Chemistry Study guide: Structure 1.1 and 1.2
5.0(2)
Studied by 9 peopleCard Sorting
1/34
There's no tags or description
Looks like no tags are added yet.
Last updated 6:32 PM on 9/11/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
1
New cards
what is the set up of distillation?
(know the parts and their functions)
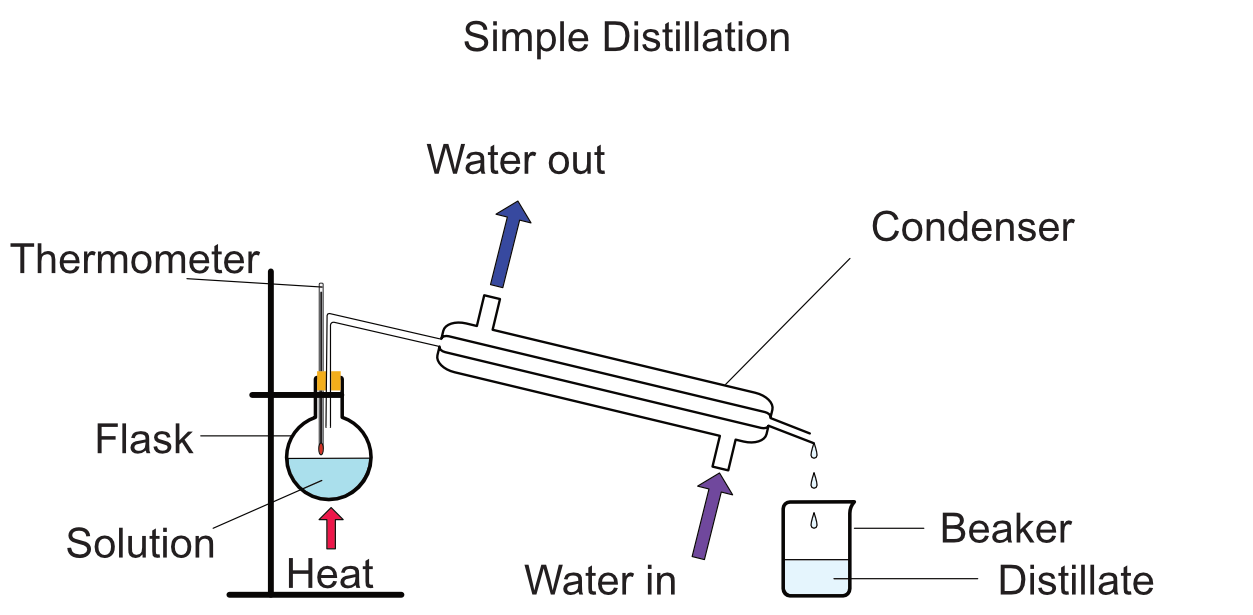
2
New cards
what is the plum pudding model?
