OCR A level Chemistry Module 3
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
Periodicity
the repeating pattern of chemical and physical properties of the elements
Groups
Vertical columns on the periodic table
Periods
Horizontal rows on the periodic table
Ionisation
The removal of one or more electrons from an atom.
First ionisation energy
The energy required to remove one mole of electrons from ine mole of gaseous atoms to form one mole of gaseous 1+ ions
Equation for IE1 of Li
Li(g) --> Li+(g) + e-
Factors affecting ionisation energy
-atomic radius
-nuclear charge
-electron shielding
How does atomic radius affect ionisation energy?
The larger the atomic radius, the smaller nuclear attraction, as they are further away from the nucleus.
How does nuclear charge affect ionisation energy?
The higher the nuclear charge, the larger the attractive force is on the outer electrons.
How does electron shielding affect ionisation energy?
Inner shells of electrons repel the outer shell electrons because they are negative, the more shells there are the smaller is the nuclear attraction experienced by the outer electrons
Successive ionisation energies
The measure of the amount of energy required to remove each electron in turn, each successive energy is higher than the one before. As each electron is removed there is less repulsion between remaining electrons and drawn closer to the nucleus.
Trends across a period
Across each period the number of protons increase, electrons are added to the same shell so the shell is drawn inwards slightly, electron shielding remains the same. First ionisation energy increases across a period.
Trends down a group
Moving down a group first ionisation energy decreases because the number of shells increase so the nuclear attraction is weaker and there is more shielding.
Metallic bonding
a bond formed by the attraction between positively charged metal ions and the electrons around them
Giant metallic lattice structure
A regular structure consisting of closely packed positive metal ions in a sea of delocalised electrons.
Properties of giant metallic lattices
-high melting and boiling points
-malleable and ductile as there are no bonds
