AP Biology - Unit 3
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Key properties of enzymes
proteins that catalyze reactions in cells, lower activation energy, increase rate of reactions.
changing this effects enzymes
temperature, pH, ion concentration, interferes with bonds which change active site
denaturation
loss of normal shape of a protein
more kinetic energy increases molecular motion which..
increases chance that enzymes will bind with substrate
reversible denaturation
restoration of optical conditions restores the enzymes function as it regains optimal shape
irreversible denaturation
enzymes shape is permanently changes and its catalytic ability is destroyed
Low substrate concentration
the probability of the enzyme meeting it substrate is low and the product is produced at a very low rate
medium substrate concentration
the reaction rate increased, collision rate increased
high substrate concentration
you get to a saturation point and all enzymes have active sites interacting with substrate, rate peaks
competitive inhibition
foreign molecule blocking enzymes active site, keeps from binding, inhibits rate of reaction

non competitive
foreign molecule binds away from active site at a allosterice site, causes ripple effect which causes a change in the active site so substrate can not bind.
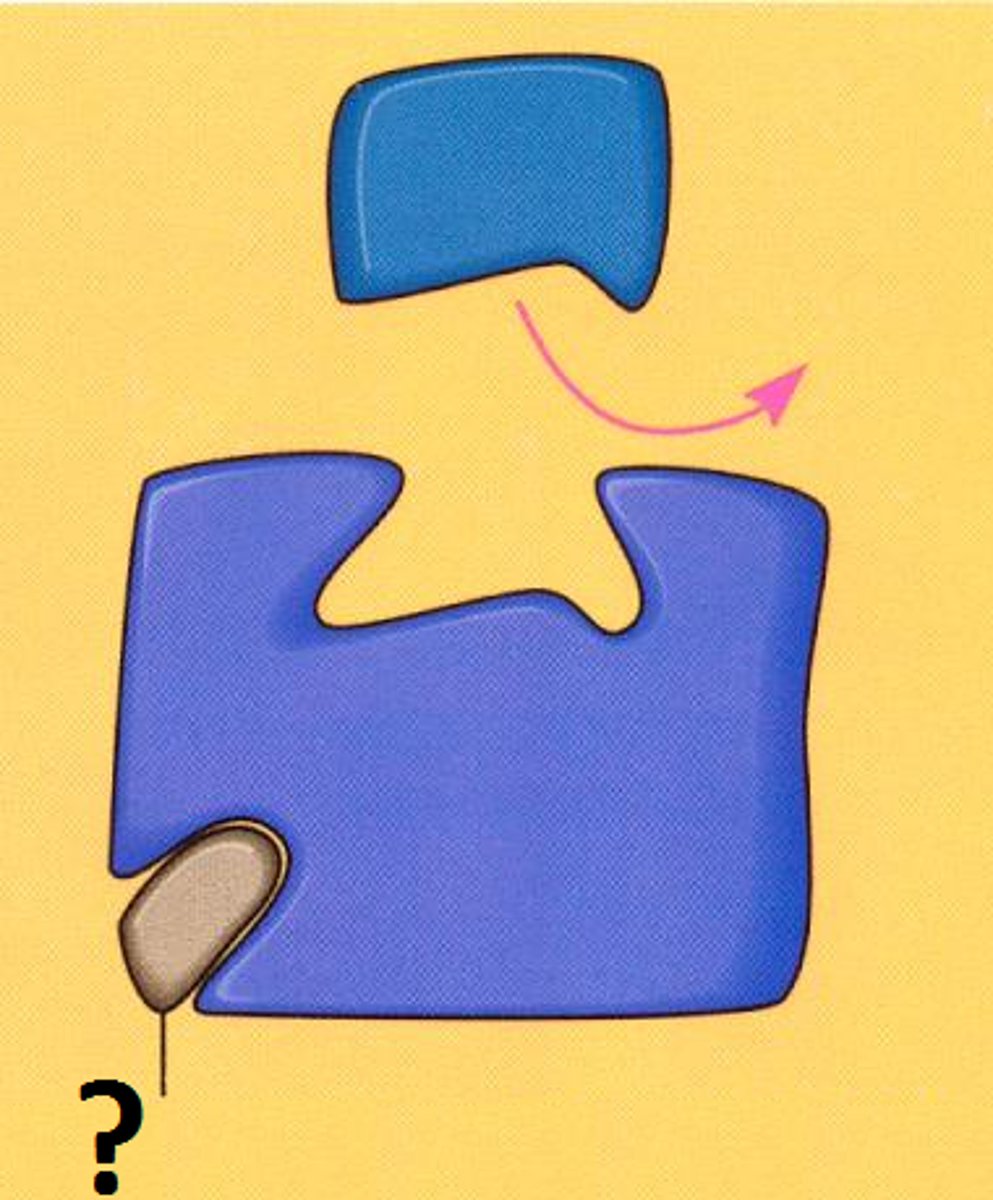
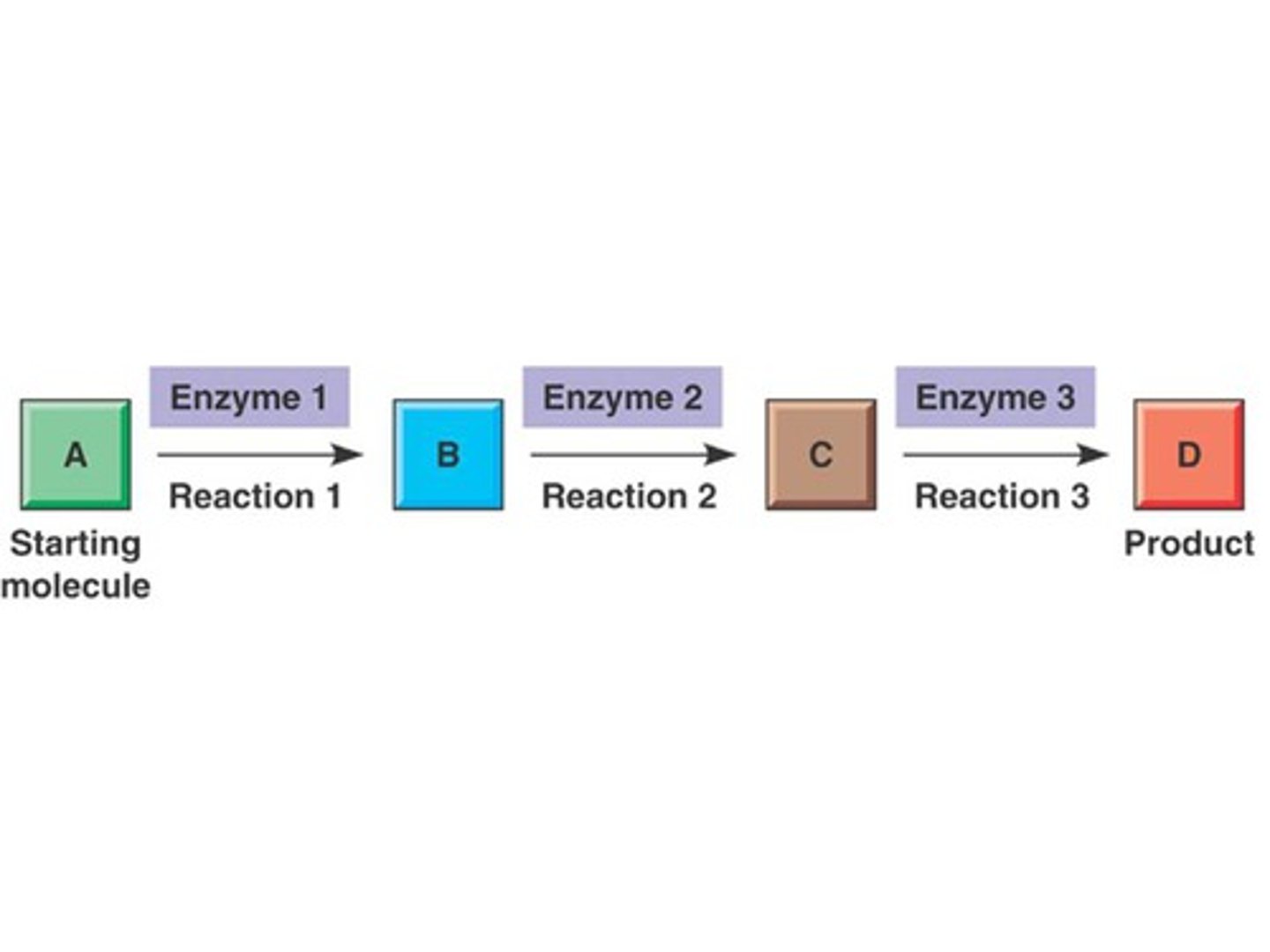
metabolic pathway
linked series of enzyme catalyzed chemical reactions occurring within a cell, can be linear or circle
autotrophs
organisms that can produce their own food, plants
chemoautotrophs
the energy for their life process comes from chemo synthesis.