* thompson
* disproved by bohr and rutherford because of the gold foil method and bohrs model
* disproved by bohr and rutherford because of the gold foil method and bohrs model
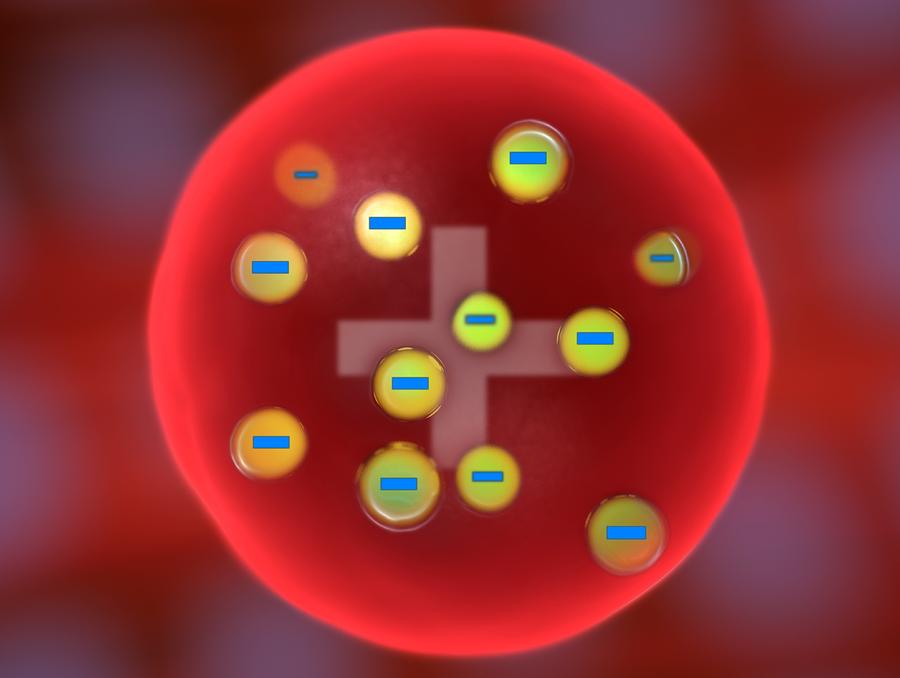
3
New cards
what is bohrs model?
disproved the plum pudding model
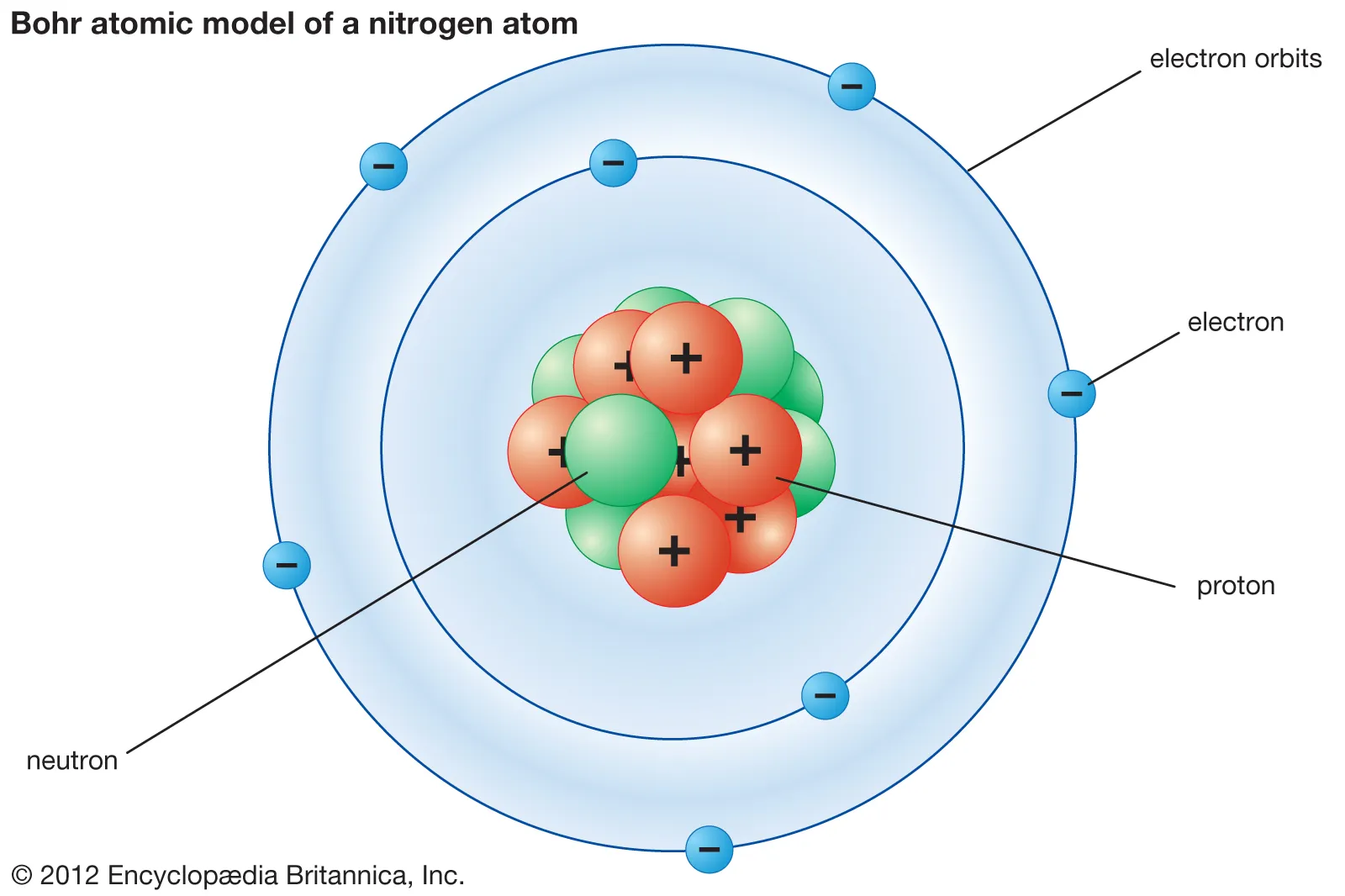
4
New cards
how many sigfigs are in 1) 10.04 2) 0.0065 3) 6000
1) 4 2) 2 3) 1
5
New cards
what are the changes of matter? (on a heating curve)
heating (left → right) cooling (right → left)
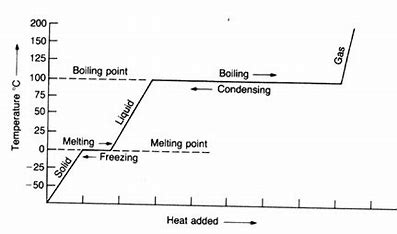
6
New cards
what are the conversions for Celsius and Kelvins?
100C → 373K, 0C → 273K, -273C → 0K
7
New cards
How do you double the kinetic energy of a state of matter?
covert the Celsius temperature to Kelvins, double it, then convert it back to Celsius
8
New cards
what are atoms?
the smallest unit of a particle (ex. H, Cl, H-Cl is two atoms, H-H is two hydrogen atoms)
9
New cards
what are elements?
a pure substance that cannot be broken down into a simpler substance by chemical means (all known elements are on the periodic table) (ex. O**2, H*2, S*8, C*60)*
10
New cards
what are compounds?
a pure substance formed from two or more different elements chemically joined in a fixed ratio (ex. HCl, H\**2O, Al*2O*2, NaCl*). compounds have different properties from the elements they are made from (ex. NaCl has different properties than the elements it is made from)
11
New cards
what are molecules?
two or more atoms (covalent → two non-metal elements)
12
New cards
what are pure substances?
a single kind of matter that cannot be separated into other kinds of matter by physical means (ex. element, compound)
13
New cards
what is a mixture (mixed substance)?
contains more than one element and/or compound that are not chemically bonded together and so retain their individual properties (ex. water and oil)
14
New cards
what is a homogeneous mixture?
has the same uniform appearance and composition throughout (ex. a salt solution)
15
New cards
what is a heterogeneous mixture?
consists of visibly different substances pr phases (ex. sand and water)
16
New cards
what are the techniques of separation?
* filtration:
* separation technique used to separate a heterogeneous mixture like sand and water
* evaporation:
* separation technique used to separate a homogeneous mixture like a salt solution
* distillation:
* separation technique used to separate two miscible (capable of being mixed) liquids with different boiling points
* chromatography:
* separation technique used to separate a mixture of solutes in a solvent
* separation technique used to separate a heterogeneous mixture like sand and water
* evaporation:
* separation technique used to separate a homogeneous mixture like a salt solution
* distillation:
* separation technique used to separate two miscible (capable of being mixed) liquids with different boiling points
* chromatography:
* separation technique used to separate a mixture of solutes in a solvent
17
New cards
what are the properties of solids, liquids, and gases?
