Chapter 20 - Electron Transport Chain
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
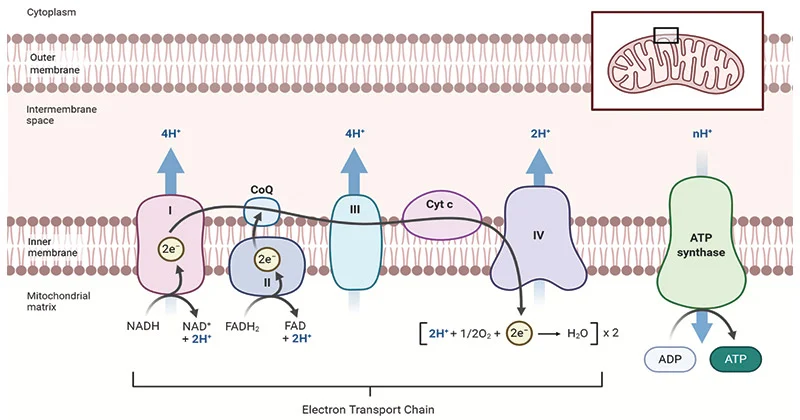
Where does the electron transport chain take place and what is it?
Four large protein complexes embedded in the inner mitochondrial membrane; three of these complexes pump protons out of the matrix into the intermembrane space, which generates a proton gradient used to power ATP synthesis
Where does the citric acid cycle occur?
In the mitochondrial matrix
Does ATP synthase aid in electron flow?
NO, it uses the proton-motive force generated by electron flow to synthesize ATP
Is the outer mitochondrial membrane permeable to small molecules and ions?
Yes, it is permeable to these because of channel proteins called mitochondrial porins → most abundant protein in outer mitochondrial membrane
Is the inner mitochondrial membrane permeable to small molecules and ions?
No, the inner membrane is impermeable to nearly all ions and polar molecules
What are the relative positions and roles of complexes of the ETC?
Complexes I and III are the major proton pumpers → each pump 4 H+
Complex II does not pump protons
Complex IV is the final electron acceptor, uses O2 → pumps 2 H+
