BI108 Quiz #2
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
Electrical Energy
Energy related to interactions among charged particles.
Thermal Energy
The energy of motion in ions and molecules, measured as temperature.
Potential Energy
Energy that is related to an object's position.
Chemical Reaction
Conversion of substances (ions or molecules) into other substances via breaking and forming chemical bonds.
Free Energy
The total energy available to do work—a combination of entropy and thermal and potential energy.
Energetic Coupling
A phosphorylation reaction that makes a nonspontaneous reaction spontaneous, because it raises the free energy of the reactants. Uses free energy released from spontaneous reactions to drive nonspontaneous reactions
Endergonic Reaction
One that results in an increase in free energy; another way of referring to a nonspontaneous reaction.
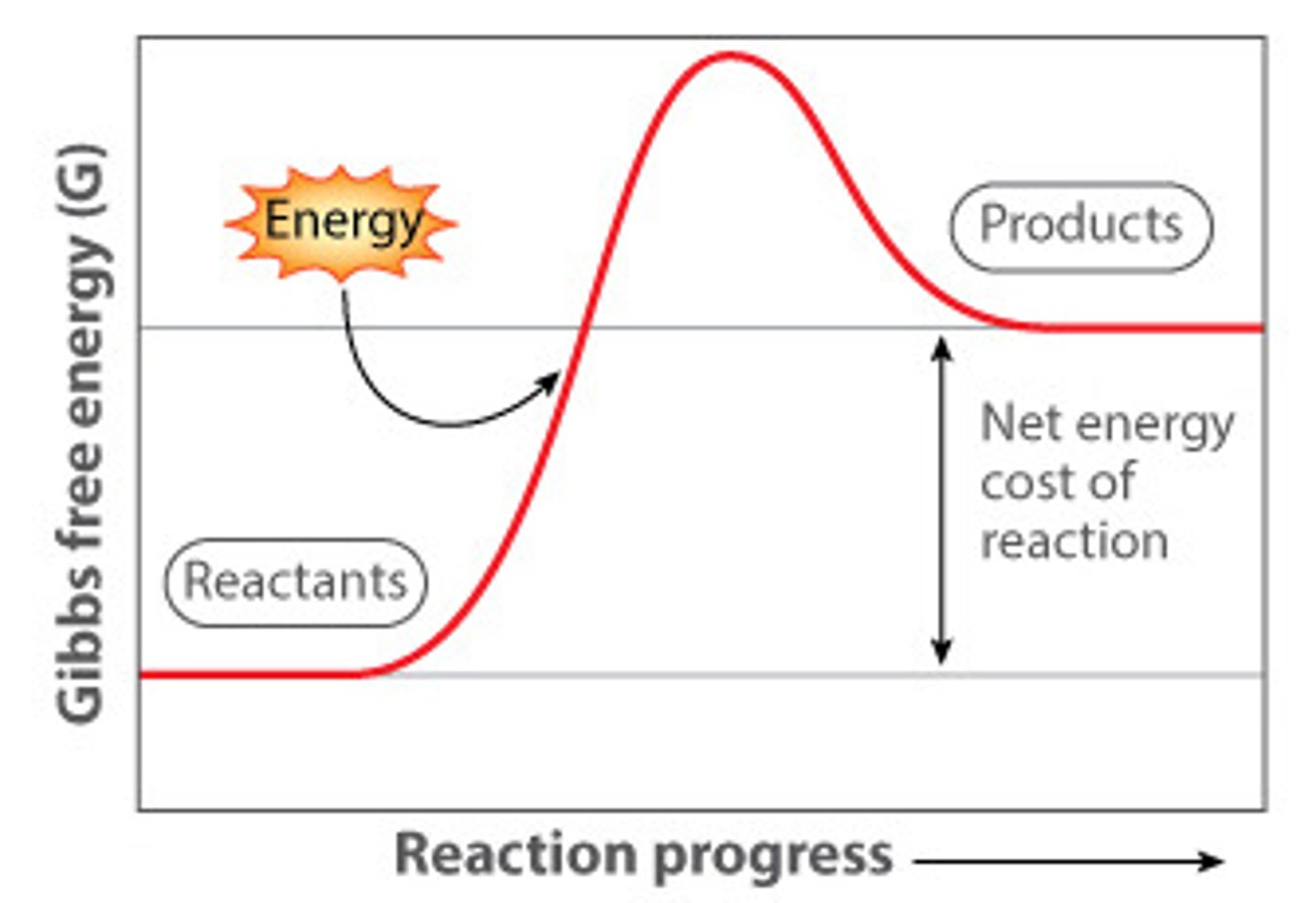
Exergonic Reaction
One that results in a decrease in free energy; another way of referring to a spontaneous reaction.
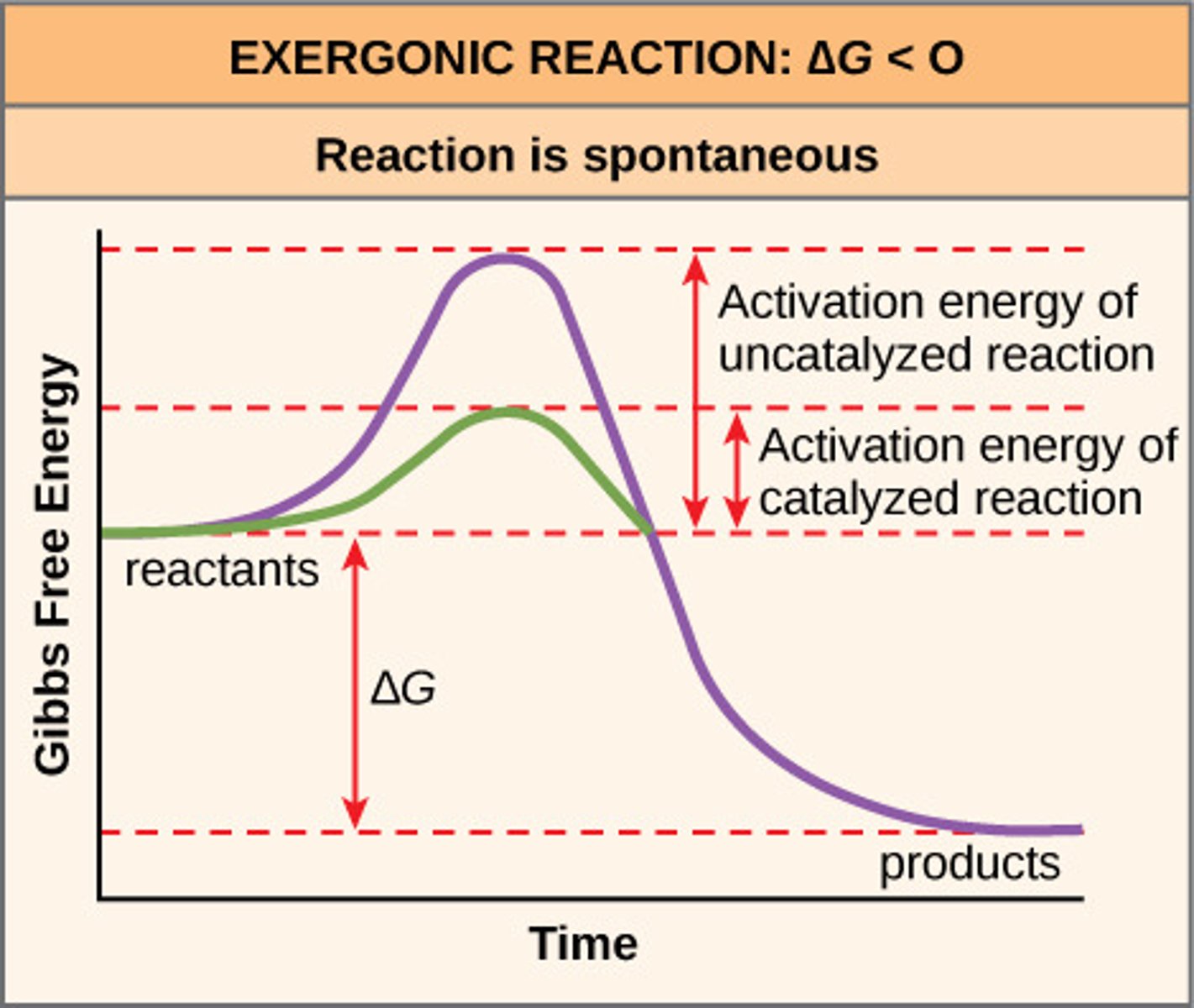
Activation Energy
The amount of energy required to get a chemical reaction through its transition state.