-good thermal and electrical conductors
-insoluble
group 2
ionisation enrgies incerase going down the group due to increasing atomic radiusand shielding effect.
reactivity increases going down, atom gets larger and electron is further from nucleus,
group 2 + water -> metal hydroxide + h
group 2 oxides are white solids
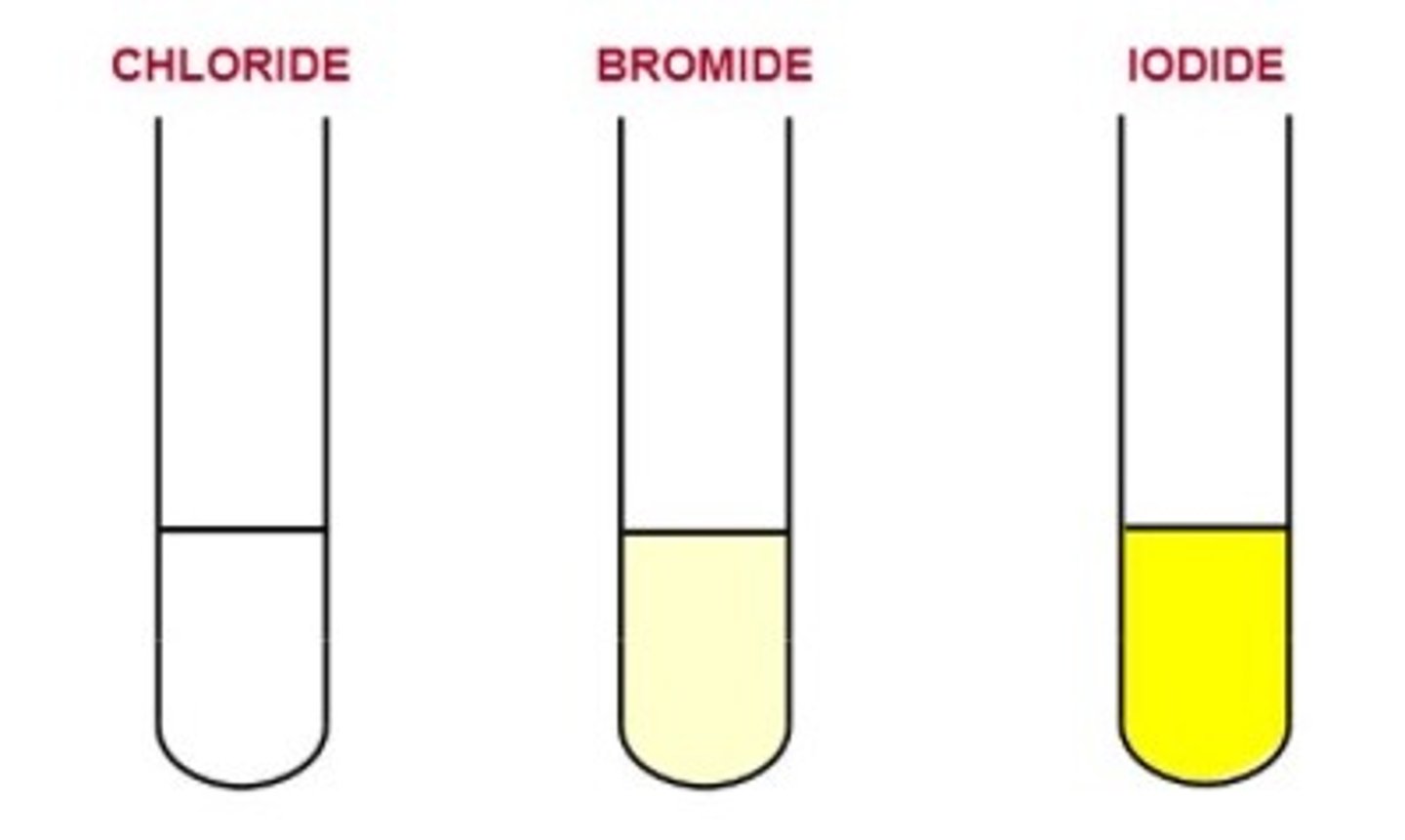
Test for Halides (Cl-, Br-, I-)
Add dilute nitric acid to remove ions that might interfere with the test, followed by silver nitrate solution. a precipitate is formed.
(Cl-) white precipitate
(Br-) cream precipitate
(I-) yellow precipitate
add amionia solution to ensure results. cl dissolves in dilute, br in concentrated, iodine doesnt dissolve
disproportionation reaction
halogens undergo disproportionation reactions with cold dilute alkali solutions, where the halogens are simultaneously oxidised and reduced.
test for ammonium ions (nh4+)
add sodium hydroxide to mystery solution, gently heat, and ammonium gas is given off. hold damp litmus paper above solution, turns red to blue
(NH4+) + (OH−) →NH3(g)+H2O
TEST FOR CARBONATES
add dilute acid, if carbonate is present co2 will be produced, when bubbled through limewater it should go cloudy
test for sulfates
add hcl to remove any carbonates in otherwise it will from baco3 another white precipitate. but not something we are after, add barium chloride. if you get a white precipitate, its barium sulfate, which tells you the substance was a sulfate.
order for testing ions
test for carbonates if none then test for sulfate, if not then test for halide ions - look out for white cream and yellow prercipitate
halogen + water
disproportionation
Cl2 + H2O -> HCl + HclO
GENERAL FORMULA :
X2+H2O⇌HX+HXO
general disproportionation reaction formula
X2 + 2NaOH -> NaXO +NaX +H2O
x is any halogen
halogens
less reactive as you go down
melting and boiling points increase as you go down
Exothermic reaction
A reaction that releases energy in the form of heat
bonds are broken to start the reaction then bonds are made which releases heat
Enthalpy
The energy associated with the making and breaking of bonds.
enthalpy change ΔH
heat energy transferred in a reaction at constant pressure.
