CHEMISTRY UNIT 3 chemical energies
1/20
Earn XP
Description and Tags
Chemical energies
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
What is enthalpy ?
Enthalpy is a measure of the chemical energy in a substance
What is an exothermic reaction ?
An exothermic reaction is a reaction or process that releases heat energy
Why in industry might exothermic reactions require heat to be removed ?
In industry, exothermic reactions may require heat to be removed to prevent the temperature rising
What are endothermic reactions ?
Endothermic reactions are reactions or processes that take in heat energy
Why in industry might endothermic reactions incur costs ?
In industry, endothermic reactions may incur costs in supplying heat energy ion order to maintain the reaction rate
What happens to energy when bonds are being broken ?
Energy is used when breaking bonds
What happens to energy when new bonds are formed ?
Energy is released when new bonds are formed
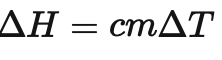
What is this formula used for ?
The formula /_\ H = cm/_\T is used to calculate the enthalpy change for a reaction
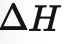
What is this ?
/_\ H is the enthalpy change (in kJ or kJmol-1)
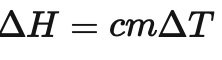
What is C in this formula ?
C is the specific heat capacity of water (4.18)
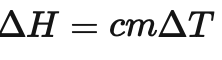
What is m in this formula ?
m is the mass of water ( in kg) (100cm3 = 0.1kg)
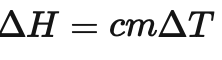
What is the /_\ T in this formula ?
/_\ T is the change in temperature ( *C )
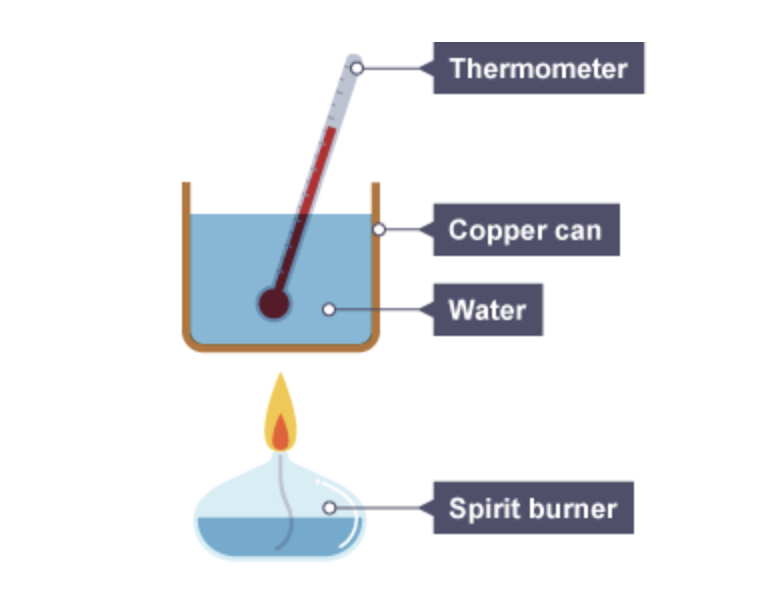
What several measurements are taken when calculating the enthalpy of combustion ?
start temperature of the water
final temperature of the water
mass of burner before experiment
mass of burner after experiment
the temperature change
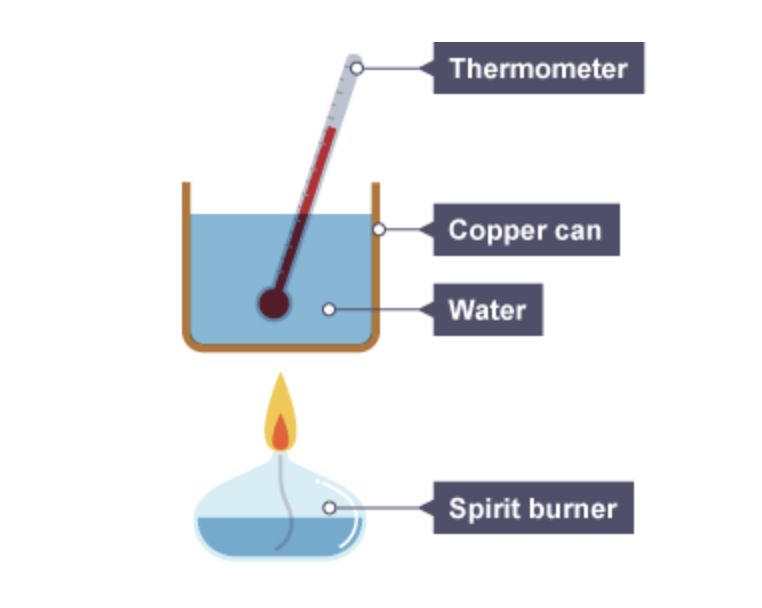
What can be added to this experiment ( calculating enthalpy of combustion ) to improve the accuracy of the results
A draught shield can be added to the set up of this experiment as this will prevent any unwanted draughts affecting the results of the experiment
What is the enthalpy of combustion of a substance ?
The enthalpy of combustion of a substance is the enthalpy change when one mole of the substance burns completely in oxygen
What is Hess’s Law ?
Hess’s law states that the enthalpy change for a chemical reaction is independent of the route taken (the enthalpy change for the overall process will be identical regardless of how many steps are taken)
What is the molar bond enthalpy ?
The molar bond enthalpy is the energy required to break one mole of bonds in a diatomic molecule
What is a mean molar bond enthalpy ?
A mean molar bond enthalpy is the average energy required to break one mole of bonds, for a bond that occurs in a number of compounds
Is bond breaking an exothermic or endothermic process and why ?
Bond breaking is a endothermic process as energy is required to break a covalent bond between two atoms to overcome the attractive force
Is bond making an exothermic or endothermic process and why?
Bond making is a exothermic process as energy is released when new chemical bonds are formed
What are bond enthalpies ?
Bond enthalpies are the energies required to break one mole of a particular bond between a pair of atoms in the gaseous state