Transition State
During a chemical reaction, an intermediate state where old bonds are being broken but new bonds have not yet formed.
Enzyme
A protein that catalyzes a chemical reaction.
Active Site
The place on an enzyme (or ribozyme) where a reaction is catalyzed.
Redox (reduction-oxidation) reactions
Chemical reactions involving the gain (reduction) or loss (oxidation) of an electron.
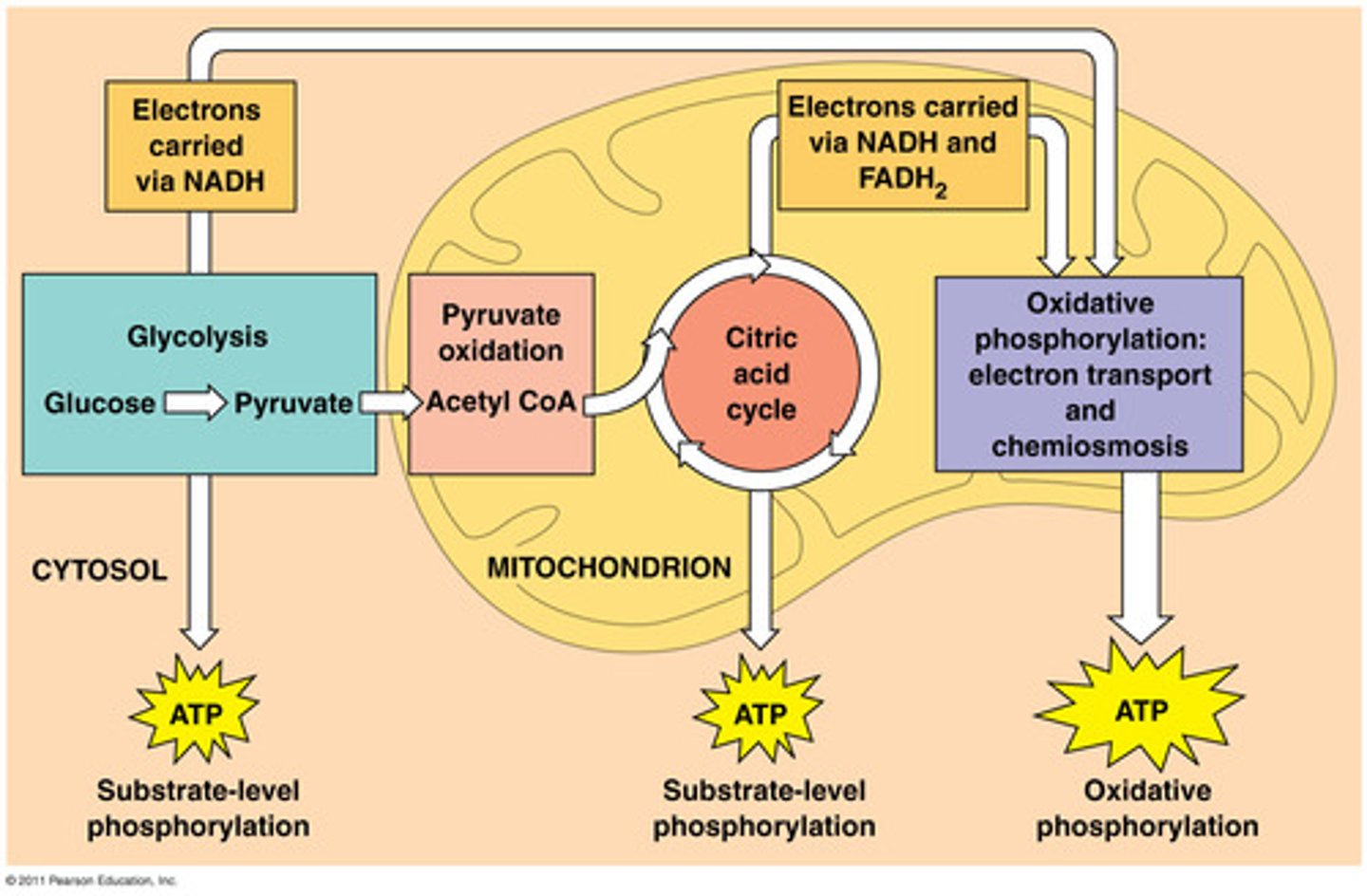
Cellular Respiration
A multi-step process that uses energy captured from carbon oxidation to power ATP production via an electron transport chain and ATP synthase.
Electron Transport Chain (ETC)
A series of machines that uses an electric current to pump protons across a membrane, establishing a proton gradient that is then used to generate ATP.