* solid:
* fixed shape
* fixed volume
* limited compressibility
* has no fluidity
* liquid:
* no fixed shape
* fixed volume
* limited compressibility
* flows easily
* gas:
* no fixed shape
* no fixed volume
* high compressibility
* diffuses easily
* fixed shape
* fixed volume
* limited compressibility
* has no fluidity
* liquid:
* no fixed shape
* fixed volume
* limited compressibility
* flows easily
* gas:
* no fixed shape
* no fixed volume
* high compressibility
* diffuses easily
18
New cards
what does endothermic mean?
energy is absorbed (hot) (solid → liquid (melting), liquid → gas (evaporation), solid → gas (sublimation))
19
New cards
what does exothermic mean?
energy is released (cold) (gas → liquid (condensation), liquid → solid (freezing), gas → solid (deposition))
20
New cards
what are the state symbols for chemical reactions?
s = solid, l = liquid, g = gas, aq = aqueous (in solution)
21
New cards
what are nucleons?
protons and neutrons (they are in the nucleus)
22
New cards
what are the relative masses and relative charges of protons, neutrons, and electrons?
* protons:
* relative mass: 1
* relative charge: +1
* neutrons:
* relative mass: 1
* relative charge: 0
* electrons:
* relative mass: 5 x 10\*-4
* relative charge: -1
* relative mass: 1
* relative charge: +1
* neutrons:
* relative mass: 1
* relative charge: 0
* electrons:
* relative mass: 5 x 10\*-4
* relative charge: -1
23
New cards
how do you draw an atom symbol?
(if it has a charge it is an ion and the charge goes in the top right corner)

24
New cards
what is a cation and an anion?
* cation:
* positive ion (more protons than electrons)
* anion:
* negative ion (more electrons than protons)
* positive ion (more protons than electrons)
* anion:
* negative ion (more electrons than protons)
25
New cards
what is the only element that has more protons than neutrons in an atom?
hydrogen; 1 proton and no neutrons
26
New cards
what are isotopes?
* isotopes are elements with different amounts of neutrons and therefore different mass numbers (same protons)
* differ in physical properties that depend on mass (density, rate of diffusion, etc)
* the different physical properties include melting point, boiling point, density, etc
* same chemical properties (react the same way)
* differ in physical properties that depend on mass (density, rate of diffusion, etc)
* the different physical properties include melting point, boiling point, density, etc
* same chemical properties (react the same way)
27
New cards
Isotopes have the same a) __ properties, but different b) __ properties
a) chemical b) physical
28
New cards
what is important about the mass spectrum?
* gives the number of isotopes
* gives the abundance of each isotope
* gives the abundance of each isotope
29
New cards
what is relative atomic mass?
* average mass of an atom according to the relative abundances of its isotopes
30
New cards
how do you find relative atomic mass?
multiply the percentage of the isotope with the mass number and add that to the next isotope. ex. Cl-35 (75%) and Cl-37 (25). (75/100 x 35)+ (25/100 x 37) = relative atomic mass amu (atomic mass unit)
31
New cards
example question of RAM (relative atomic mass)
iridium has a RAM of 192.22 and has isotopes Ir-191 and Ir-193. Calculate the percentage composition.
Work:
191 \~ (1 - x) because it is 100- the percentage of 193
193 \~ x because it is the percentage being looked for
191 (1 - x) + 193x = 192.22
Answer:
x = 0.61, so
Ir-193 = 61%
Ir-191 = 39%
Work:
191 \~ (1 - x) because it is 100- the percentage of 193
193 \~ x because it is the percentage being looked for
191 (1 - x) + 193x = 192.22
Answer:
x = 0.61, so
Ir-193 = 61%
Ir-191 = 39%
32
New cards
what is a separating funnel?