What is reduction/redox potential?
A measure of a molecule’s tendency to donate or accept electrons
Negative reduction potential → readily donates electrons, strong reducing agent, wants to get oxidized; ex. NADH
Positive reduction potential → readily accepts electrons, strong oxidizing agent, wants to get reduced; ex. O2
High reduction potentials will release more free energy when reduced, allowing more protons to be pumped
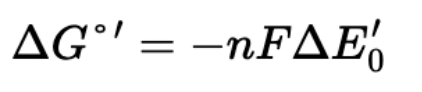
How do electrons flow?
From low reduction potential (more negative, NADH) to high reduction potential (more positive, O2)
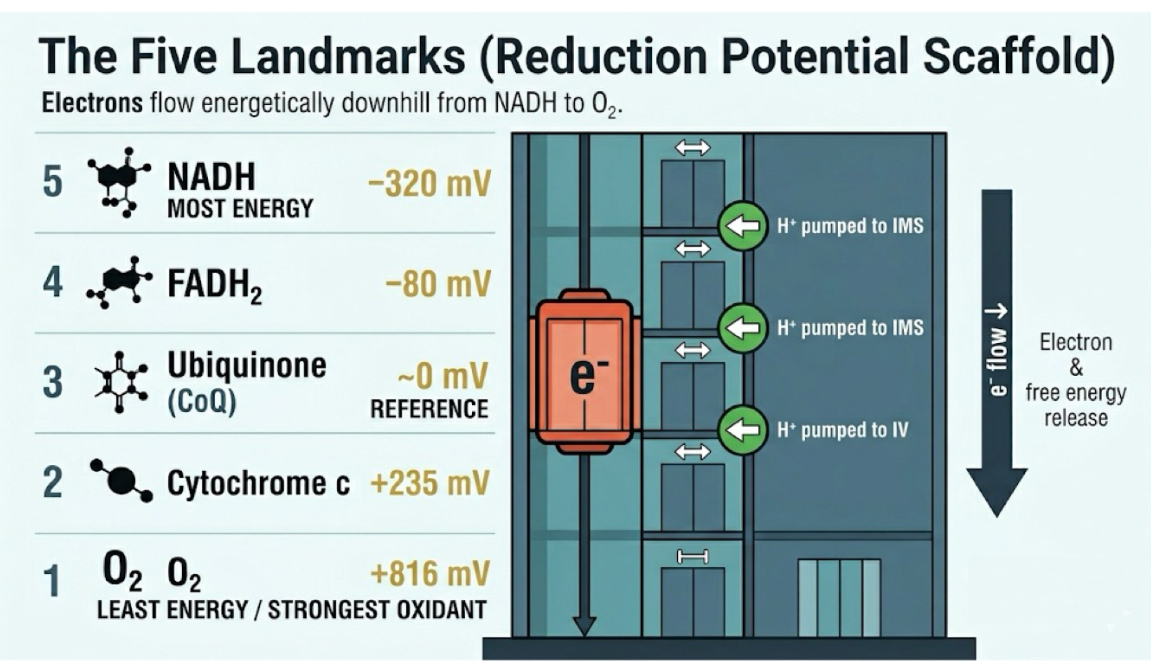
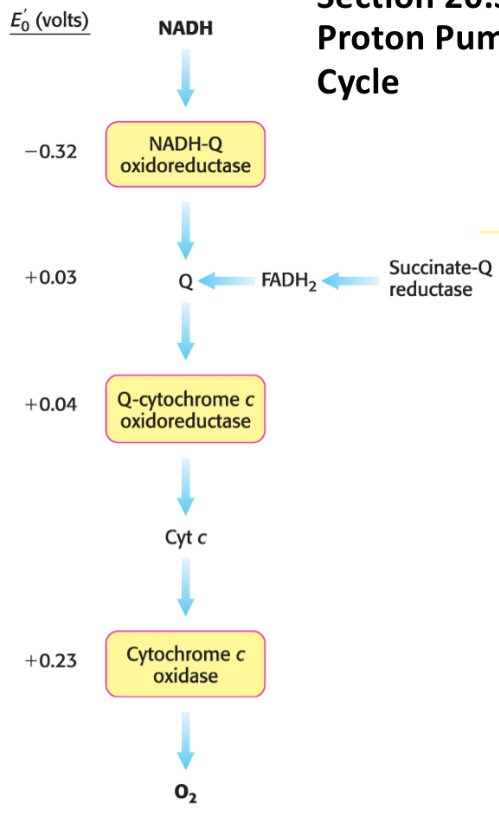
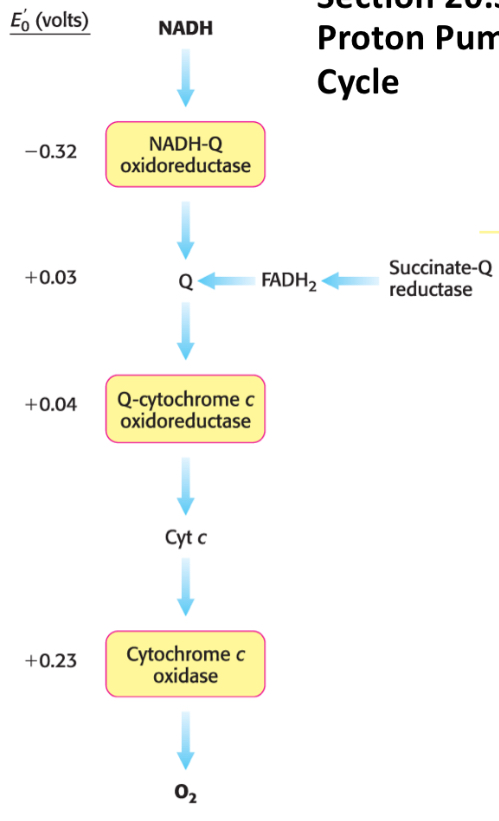
What are the 5 landmark reduction potentials in the ETC?
Top (most energy, strongest reductant)
NADH → -320 mV
FADH2 → -80 mV
Ubiquinone (CoQ) → 0 mV
Cytochrome C → +235 mV
O2 → +816 mV
Bottom (least energy, strongest oxidant)
Where does NADH enter the ETC and how much ATP does it yield?
NADH enters at Complex I via flavin mononucleotide (FMN) and yields ~2.5 ATP
FMN accepts 2 e- and 1 H+ from NADH, becoming FMNH2
Where does FADH2 enter the ETC and how much ATP does it yield?