Standard conditions
100kPa, 298K, 1 mol dm-3
Endothermic reaction
A reaction in which energy is absorbed, more energy is needed to break bonds than is released when new bonds are formed
why does temperature fall in endothermic reactions
Energy is absorbed from the surroundings
That energy is used to break bonds in the reactants
The energy taken comes from the thermal energy of the surroundings
👉 Because energy is taken from the surroundings:
The particles in the surroundings have less kinetic energy
So the temperature of the surroundings falls
why is change in heat positive for endothermic reactions
although temp. falls, energy enters the system so ΔH changes
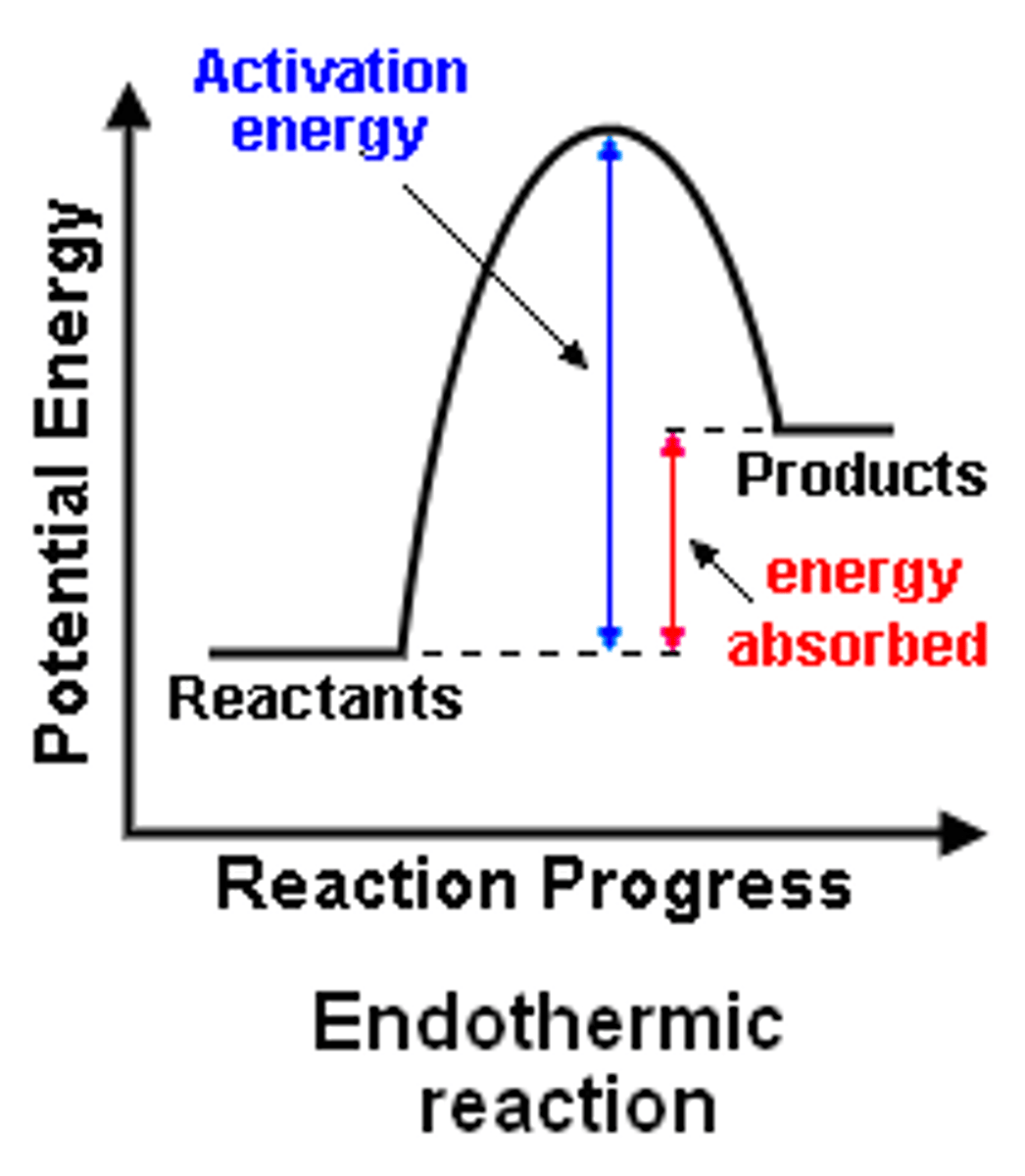
Endothermic enthalpy profile
in endo, products have higher energy that reactantts
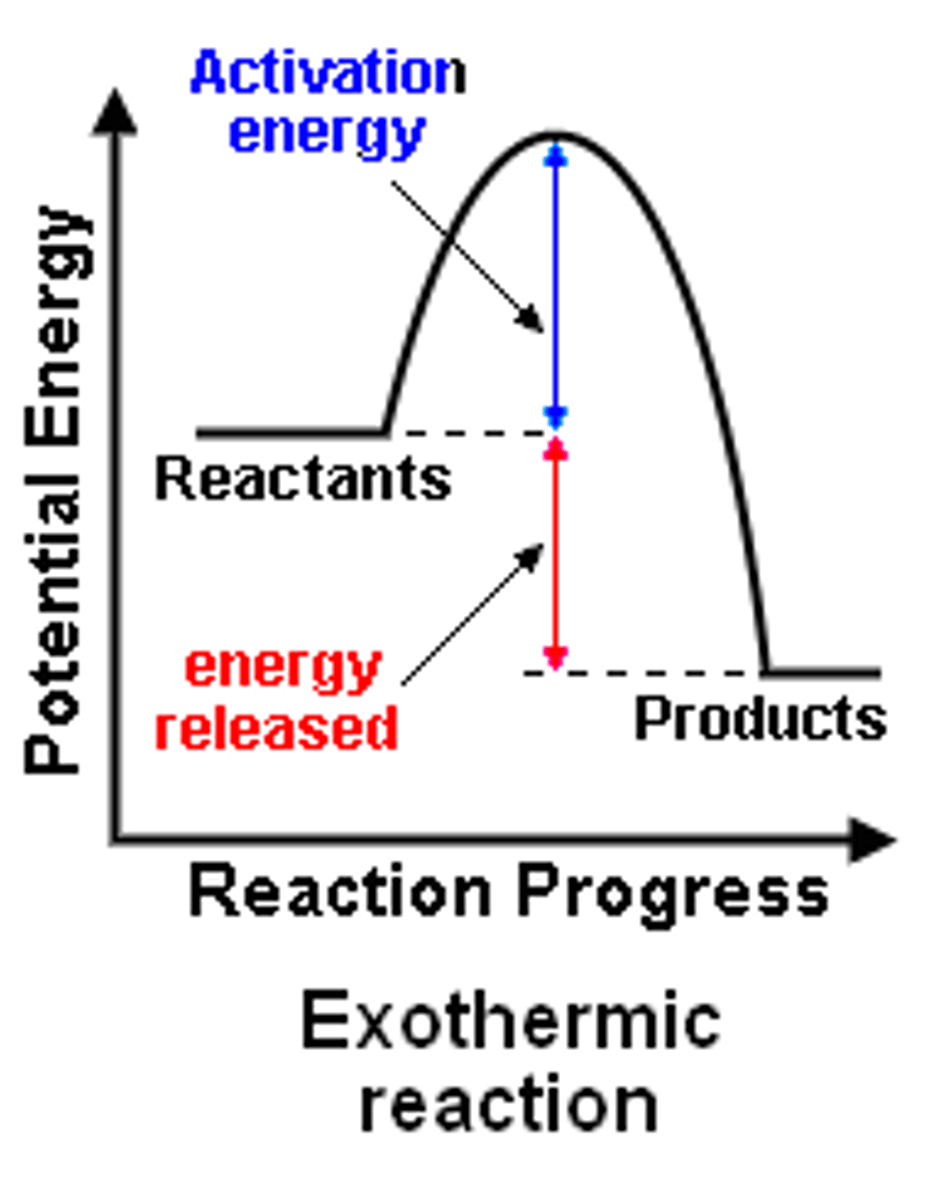
Exothermic enthalpy profile
in exo, products have higher energy than reactants
activation energy
