Addition Reactions
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
general features of addition reactions
2Xs added across pi bond or X & Y are added arcoss pi bonds
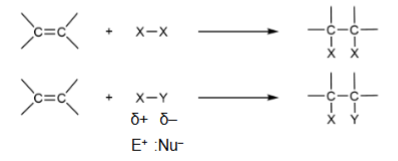
Syn addition
add at same time so on the same side
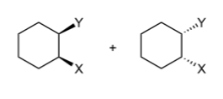
Anti addition
add at different times through bridged intermediate so on different sides
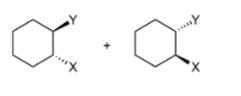
Random Additionally
add at different times, carbocation/carbon radical intermediate forms; can have same side or different sides
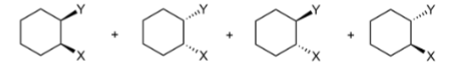
syn additions result in (stereochemistry)
enantiomers (mirror images) → 50:50 racemic mixture
anti addition results in (stereochemistry)
identical; only mesocompound bc achiral, internal mirror plane, & 2 chiral centers
Hydrohalogenation
adding H and halogen to C=C
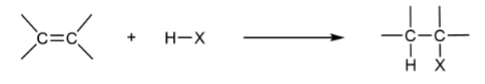
Hydrohalogenation produces
Markovnikov product (major) and Anti-Markovnikov products (minor)
Markovnikov’s Rule
When H-A is added to a C=C, the H+ adds to the least substituted carbon so a more stable carbocation can form
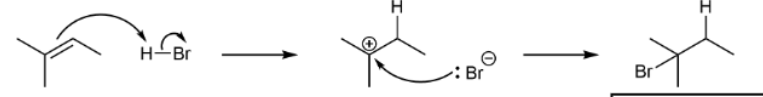
Hydrohalogenation using just H-X
produce Markovnikov as major product and anti-Markovnikov as minor product; all heterolytic cleavages & H bonds first & carbocation intermediates
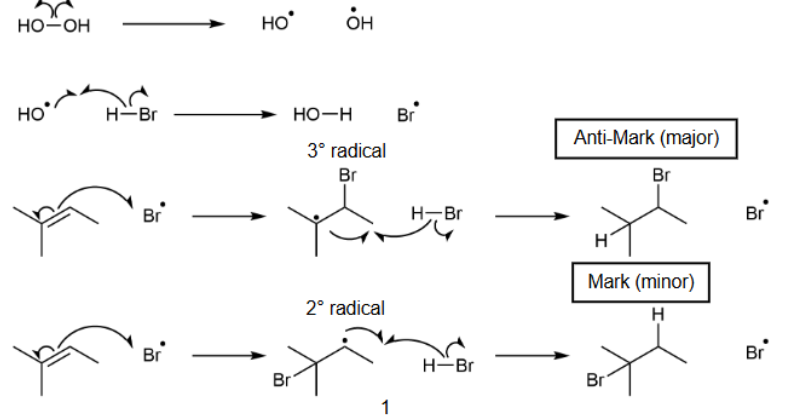
Hydrohalogenation with H-X and H2O2
gets Anti-Markovnikov as major product and Markovnikov as minor product; uses homolytic cleavage & carbon radical intermediates & Br binds first
Which halogenation (H-X or H-X & H2O2) gets markovnikov as major product?
with H-X
Which halogenation (H-X or H-X & H2O2) gets markovnikov as minor product?
