cation/ radius of anion
radius ratio predicts
the crystal structure based on the size of the hole the cation can stably occupy (ion size determines structure)
crystalline versus amorphous properties
amorphous tend to be transparent and let light pass though, crystalline show greater strength, stability at high T, and are opaque from scattering light
polymers (many repeating units)
type of macromolecule, have large molecular weights
a long chain made of repeating small units
polymer (monomer)
the __ of polymer chains determines mechanical behavior
structure
chain flexibility in polymers
polymers chains are not straight, can bend twist and rotate
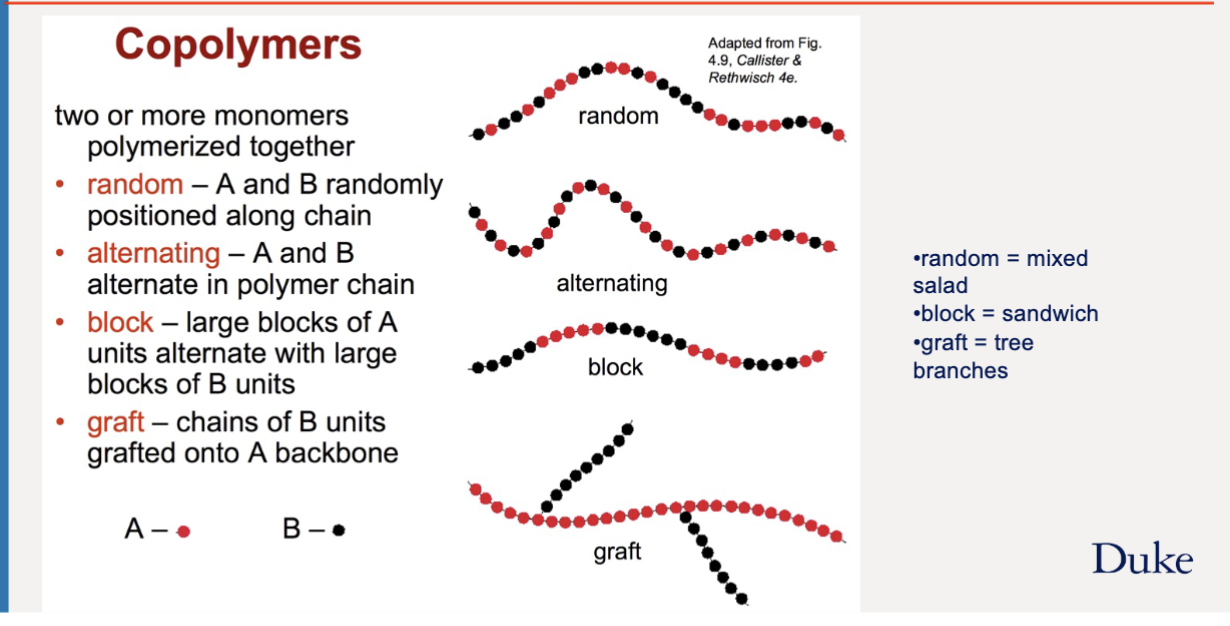
copolymer shapes
two or more monomers polymerized together
molecular weight
mass of a mole of chains (longer chain has higher M)
polymers have chains of different length so use:
average molecular weight
resistance (R) of a wire depends on:
resistivity of the material and geometry of the wire
resistivity (rho)
property of the material itself rho = R * A/L
electrical conductivity is the inverse of
resistivity (sigma = 1/ rho)
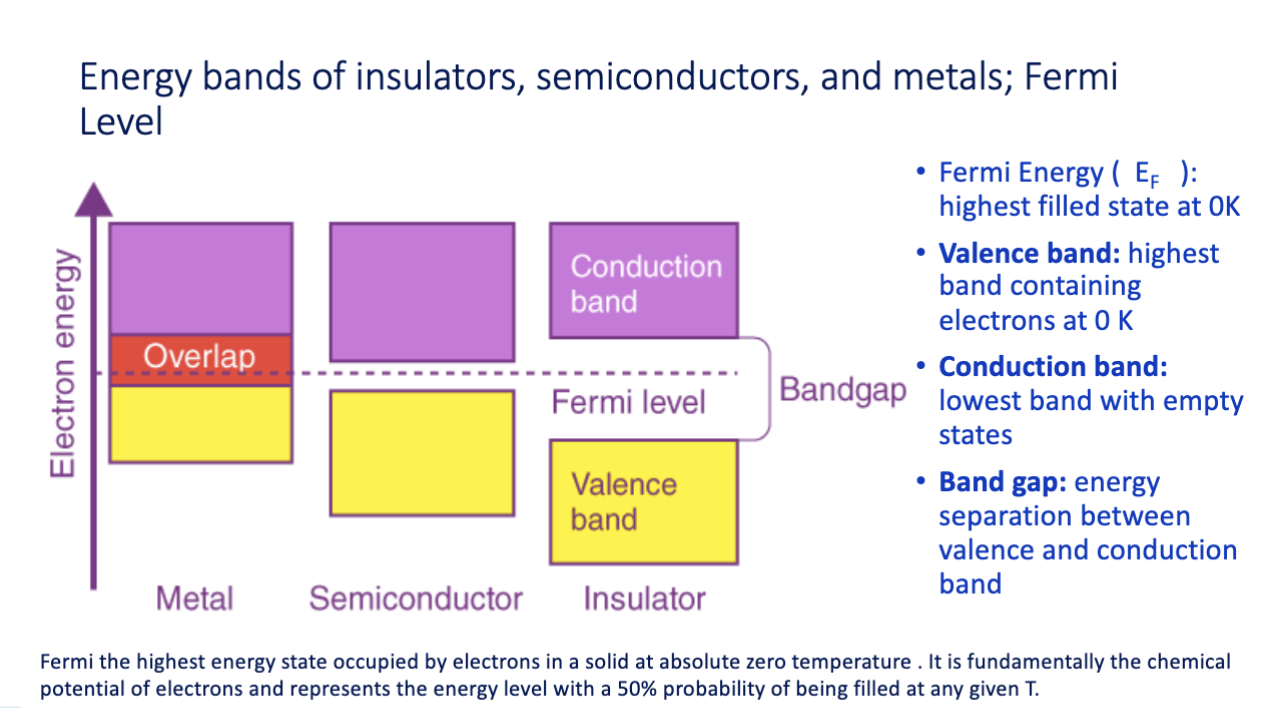
band gaps graphic