Inputs: NADH, FADH2, O2, H+
Outputs: NAD+, FAD, H20, Proton Gradient (low concentration in matrix, high in membrane)
-Gives up electrons (oxidation) to make pumps that push protons into inner membrane
- Oxygen allows process to continue. Its the final electron acceptor to create H2O
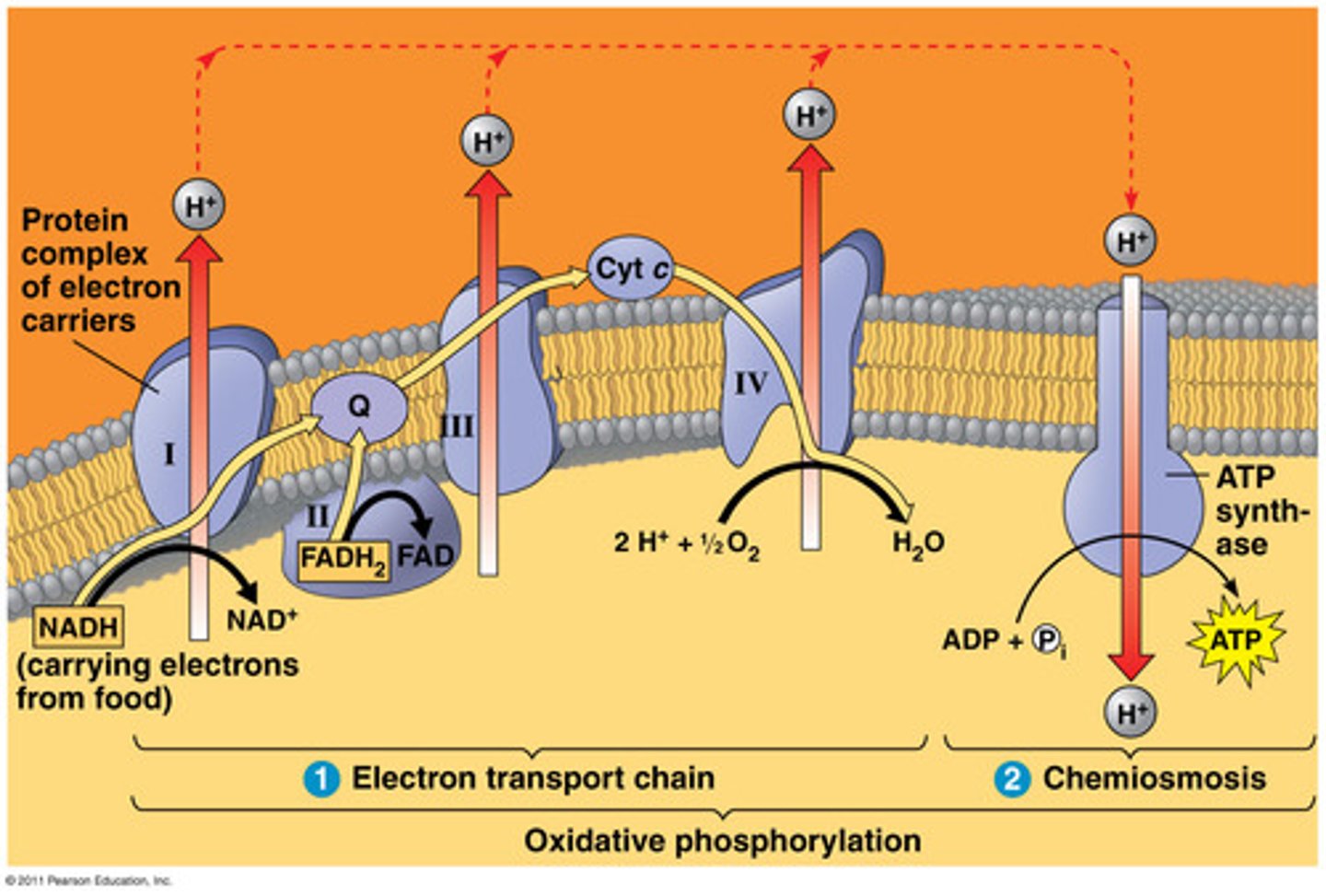
Oxidative Phosphorylation
ATP synthesized by oxidation of electron carries in the presence of O2. When the ETC uses oxygen as an electron acceptor, ATP production via the combined action of the ETC and ATP synthase.
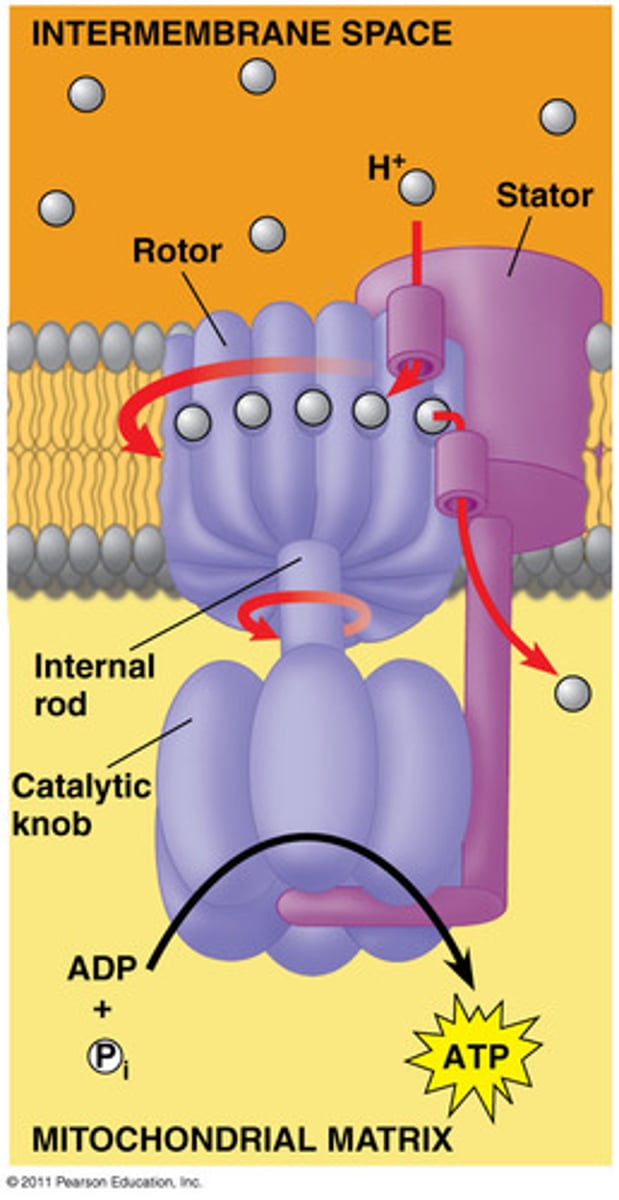
ATP Synthase
The multi-protein machine that transforms the kinetic energy in a flow of protons to mechanical energy that catalyzes the addition of a phosphate group to ADP to form ATP.
Inputs: 4H+ per ADP, ADP + Pi
Outputs: ATP (around 28 per glucose)
- move protons down the gradient from high to low concentration
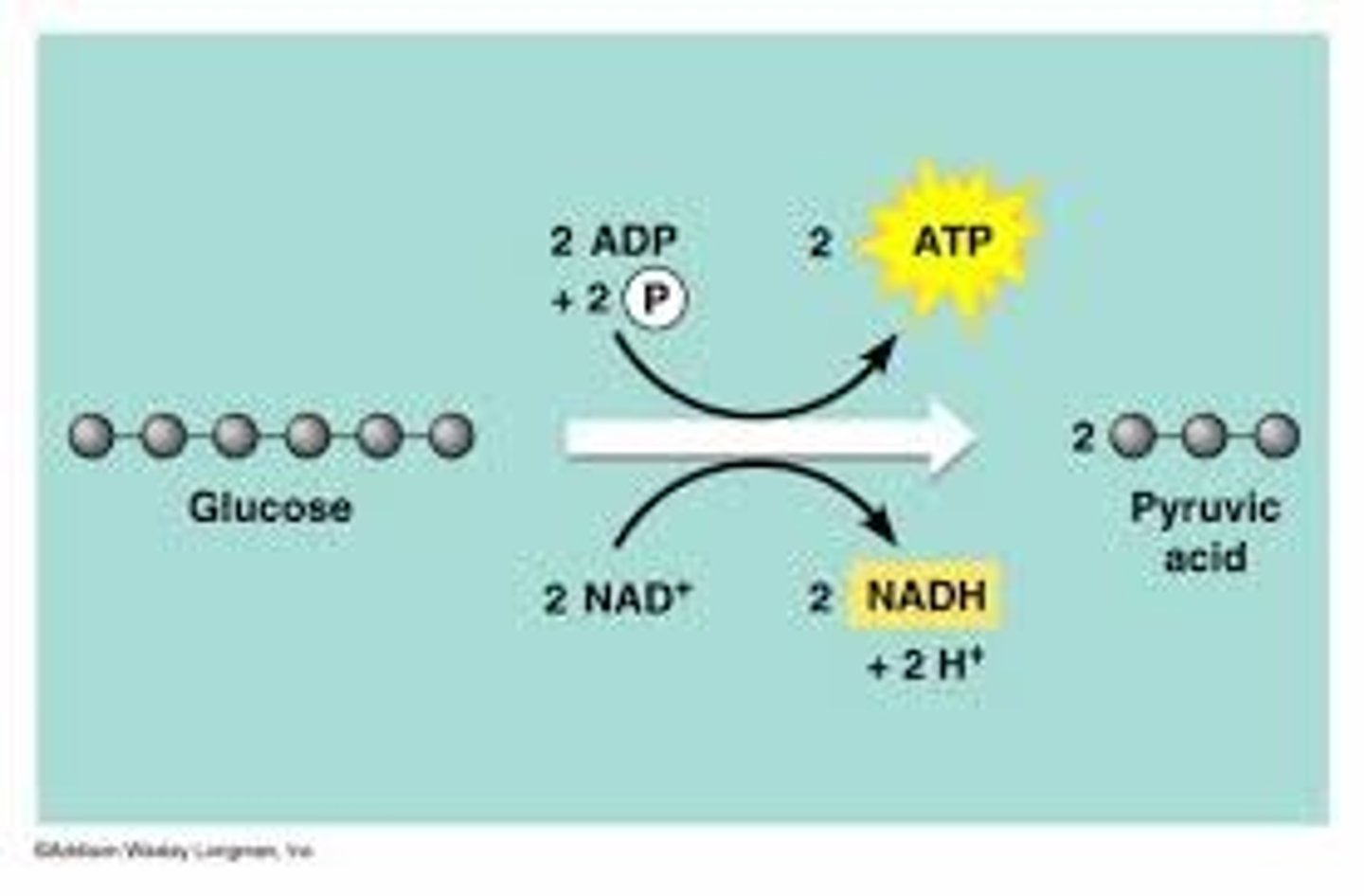
Glycolysis
Takes place in cytosol. A sequence of ten enzyme-catalyzed reactions that begins with glucose and ends with 2 pyruvate, producing 2 net ATP and 2 NADH per molecule of glucose.