* technique used for immiscible liquids (liquids that don’t mix together)
* separating funnel used in the progress
* method:
* once liquids are in the funnel you wait until they form two layers
* denser liquid sinks to bottom and other liquid goes to top
* conical flask is placed at bottom to collect the denser liquid
* valve allows you to control when and how the liquid is let through down to the conical flask
* separating funnel used in the progress
* method:
* once liquids are in the funnel you wait until they form two layers
* denser liquid sinks to bottom and other liquid goes to top
* conical flask is placed at bottom to collect the denser liquid
* valve allows you to control when and how the liquid is let through down to the conical flask
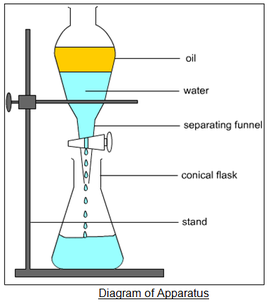
33
New cards
what is gravity filtration?
* filtration also separates an insoluble solid from a liquid, but it is a more reliable way
* method:
* place filtration paper (making sure it is folded correctly) into the filter
* either use a stand to support the filter over a beaker
* pour the mixture into the funnel and the liquid should be collected in the beaker/conical flask while the solid, now called the residue, stays on the filter paper
* the liquid is now called the filtrate
* to gain back the solid from the residue, evaporate the water from the filter paper
* method:
* place filtration paper (making sure it is folded correctly) into the filter
* either use a stand to support the filter over a beaker
* pour the mixture into the funnel and the liquid should be collected in the beaker/conical flask while the solid, now called the residue, stays on the filter paper
* the liquid is now called the filtrate
* to gain back the solid from the residue, evaporate the water from the filter paper
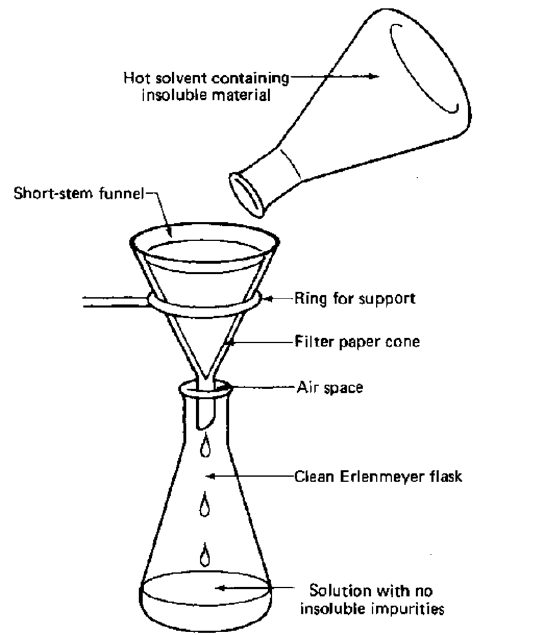
34
New cards
what was the Gold Foil Experiement?
* Rutherford
* demonstrated the atom is largely empty space occupied by the negatively charged electrons surrounding a very dense positively charged nucleus.
* demonstrated the atom is largely empty space occupied by the negatively charged electrons surrounding a very dense positively charged nucleus.
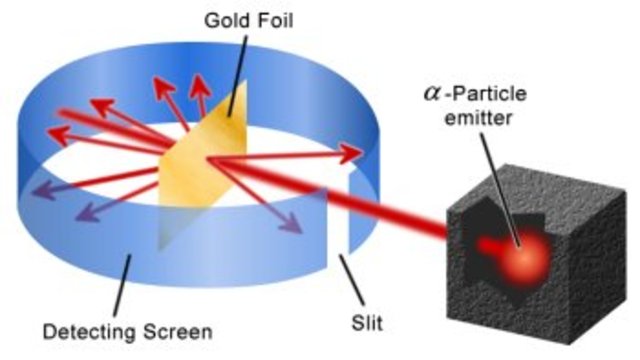
35
New cards
WHAT IS THE FORMAT OF THE TEST?
15 MULTIPLE CHOICE
10 SHORT/LONG ANSWER
10 SHORT/LONG ANSWER