chemosynthesis
oxidizing inorganic substances like iron, sulfur, or hydrogen sulfide.
heterotrophs
capture the energy present in organic compounds produced by other organisms, animals
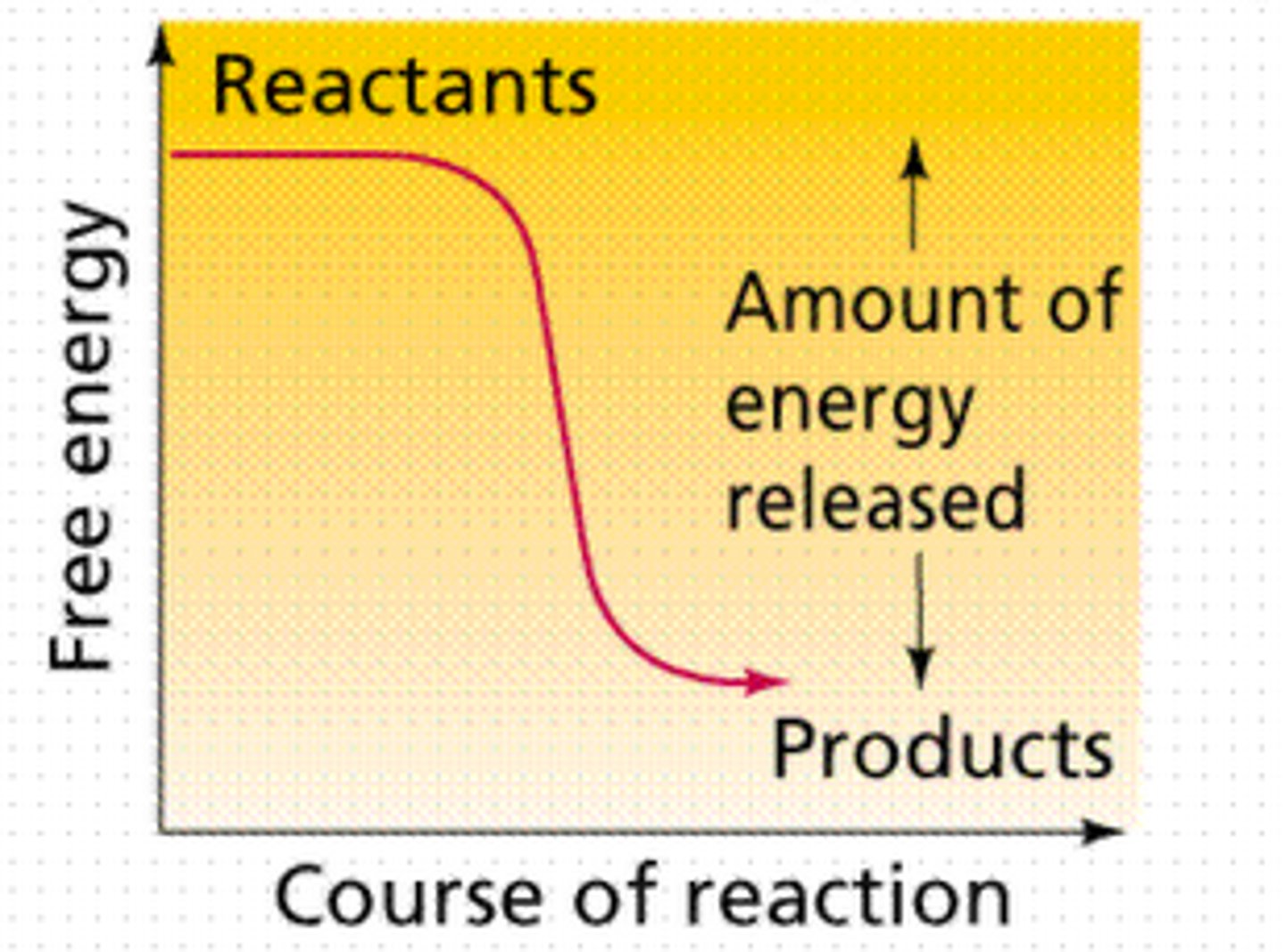
exergonic reactions
release energy and increase entropy, energy of reactants is less than energy of products
entropy
how spread out energy is, higher =harder to get work from it
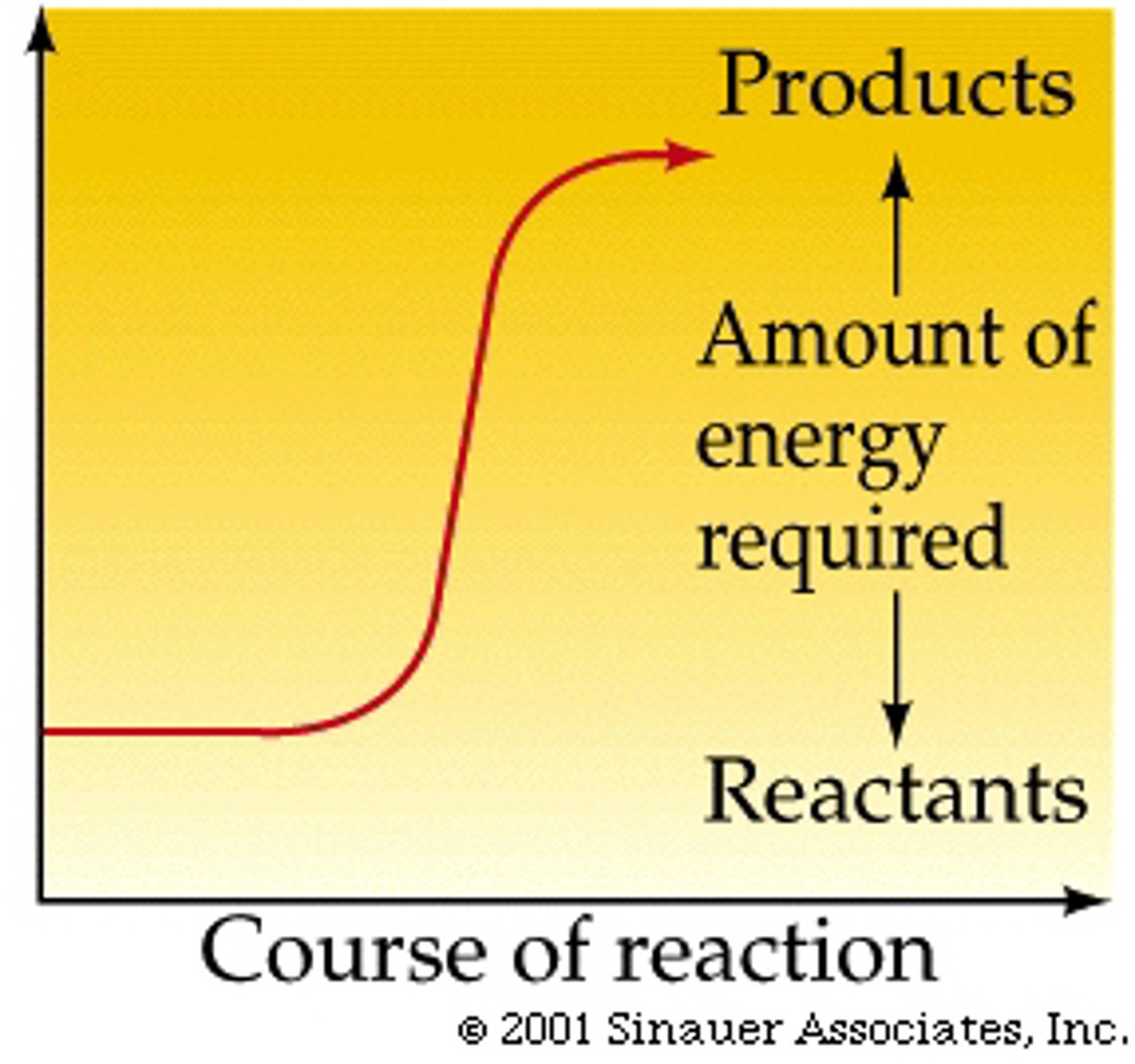
Endergonic reactions
require energy and decrease entropy, ex photosynthesis
ATP is used to
power work within cells, no sharing ATP between cells
to store energy cells must
take energy from food via cellular respiration or light and use it to make ATP from ADP and P
to release energy for work cells must