FADH2 enters at Complex II and yield ~1.5 ATP
What are the electrons carriers in the ETC?
FMN → where NADH passes e- to in Complex I
Fe-S cluster proteins → present in Complexes I, II, and III
Fe-Heme cytochromes → present in Complex III, cytochrome c, and Complex IV
Coenzyme Q → lipophilic, mobile shuttle b/w Complex I/II and III
What is the role of coenzyme Q (CoQ)?
CoQ carries electrons between Complex I/II and Complex III
lipophilic, can move through IM membrane
Three oxidation states: full oxidized ubiquinone (Q), semiquinone radical (QH), fully reduced ubiquinol (QH2)
bridge between fixed complexes in ETC
carries 2 e-
What is fraxatin and what can mutations in the fraxatin gene cause
Fraxatin is a protein required for synthesis of Fe-S proteins, required for Complexes I, II and III, so without it the ETC and ATP synthesis fails causing neurodegeneration and cardiomyopathy
What three complexes in the ETC pump protons?
Complexes I, III, and IV
Where is [H+] high and where is [H+] low?
[H+] high in IM space, [H+] low in matrix
![<p>[H+] high in IM space, [H+] low in matrix</p>](https://assets.knowt.com/user-attachments/71adeb51-c3a4-4a5e-921e-32b8da514219.jpg)
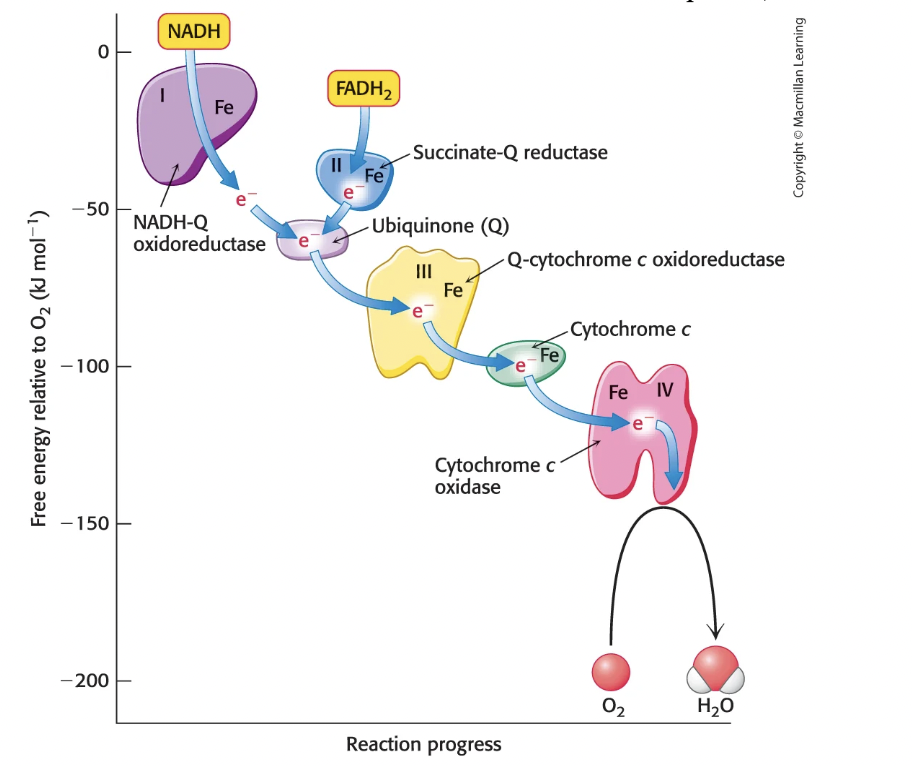
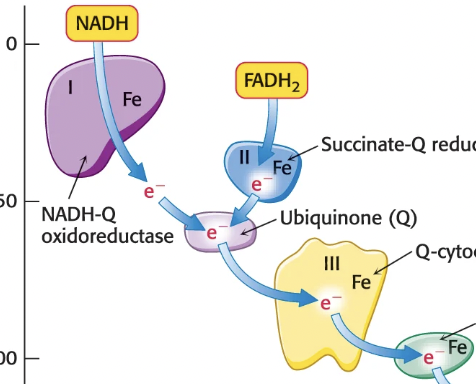
How do electrons move from NADH to O2?