Steps 1-5 require ATP (energy-investing reactions) gain
input: 1 glucose, 2 ATP, 2 NAD + Pi
Steps 6-10 yield NADH and ATP (energy-harvesting reactions) release
Outputs: 2 Pyruvate, 4 ATP (2 net), 2 NADH
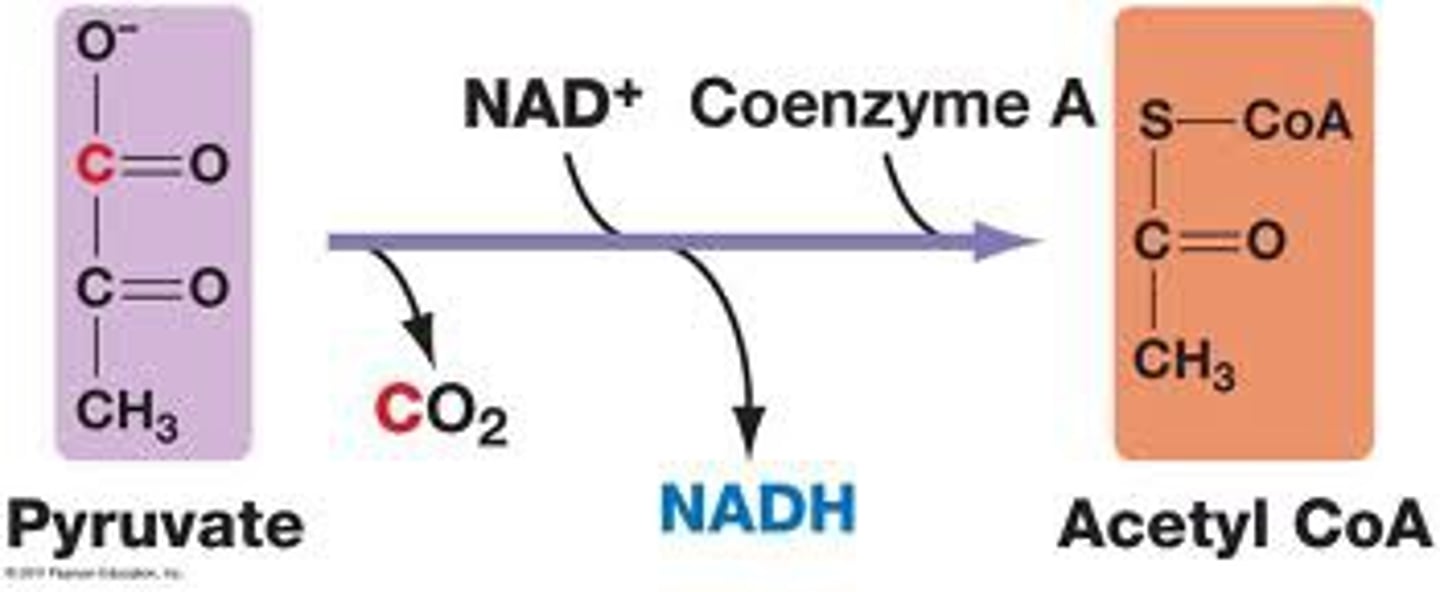
Pyruvate Processing
Pyruvate is transferred to the mitochondrial mix. A series of enzyme-catalyzed reactions that begins with pyruvate as a substrate and produces acetyl-CoA and NADH.
Inputs (per 1 glucose): 2 pyruvate, 2 NAD+, 2 coenzyme A - oxidizes Carbon in glucose and makes Co2
Outputs (per 1 glucose): 2 Acetyl CoA, 2 CO2, 2NADH
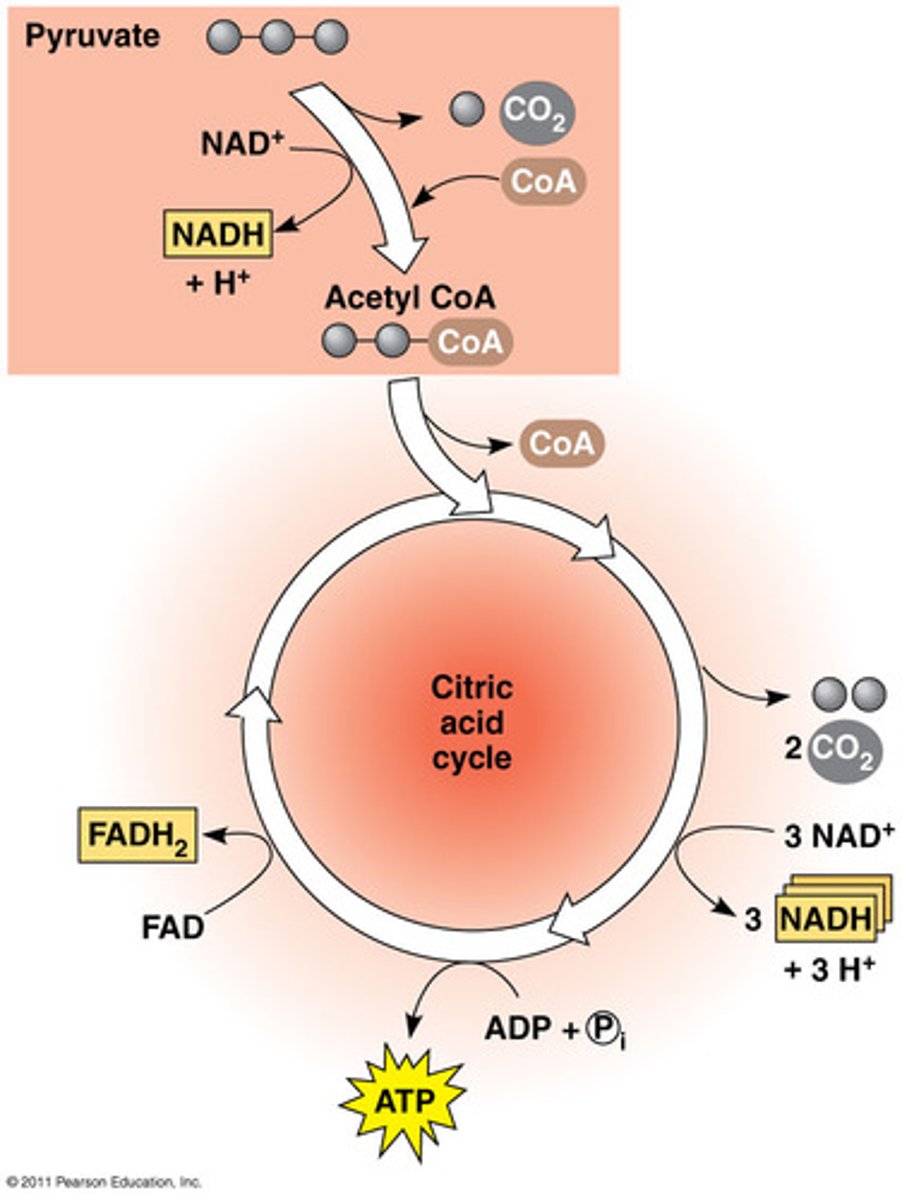
Citric Acid Cycle
A sequence of nine enzyme-catalyzed reactions that begins with acetyl-CoA, completes the oxidation of glucose to CO2, and produces ATP, NADH, and FADH2.