remove a phosphate group from ATP which creates ADP and P
energy coupling
linking an exergonic reaction to an endergonic one, provides energy to drive the endergonic reaction forward
formula for photosynthesis
6CO2+6H2O+light energy --> C6H12O6+6O2
the two phases of photosynthesis
light dependent and light independent (calvin cycle) reactions
light reactions
convert light energy into chemical energy (ATP)
calvin cycle
converts chemical energy in ATP and NADPH into carbohydrates which "fixes" carbon dioxide into high energy sugars
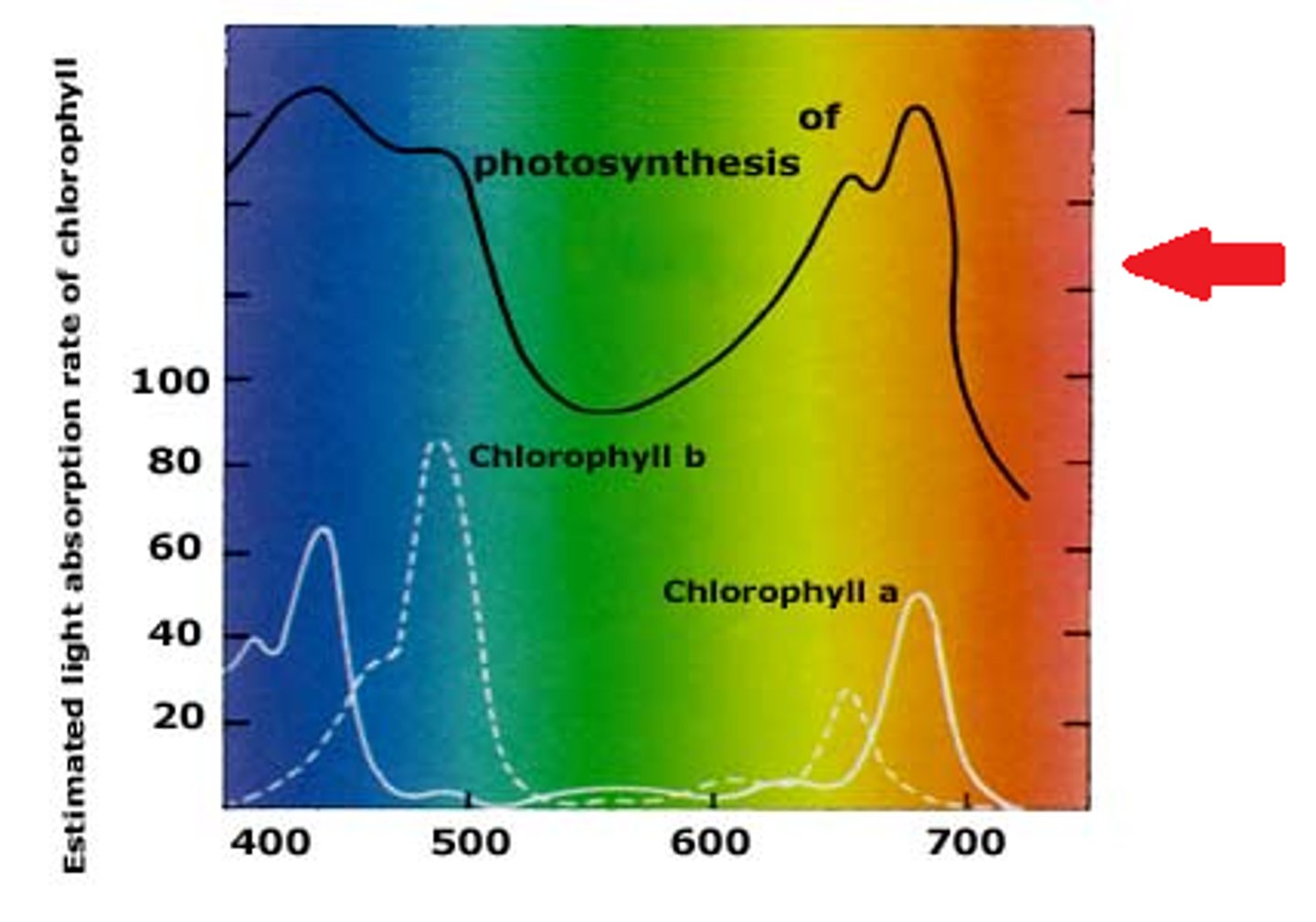
Chlorophyll function
the pigments that absorbs light energy in plant, absorbs most blue and red light and least green light
absorption spectrum