Through Complex I (NADH-Q oxioreductase), then Complex III (Q-cytochrome c oxioreductase), then Complex IV (cytochrome c oxidase), then O2
How do electrons move from FADH2 to O2?
Through Complex II (succinate-Q reductase), to ubiquinone (generates QH2), to Complex III (Q-cytochrome c oxioreductase), then Complex IV (cytochrome c oxidase), then O2
Which complex in the ETC is the largest?
Complex I NADH-Q oxioreductase
Which complex in the ETC contains the succinate dehydrogenase used in the citric acid cycle?
Complex II succinate-Q reductase
Why do electrons from FADH2 feed into the chain downstream and NADH and what is the result?
They feed into the chain downstream because FADH2 has a lower reduction potential (more positive); this is why Complex II does not pump protons and FADH2 yields less ATP overall
What does cytochrome c do?
shuttles electrons from Complex III to Complex IV
carries 1 e-
soluble in water
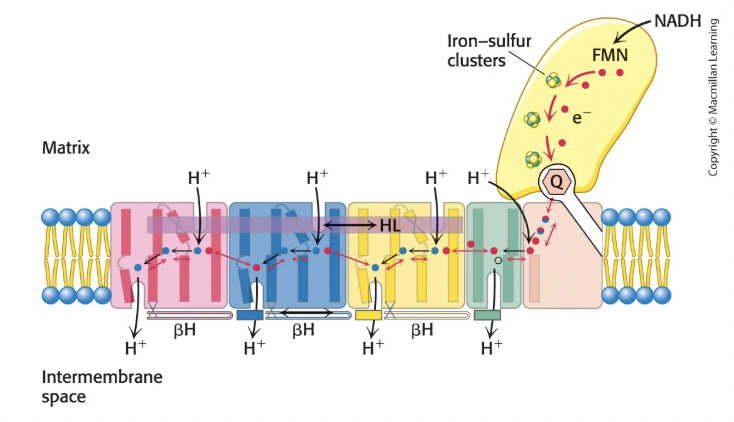
How does Complex I NADH-Q oxioreductase work?
Electrons are passed from NADH to FMN and then through series of Fe-S clusters until they reach CoQ, forming QH2 → all occurs in extramembranous part of Complex I in matrix
QH2 leaves Complex I for the Q pool in the hydrophobic interior of the IM membrane
QH2 travels to Complex III via CoQ
Flow of electrons leads to pumping of 4 H+ into IM space
L-shaped structure of Complex I separates substrate recognition from proton pump
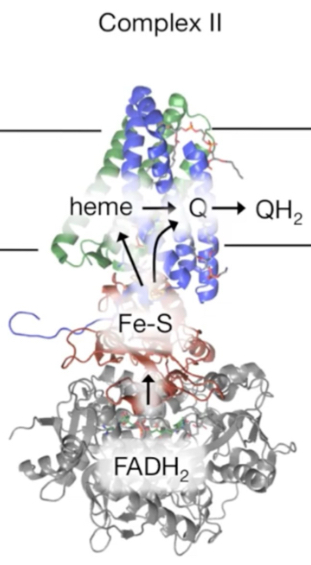
How does Complex II succinate-Q reductase work?
Succinate dehydrogenase within Complex II oxidizes succinate → fumarate in TCA cycle, forming FADH2
Electrons are transferred from FADH2 to Fe-S clusters, then to CoQ, forming QH2 that enters Q pool in hydrophobic interior of IM membrane
QH2 travels to Complex III via CoQ
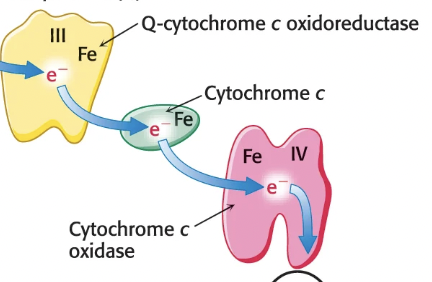
How does Complex III Q-cytochrome c oxidoreductase work?