the minimum amount of energy required to start a chemical reaction
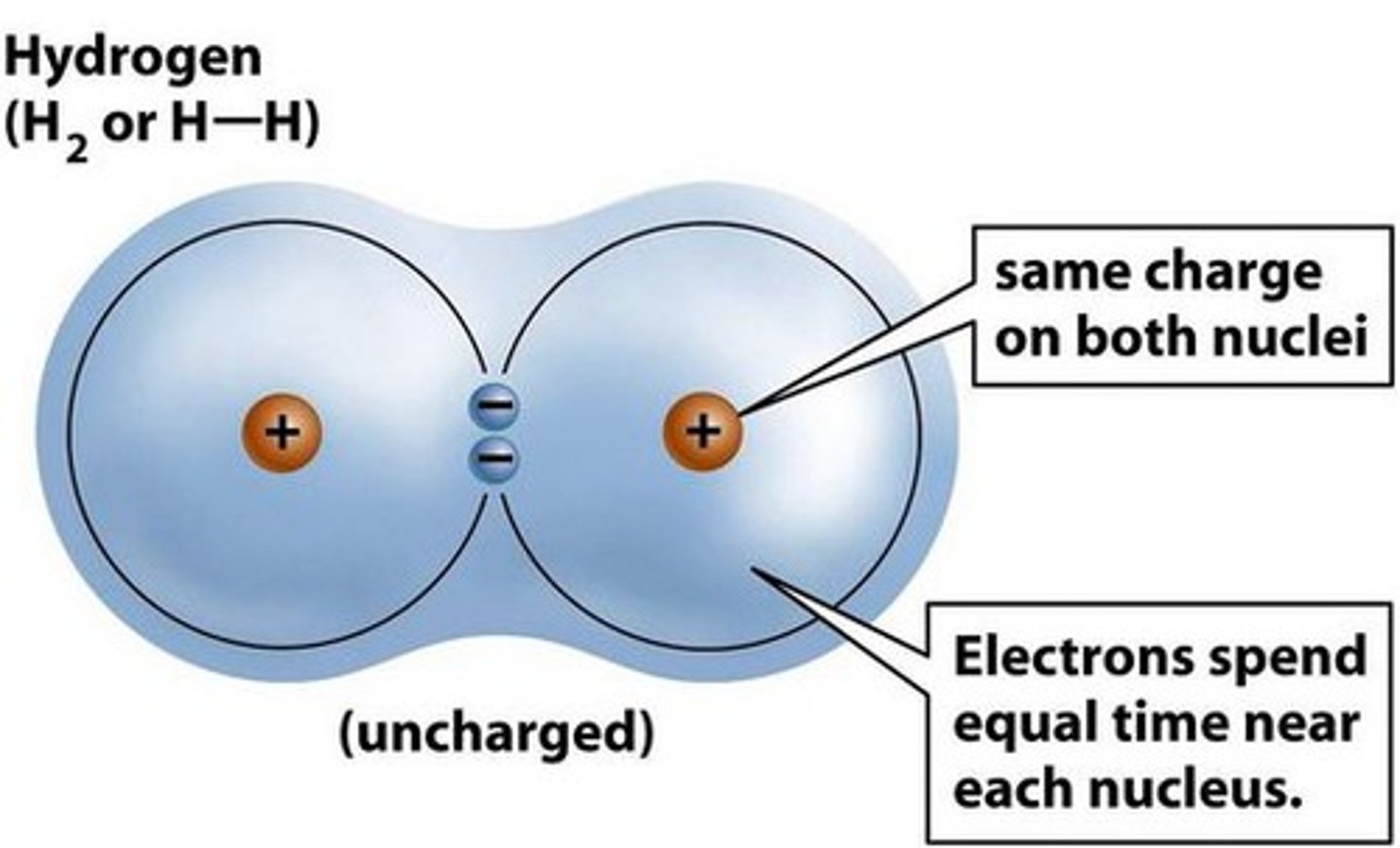
covalent bond
The shared electrons sit between the two nuclei
Each positive nucleus is attracted to these shared negative electrons
This attraction pulls the atoms together
That attraction is the covalent bond
Standard Enthalpy change of reaction
The enthalpy change when, the reaction occurs in the molar quantities shown in the chemical equation, under standard conditions
Standard Enthalpy change of formation
the enthalpy change when, 1 mole of a compound is formed, from its elements in their standard states, under standard conditions
Standard enthalpy change of combustion
the energy change when one mole of the substance in its standard state is burned in excess oxygen, under standard conditions
Standard enthalpy change of neutralisation
The enthalpy change when 1 mole of water forms from a neutralisation reaction.
enthalpy change formula if not given value
total energy to break bonds - total energy released forming bonds
calorimetry
used to work out enthalpy change of combustion of flammable liquid
a fuel is burned to raise the temp of water by a specific amount and we can calculate heat absorbed if we know the water's s.h.c, mass and temp using q=mcΔT
q=
mc∆T
units of q
J, Joules
heat lost or gained.
enthalpy (kJ/mol)= q / moles
to find enthalpy change from q , divide by the number of moles using mass/rm
units of T
K, Kelvin
Average bond enthalpy
The mean average energy required to break 1 mole of a given type of bond, in gaseous molecules
Bond dissociation enthalpy
The enthalpy change required to break one mole of a specified covalent bond in the gas phase by homolytic fission
Hess's Law
The total enthalpy change of a reaction is independent of the route taken ie the total enthalpy change of a reaction is the same, no matter what route or steps the reaction takes.
