1. Transition Metals - The Basics + Complex Ions
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Transition Metal definition
A transition metal is a metal that can form one or more stable ions with an incomplete d sub-shell.
What are the two elements in the d block which aren’t transition metals?
Scandium and zinc
Why aren’t Scandium and zinc transition metals?
Their stable ions don’t have incomplete d sub-levels.
Physical properties - 2
high density
high melting and boiling points
Chemical Properties of transition metals - 4
They form complexes
They form coloured ions
Variable oxidation states
Good catalysts
what causes the special chemical properties of transition metals?
their incomplete d sub-levels.
what is a complex?
These form from transition metals, consisting of a central metal ion, surrounded by ligands.
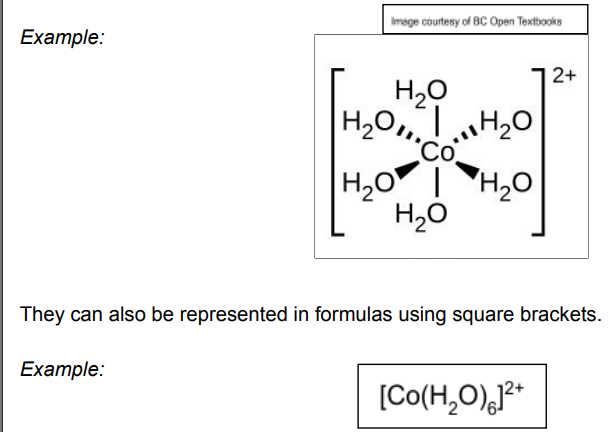
What are ligands?
An atom, ion or molecule that donates a pair of electrons to a central metal ion.
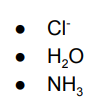
list the monodentate ligands
What are the bidentate ligands
ethane-1,2-diamine
ethandioate ions
What are the multidentate ligands
EDTA4-
Haem groups
Coordination number
The number of co-ordinate bonds that are formed with the central metal ion. (usually 6 and 4)
equation for the oxidation state of the metal ion
the oxidation state of the metal ion = The total charge of the complex - The sum of the charges of the ligands.
Ti ions, oxidation state and colour
Ti2+ → +2 → violet
Ti3+ → +3 → purple
V ions, oxidation state and colours
V2+, +2, violet
V3+, +3, green
VO(2+), +4, blue
VO2 (+), +5, yellow
Cr ions, oxidation state and colours
Cr3+, +3, green
Cr2O7 (2-), +6, orange
Mn ions, oxidation state and colours
Mn2+, +2, very pale pink/ colourless
MnO4 (2-), +6, green
MnO4 (-), +7, purple
Fe ions, oxidation state and colours
Fe2+, +2, pale green
Fe3+, +3, yellow/purple
Co ions, oxidation state and colours
Co2+, +2, pink
Ni ions, oxidation state and colours
Ni2+, 2+, green
Cu ions, oxidation state and colours
Cu2+, +2, pale blue