Receives electrons from QH2 in CoQ → QH2 carries two e-, but cytochrome c can only accept one e-, so the other e- must move through the Q cycle
This results in 4 H+ pumped into IM space and electrons passed to cytochrome c, which carries them to Complex IV
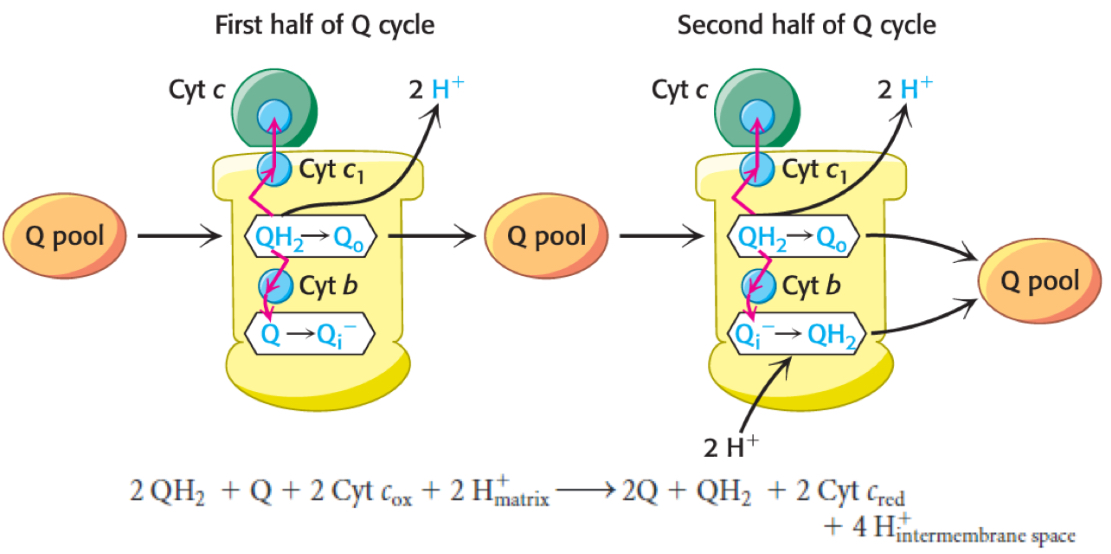
How does the Q cycle work?
Two QH2 required per cycle
QH2 is oxidized, releasing two H+ and two e-; one e- travels to cytochrome c and continues along ETC
simultaneously, the other e- piles onto Q in Complex III, forming semiquinone Q.-
Semiquinone needs a second e- to become fully reduced to QH2, which is provided when the second QH2 enters and becomes oxidized, passing one e- to cytochrome c and the other to semiquinone, forming QH2 that enters the Q pool
2 QH2 + 2 cytochrome cox → 2 cytochrom cred + 1 QH2 + 4 H+
How does Complex IV cytochrome c oxidase work?
Complex IV accepts four e- from four molecules of cytochrome c to catalyze reduction of O2 to H2O
electrons moves from cytochrome c to Cu center to Heme to O2
Removes 8 protons from matrix → 4 used to reduce O2 to H2O, 4 pumped into IM space
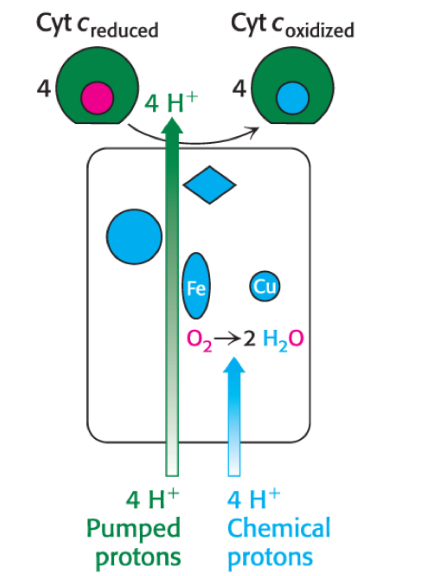
Does Complex IV pump 2 H+ or 4 H+ into the IM space?
Complex IV requires 4 cytochrome c molecules to fully reduce O2 to H2O, in which it pumps 4 H+ into the IM space, however each molecule of NADH/FADH2 results in only 2 cytochrome c molecules being reduced by Complex III, so only 2 H+ are pumped into the IM space by Complex IV PER NADH, but in the complete reduction of O2 to H2O (which would require 2 NADH/FADH2 molecules), 4 H+ are pumped into the IM space
Per NADH, how many H+ are pumped into the IM space and how much ATP is produced?