Step 1: Acetyl CoA reacts with Oxaloacerate (4 carbons) to create citrate (6 carbons) --- Coenzyme A is released (4 carbons) are supports
Step 3-4: acetyl group is oxidized to CO2, resulting in a 4 carbon molecule in the cycle
Step 8: malate is oxidized to regenerate oxaloacerate
Inputs (per 1 starting glucose): 2 Acetyl CoA, 6 NAD+, 2 FAD+, 2 GDP (like ADP) + Pi
Outputs (per 1 starting glucose): 2 GTP (like ATP they are interchangeable), 4 CO2, 6 NADH, 2 FADH2
NADH, FADH2, Q
Molecules that function as electron carriers during cellular respiration, meaning that they transport electrons to or within the electron transport chain.
Fermentation
A pathway that transfers electrons from NADH to a carbon-based molecule to regenerate NAD+ and keep glycolysis running to produce small amounts of ATP.
Aerobic Respiration
Cellular respiration that uses O2 to accept electrons from the electron transport chain and produces water as a byproduct.
ex: O2 + 4e- + 4H+ --> 2H20 -redox reaction
Anaerobic Respiration
Cellular respiration that uses any ion or molecule other than O2 to accept electrons from the electron transport chain.
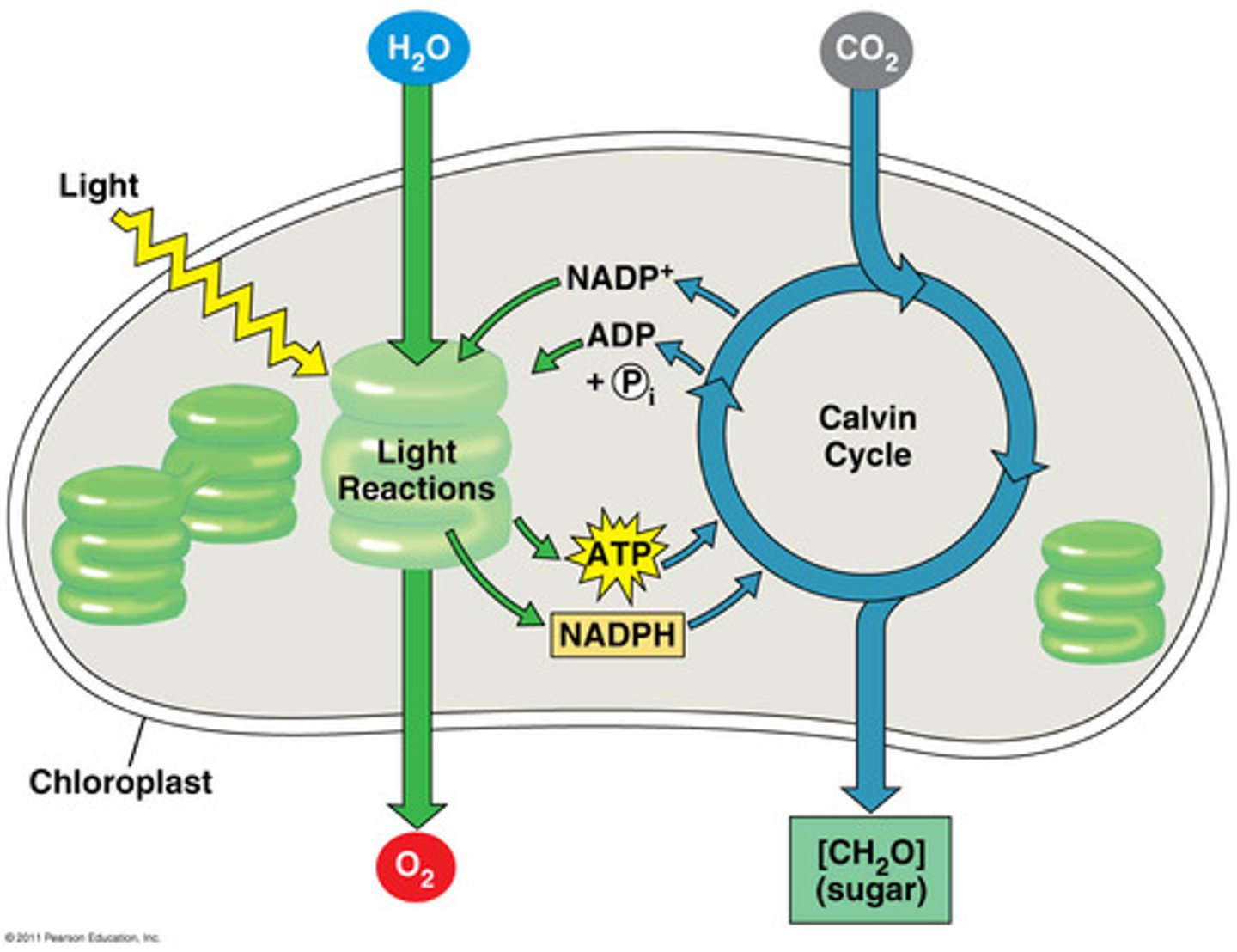
Photosynthesis
A process that transforms light energy into chemical energy — meaning the potential energy found in electrons that participate in covalent bonds, usually in sugars or other carbohydrates.
Pigment
A molecule that absorbs specific wavelengths of light. Pass energy from excited electrons until the excited electron arrives at a specific location where reduction - and thus energy transformation - takes place
Chlorophyll
The primary photosynthetic pigment in land plants and most algae.