H-X and H2O2
Hydration with acid-catalyzed
Markovnikov product is major; rearrangement; regenerates acid so only a small amount is needed; heterolytic cleaves & H bonds first & carbocation forms
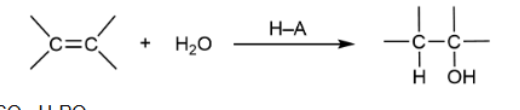
Hydration with Acid-catalyzed mechanism
heterolytic cleavage; H bonds first, then OH2, then hydrogen is removed by H2O to get OH on compound
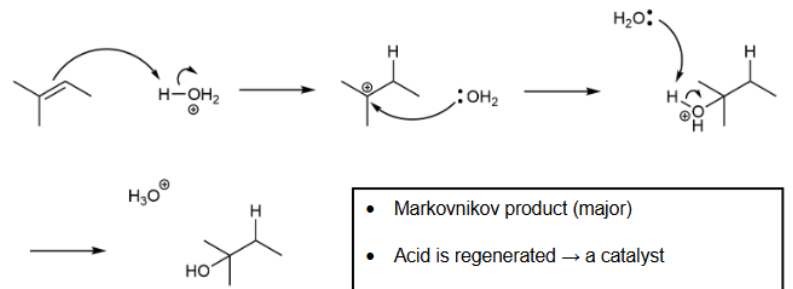
Common acids for acid-catalyzed hydration
H2SO4 & H3PO4
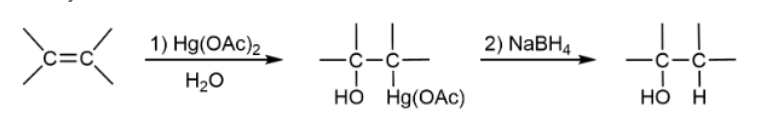
Oxymercuration-Demercuration Hydration
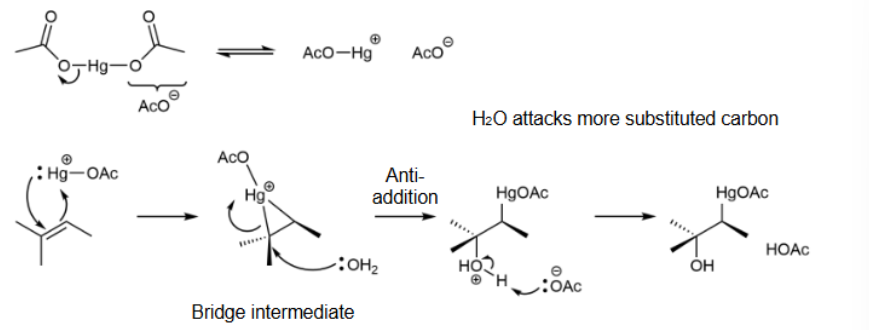
adds and removes mercury to get OH and H on compound; anti-addition & favors Markovnikov product; no rearrangment; heterolytic cleavages & forms bridged intermediates
Oxymercuration mechanism
bridged intermediate; H2O attacks carbon that is more substituted & anti-addition
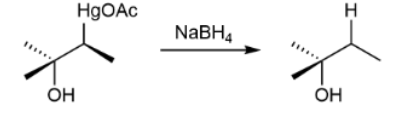
Demercuration mechanism
just know this; favors Markovnikov product & no rearrangement
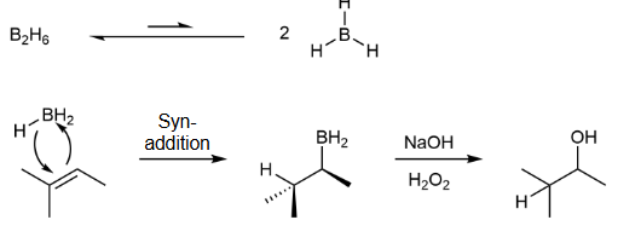
Hydroboration-Oxidation
favors Markovnikov; gets product with OH and H on compound; two steps
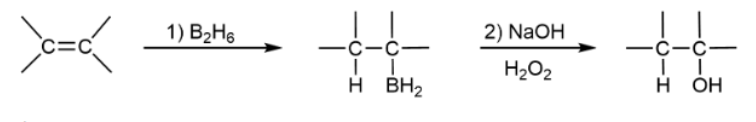
Hydroboration-Oxidation Mechanism
favors anti-Markovnikov; all heterolytic cleavages & syn addition & H bonds to the more substituted carbon because of sterics; no rearrangment
Types of Hydrohalogenation
use H-X or use H-X and H2O2
Types of Hydration
Acid-Catalyzed, Oxymercuration-Demercuration, Hydroboration-Oxidation
Which hydration methods favor Markovnikov products?
acid-catalyzed, Oxymercuration-Demercuration
Which hydration methods favor anti-Markovnikov products?