shows the amount of light absorbed at different light wavelengths
action spectrum
shows how various light wave length drive photosynthesis
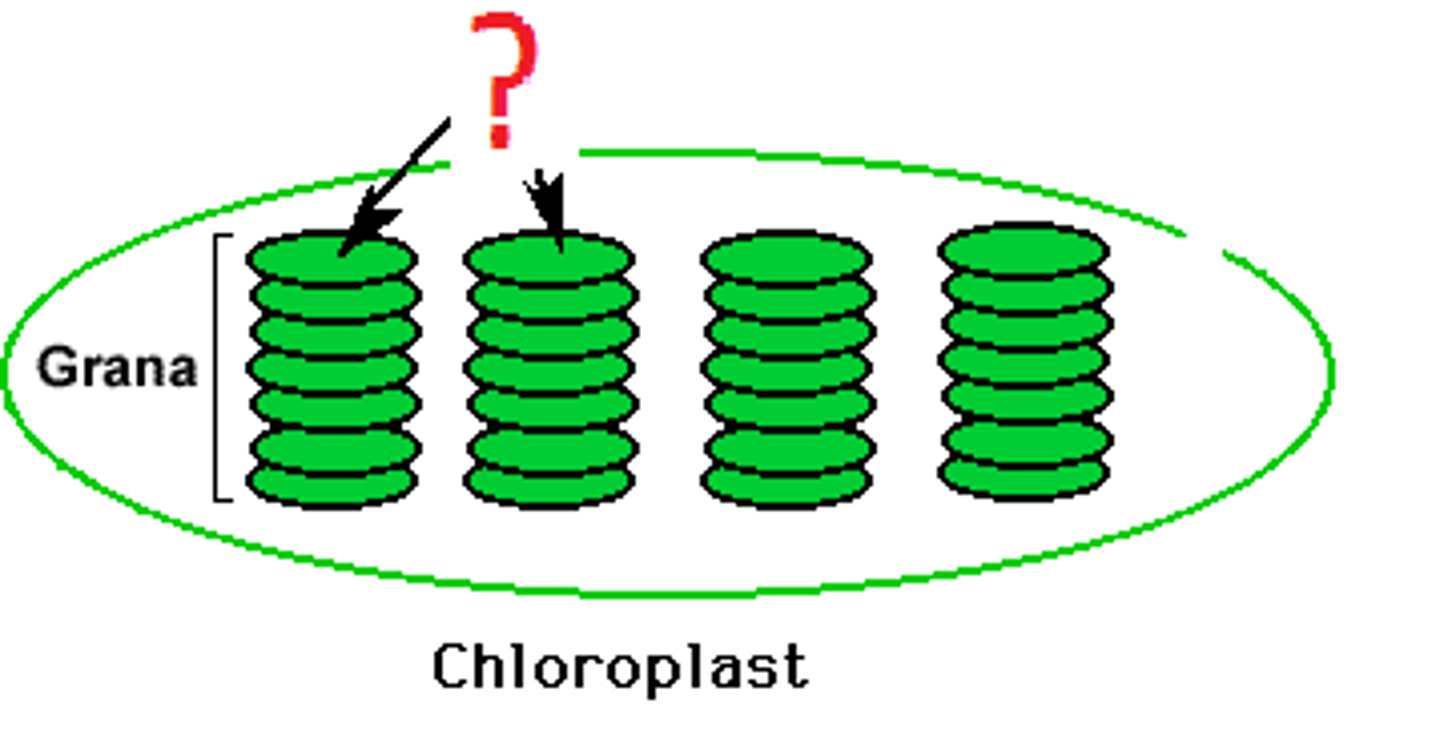
thylakoids
membrane bound sacs, contain membrane bound photosystems and chlorophyll for light reactions, organized into stacks called grana
stroma
the cytoplasm of the chloroplast
phase 1 of calvin cycle; carbon fixation phase
carbon dioxide gas is brought into the biosphere
Phase 2 of calvin cycle; energy investment and harvest phase
matter is pulled out and becomes part of the plant and part of you
phase 3 of clavin cycle; regeneration of RuBp
the regeneration of the starting compound.
how light dependent reaction gets light
thylakoids absorb it
formula for cellular respiration
C6H12+6O2-> 6 CO2+6 H2O+ Energy (Heat+ATP)
the three steps of cellular respiration
glycolysis, krebs cycle, electron transport chain
what glycolysis does
glucose is broken down to form 2 molecules of pyruvate, glucose is oxidized (loses electrons) and 2 ATP are generated
what the krebs cycle does
pyruvate enters the mitochondria where its further oxidized to yield intermediate molecules, oxidaion of intermediate is completed, extracted electrons are picked up again by NAD+ to form NADH and another FAD to form FADH2
what the electron transport chain does
series of proteins embedded in inner mitochondrial membrane, provides energy needed to provide energy for active transport and chemical reactions