Photosystem I (PSI)
A complex of molecular machines that receive low-energy electrons at the end of the electron transport chain, use the energy in sunlight to excite those electrons to a high-energy state, and pass them on to electron carriers that either feed the electron transport chain or an enzyme that catalyzes the reduction of NADP+ to NADPH.
Photosystem II (PSII)
A complex of molecular machines that acquire electrons by oxidizing water, use the energy in sunlight to excite those electrons to a high-energy state, and pass them on to an electron carrier that feeds the electron transport chain, leading to ATP production by ATP synthase.
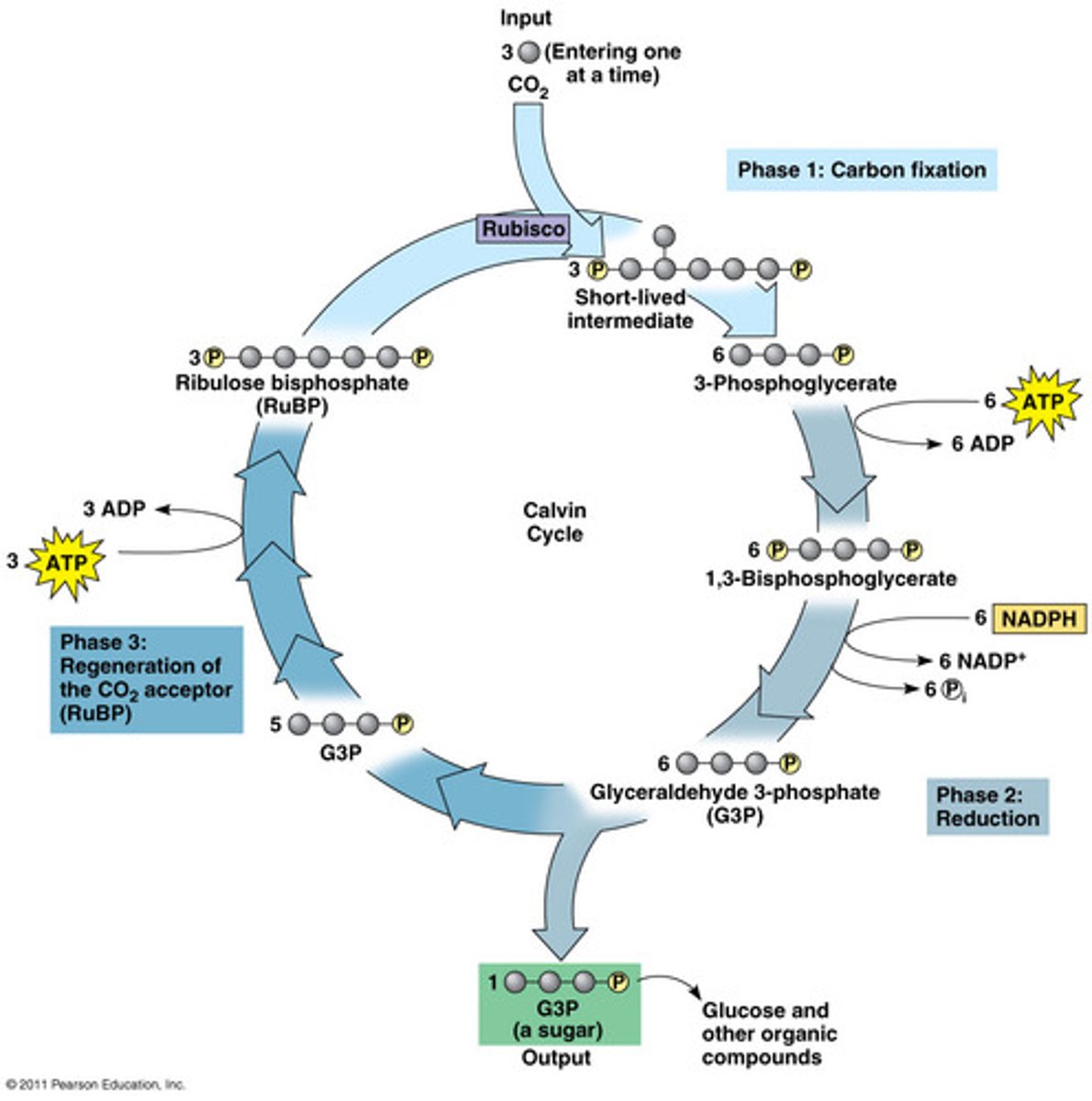
Calvin Cycle
A series of reactions that results in carbon from CO2 being "fixed," or reduced, and used to synthesize sugars.
Rubisco
The enzyme that catalyzes the reduction of CO2 and its incorporation into sugars. Most abundant and important enzyme. It is slow (only catalyzes 3 reactions per second) and when CO2 levels in the chloroplast are low, O2, instead of CO2 can bind to the active sites and be added to the 5-carbon sugar that acts a substrate. Can be summarized as an insufficient enzyme.
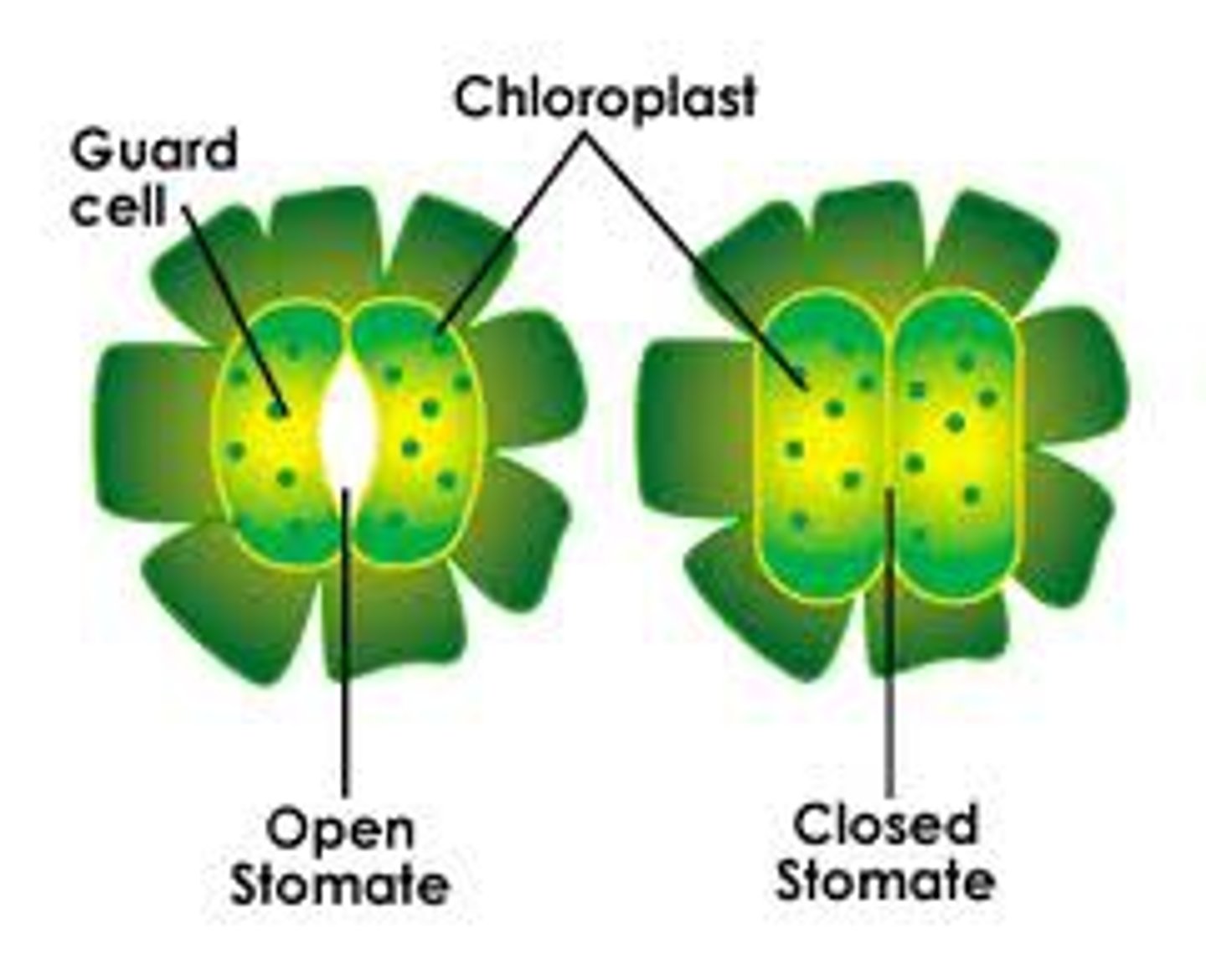
Stomata
Openings in stems and leaves that allow gas exchange via diffusion along their concentration gradients — most importantly, CO2 to enter and O2 to leave.