enthalpy change of formation
total of products - total of reactants
Effect of concentration on rate of reaction
Increasing the concentration of a reactant will increase the rate of reaction as there are more particles per unit volume, therefore more frequent collisions occur
reactions rates
a reaction wont take place between two particles unless they collide in the right direction (facing each other the right way) and they need to collide with a certain minimum amount of kinetic energy (activation energy)
How to calculation rate with concentration
change in concentration/time
units of rate, from concentration v time data
mol dm-3 s-1
rate equation
product made/time
Effect of temperature on rate of reaction
~ Molecules have more kinetic energy so move faster, more frequent collisions occur
~ More molecules have E>Ea of reaction, so more collisions are successful
Effect of surface area on rate of reaction
Increasing SA of a reactant will increase the rate of reaction as there are more particles in a unit volume, therefore more frequent collisions occur
Effect of pressure on rate of reaction
Increasing the pressure of a gas will increase the rate of reaction as there are more particles per unit volume, therefore more frequent collisions occur
Effect of catalyst on rate of reaction
A catalyst increases rate by providing an alternative reaction pathway, with a lower Ea, so more collisions are successful
Collision theory
For a reaction to occur, the particles must collide, with the appropriate orientation, and with sufficient energy - E>Ea.
activation energy
the minimum amount of kinetic energy particles need in order to react
catalyst
speeds up the rate of the reaction by providing an alternative pathway with a lower activation energy. the catalyst is chemically unchanged at the end of the reaction.
Homogeneous catalyst
a catalyst that is in the same phase as all the reactants
how do homogenous catalysts work
reactants combine with catalyst to form intermediate species, which then reacts to form the products and reform the catalyst
Heterogeneous catalyst
