orgo 2 reactions
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
2 degree alcohol to ketone
jones or any oxidizing agent
1 degree alcohol to carboxylic acid
strong oxidizing agent
1 degree alcohol to aldehyde
mild oxidizing agent
3 degree alcohol
no reaction
aldehyde to 1 degree alcohol
NaBH4, MeOH or 1) LAH 2) H2O
ketone to 2 degree alcohol
NaBH4, MeOH or 1) LAH 2) H2O
carboxylic acid or ester to 1 degree alcohol
1) LAH 2) H2O
strong oxidizing agents
H2CrO4, KMnO4, jones
mild oxidizing agents
PCC, HOCl via NaOCl with CH3COOH (acetic acid) at 0 degrees
ester to aldehyde
1) DiBAL-H 2) H2O
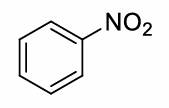
benzene + HNO3 and H2SO4
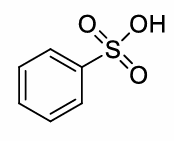
benzene + H2SO4
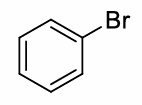
benzene + Br2 and FeBr3 (or AlCl3)
benzene + RCl and AlCl3
benzene + ketone structure with Cl and AlCl3
acid chloride and carboxylic acid with pyridine or TEA
anhydride
acid chloride and alcohol with pyridine or TEA
ester
acid chloride and amine
amide
acid chloride and water
carboxylic acid
anhydride and alcohol
ester
anhydride and amine
amide
anhydride and water
carboxylic acid
carboxylic acid and heat
anhydride
carboxylic acid and alcohol
ester
birch reduction
benzene and Na, NH3, MeOH make diene
polymerization of styrene
styrene and ROOR make chains of styrene
SNAR
benzene with X and KOH, HCl at 70° make benzene with alcohol (replaces X)
strong nucleophilic addition to carbonyl
carbonyl and KOH (aq) make acetal
weak nucleophilic addition to carbonyl
carbonyl and water, HA make acetal
carbonyl with excess alcohol
carbonyl and 2 alcohols make acetal
acetal formation with chained alcohol
carbonyl and chained alcohol makes acetal with o’s connected
imine formation (Shiff base)
carbonyl and primary amine/ammonia make imine
enamine formation
carbonyl with secondary amine/ammonia make enamine
Wolff-Kishner
aldehyde/ketone to imine, imine and KOH, H2O, heat make alkane
acetal back to carbonyl
acetal and water make carbonyl
Wittig reaction
alkane bromine and 1) PPh3 2) BuLi 3) carbonyl make alkene (Z product)
HWE Wittig
alkane bromine and 1) P(OEt)3 2) BuLi 3) carbonyl make alkene (E product)
aldehyde to carboxylic acid (slow)
aldehyde and oxygen make carboxylic acid
aldehyde to carboxylic acid (fast)
aldehyde and Ag2O or KMnO4, KOH make carboxylic acid
benzylic oxidation (alkyl benzene)
alkyl benzene and KMnO4 make benzoic acid
benzylic oxidation (benzyl alcohol)
benzyl alcohol and MnO2 or PCC make benzaldehyde
benzylic bromination
benzene with group attached and NBS or Br2, heat make benzylic bromide
reductive amination
imine and NaBh3CN,[H+] make cyclohexane with amine group
Fischer esterification
benzoic acid and excess alcohol, HA make benzyl ester
cyclic esterification
6 chain carboxylic acid and HA make lactone