ATP
Source of energy that makes cells and organisms run
Phosphorylation
Provides the input of energy that makes nonspontaneous chemical reactions increase potential energy.
First Law of Thermodynamics
Energy is neither created nor destroyed but can be converted from one form to another.
Second Law of Thermodynamics
When energy is converted from one form to another, some of that energy becomes unavailable to do work. No energy transformation is 100% efficient - some energy is lost as heat
Energy
Capacity to do work. To produce a change, it must be transformed from one state to another or transferred from one location to another.
Change in free energy (G)
Difference in free energy between the products and the reactants
Anabolic Reactions
Complex molecules are made from simple molecules and energy input is required.
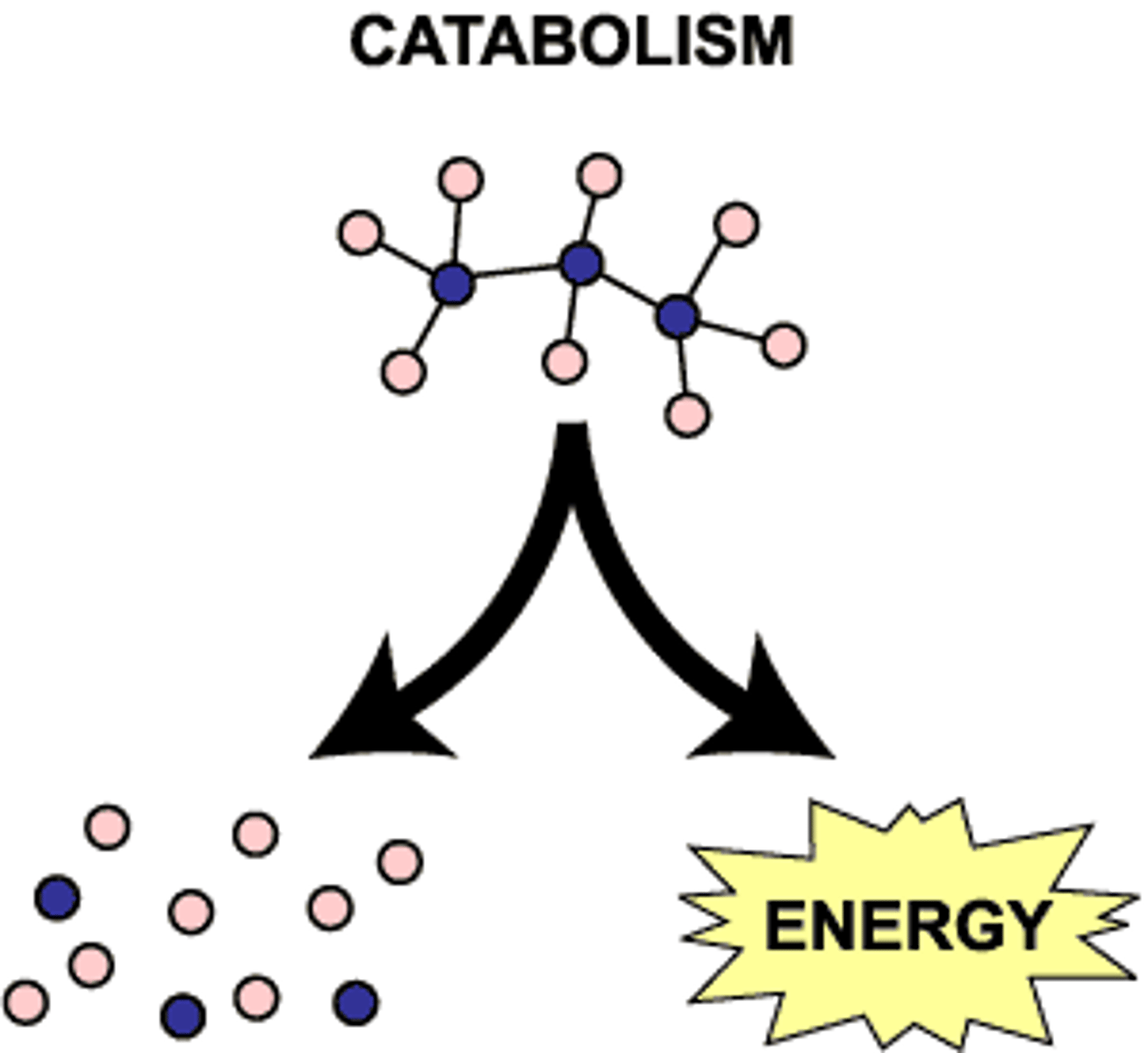
Catabolic Reactions
Complex molecules are broken down into simpler ones and energy is released.
Characteristics of Enzymes
1. lowers the activation energy, which speeds up the rate of reaction
2. they don't change reactions from spontaneous to nonspontaneous and vice versa
3. specific: bind only to their specific reactions
4. recycled: not altered during the reaction, they can be used again.