A catalyst that is in a different phase from that of the reactant substances., eg everything in haber process (all gases) solid iron catcalyst is used ( its solid not gas so its heteregeneous)
How do heterogeneous catalysts work?
Reactants adsorb to the catalyst's surface at active sites. This weakens bonds within the reactants, holds reactants close together on the surface and in the correct orientation to react. Once the reaction has occurred, products desorb from the active sites.
Economic importance of catalysts
-they lower the energy demands, by doing so they reduce costs
-less carbon dioxide emissions
-less waste by allowing different reaction to be used with better atom economy(eg instead of 5 steps would now take 2 steps)
Boltzmann distribution
The distribution of energies of molecules at a particular temperature, usually shown as a graph.
(a skewed normal distribution, with no limit to energy of particles)
Boltzmann distribution- temperature increase
- kinetic energy of all molecules increases
- maximum lowers and shifts to the right
- faster particles, more frequent collisions
- greater proportion of particles have E>Ea, more successful collisions
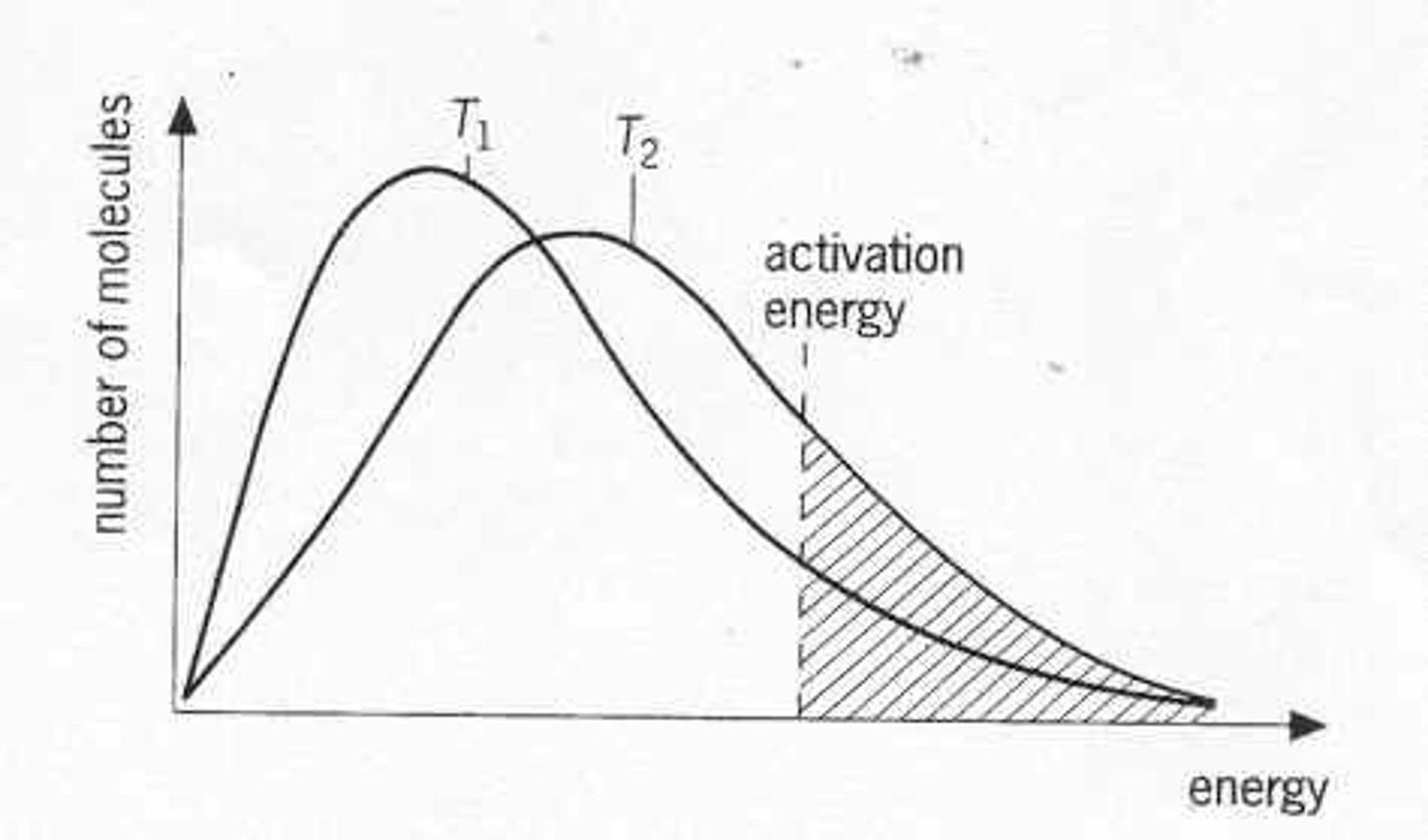
Boltzmann distribution-catalysts
-Ecat < Ea
-more particles have E>Ecat
-more successful collisions
how to measure reaction rate ( gas)
if product is gas, keep reaction on balanceand measure mass loss as gas is lost.could also use bung and syringe, time every minute how much gas is in the syringe
how to measure reaction rate ( precipitate)
place white sheet with cross underneath the precipitate time how long it takes to disappear - highly subjective
Dynamic equilibrium
Occurs when a reversible reaction in a closed system, has equal rates of forward and reverse reactions, and concentrations of species remains constant. can only happen in closed system.
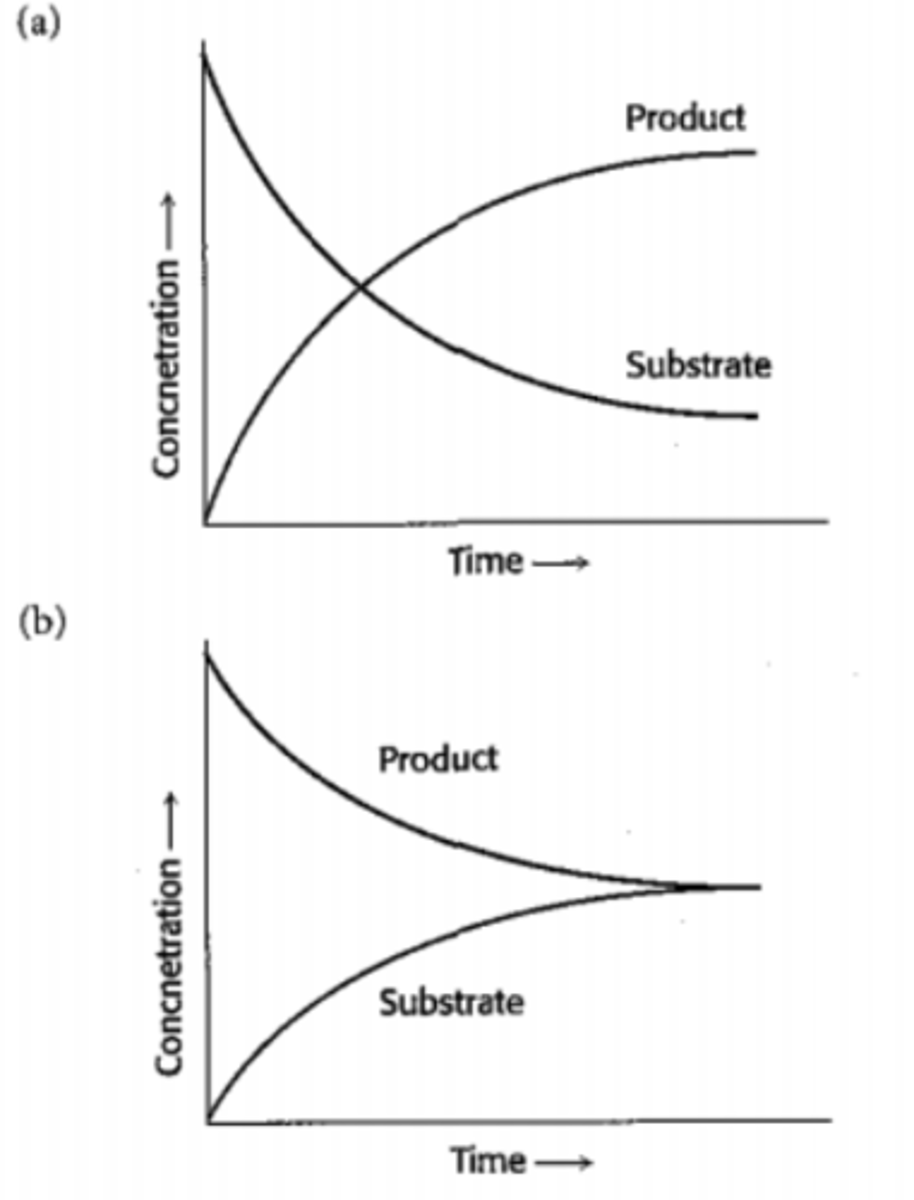
Le Chatelier's Principle
States that if a change is made to an eqilibrium system, the system shifts in the direction to counteract the change.
what happens if equilibrium moves left
if equilibrium moves left, you get more reactants
increasing conc of product/reactant