Complex I → 4 H+
Complex III → 4 H+
Complex IV → 2 H+
Total: 10 H+ pumped into IM space per NADH
1 ATP molecule requires ~4 H+, so ~2.5 ATP produced per NADH
Per FADH2, how many H+ are pumped into the IM space and how much ATP is produced?
Complex II → 0 H+
Complex III → 4 H+
Complex IV → 2 H+
Total: 6 H+ pumped into IM space per FADH2
1 ATP molecule requires ~4 H+, so ~1.5 ATP produced per FADH2
What is a respirasome?
Three of the ETC components organized into a supercomplex
Two copies of Complex I and IV surround a dimer of Complex III
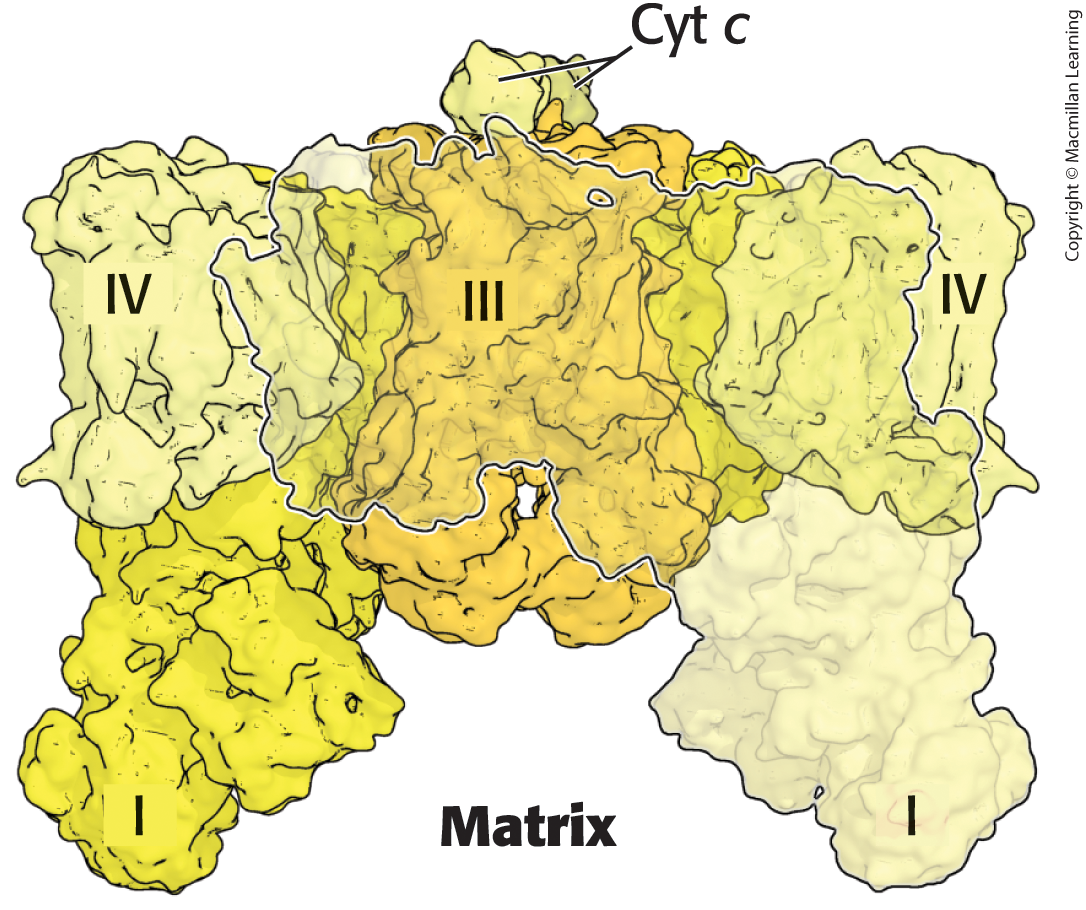
What are the two common electron leak sites that can produce ROS by partially reducing O2?
Complex I and Complex III
What enzymes protect against ROS?
Superoxide dismutase → scavenges radicals to form O2 and H2O2
Catalase → further neutralizes H2O2 into H2O and O2