Hydroboration-Oxidation
Side of Addition (Stereoselectivity) of Acid-Catalyzed Hydration
random bc C+ intermediate; 50:50 racemic mixture
Side of Addition (Stereoselectivity) of Oxymercuration-Demercuration
anti bc stepwise
Side of Addition (Stereoselectivity) of Hydroboration-Oxidation
syn bc concerted
Rearrangment in hydration?
only in acid-catalyzed hydrations
Acid-Catalyzed reagents
H20, H+catylase
Oxymercuration-Demercuration reagents
1) Hg(OAc)2, H2O; 2) NaBH4
Hydroboration-Oxidation reagents
1) BH3; 2) NaOH, H2O2
Acid-Catalyzed regioselectivity
markovnikov
Oxymercuration-Demercuration regioselectivity
markovnikov
Hydroboration-Oxidation regioselectivity
anti-markovnikov
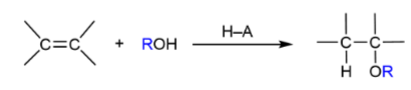
Alkene → Ether
acid-catalyzed or oxymercuration-demercuration; like hydration but with ROH; get H and OR
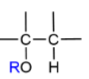
Alkene → ether acid-catalyzed method mechanism
OR on more substituted carbon; add H and OR
common acids for alkene → ether with acid-catalyzed
H2SO4, H3PO4
Alkene → ether with acid-catalyzed method reagents
ROH, HA
Alkene → ether with acid-catalyzed method regioselectivity
Markovnikov
Alkene → ether with acid-catalyzed method side of addition (stereoselectivity)
random; 50:50 mixture
Alkene → ether with acid-catalyzed method rearrangment
yes
Alkene → ether with oxymercuration-demercuration method mechanism
get RO and H in place of double bond; like hydration but with OR
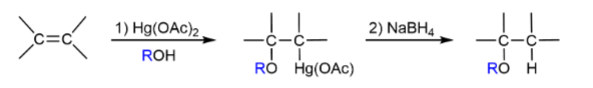
Alkene → ether with oxymercuration-demercuration method reagents
1) Hg(OAc)2, ROH; 2) NaBH4
Alkene → ether with oxymercuration-demercuration method regioselectivityq
stepwise/anti
Alkene → ether with oxymercuration-demercuration method rearrangement
no
Alkene → Di-Halogen
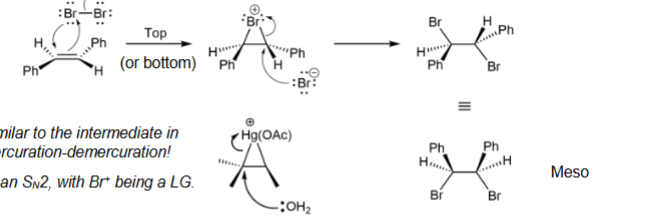
replace double bond with two hydrogens; meso and racemic mixture products with meso predicted in lab
Alkene → di-Halogen mechanism for meso
anti-addition; similar to intermediate in oxymercuration-demercuration and SN2
What does free radical halogenation target?
sp3 C-H; with heat and light
What does alkene → di-halogen target?
double bond
What are other common non-X2 halogenation reagents?
Alkene → Di-Halogen reagents
X2 or common non-X2
Alkene→Di-Halogen side of addition (stereoselectivity)
anti or syn
Alkene → Di-Halogen rearrangement
no
Alkene → Alcohol + Halogen
halohydrin
Halohydrin
X (Br, Cl, or I) and OH replace double bond
Halohydrin mechanism
anti-addition with the nucleophile attacking the more substituted carbon; bottom and top attacks; same first step as di-halogen
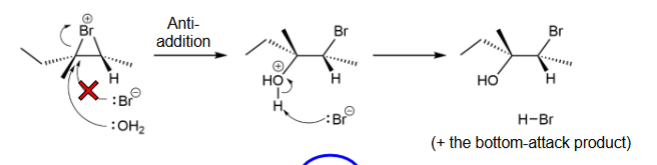
Stronger nucleophile: Br- or H2O
H2O
Where do nucleophiles attack?
more substituted carbon
Halohydrin reagents
X2 (Cl, Br, I) and H2O
Halohydrin side of addition (stereoselectivity)
anti-addition bc stepwise
Halohydrin rearrangement
no
Alkene → Alkane
hydrogenation
Hydrogenation
two hydrogens replace double bond
Hydrogenation mechanism
diastereomer products
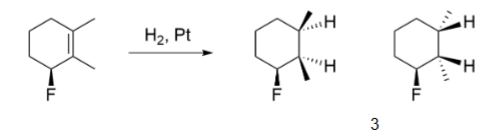
Hydrogenation reagents
H-H, metal catalyst (Pt, Pd, Ni)
Hydrogenation side of addition (stereoselectivity)
syn-addition
Hydrogenation rearrangement
no