if you increase conc of reactant, equilibrium tries to get rid of extra reactant so shifts to the right so more product is formed
if you increase conc. of product, equilibrium will shift to the left and try to remove the extra product so the reverse reaction goes faster.
only works in homegenous equilibria
How does a catalyst affect equilibrium?
It doesn't, it increases rate of forward and backward reaction equally
Effect of increasing concentration of a species on equilibrium
-equilibrium shifts in the direction that decreases changed concentration
- towards the side away from the increased concentration
Effect of increasing pressure on equilibrium
- equilibrium shifts in the direction which reduces pressure
- the side with fewer gas molecules
3H2 +N2 -> 2NH3
equilibrium will shift to right because there are 4 on the right and 2 on the left, to reduce pressure. more nh3 produced.
effect of decreasing pressure on equilibrium
The equilibrium position shifts towards the side with the larger number of gasparticles
The effect of increasing temperature on equilibrium
- equilibrium shifts in the direction to reduce the temperature
- in the Endothermic direction
3H2 + N2 -> 2NH3
equilibrium will shift to right
if we increase temp why do we move to the endothermic side
Adding heat = increasing temperature
System wants to absorb the extra heat → shifts toward the endothermic direction (the reaction that takes in heat)
catalysts have no effect on the position of equilibrium. they speed up the forward and revese reaction by same amoun. equilibrium is reached faster.
Equilibrium constant, in terms of concentration
Kc= [product] raised to power of stoichiometries / [reactants] raised to power of stoichiomteries
What does it mean when Kc is greater than 1
-reaction favours the products/RHS
What does it mean when Kc is smaller than 1
-reaction favours the reactants/LHS
When does Kc change?
as Temperature changes
If concentration of a species is changed, does Kc change?
no, Kc only changes if Temperature changes.
kc and temp
kc is only valid for one temp, changing the temp will change equilibrium conc and hence kc too. if temp causes equilibrium to shift right kc increases and vice versa.