Enzyme-Substrate Complex (ES)
Held together by hydrogen bonds, electrical attraction, temporary covalent bonds, and van der waals
Substrate Binding
Bind to active sites by noncovalent interactions
Enzymes are controlled by:
1. Regulation of gene expression
2. regulation of enzyme activity
3. enzymes can be positively or negatively regulated
Inhibitors
slow or stop reaction rates
Active Site Inhibition (competitive inhibition)
prevents substrates from entering the active site (wall)

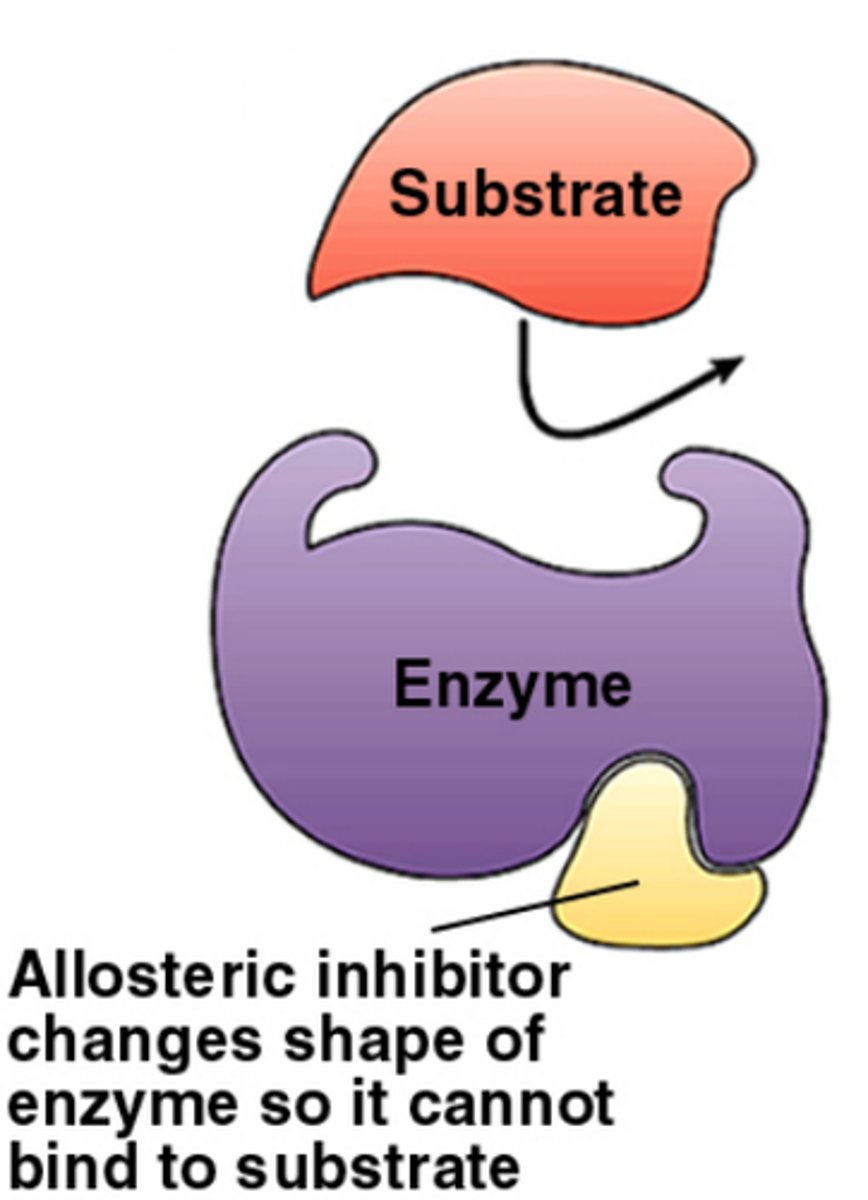
Allosteric Inhibition
Inhibitors bind elsewhere on enzyme and alters shape of active site, preventing substrate holding. Can be permanent or reversible.
Activators
Increase reaction rates
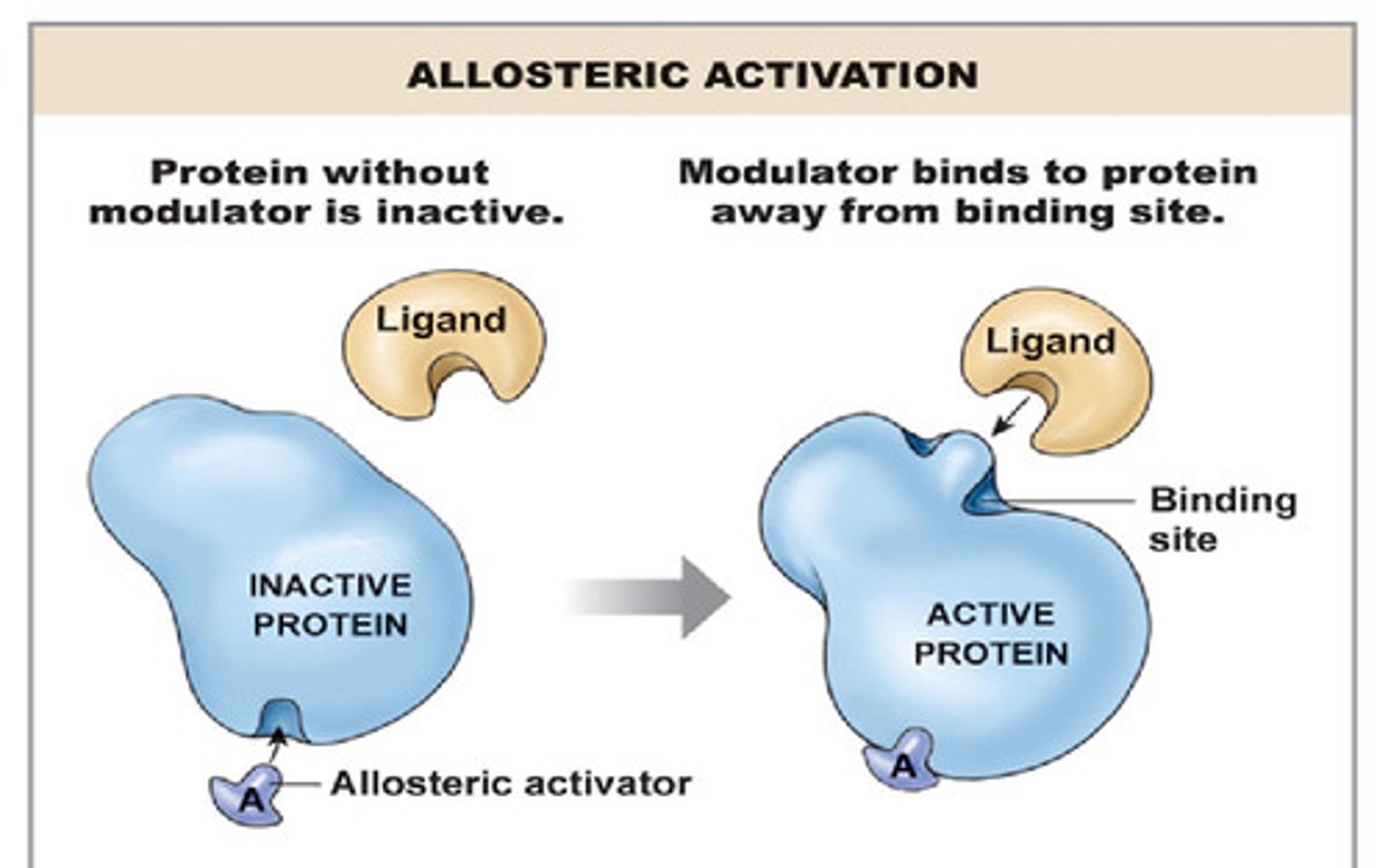
Allosteric Activation
Activator binds elsewhere on enzyme and altars shape of active site to encourage substrate binding.
Allostery
Other molecules can cause a conformational change in an enzyme which then alters its activity.
Cellular respiration involves 2 things:
1. a fundamental category of chemical reactions (reduction-oxidation reactions)
2. a fundamental principle about how the universe works (first law of thermodynamics)
-chemical reactions are about transferring energy, and the first law of thermodynamics is about transforming energy.
Oxidation
loss of electrons, almost always a spontaneous process
Reduction
Gain of electrons and is nonspontaneous. Electron is often accompanied by an H+ ion
Cellular Respiration Cycle
Glycolysis (cytoplasm), ------> (mitochondria) Pyruvate Processing, Citric Acid Cycle, Electron Transport Chain, and ATP Synthase
Fermentation
Occurs right during glycolysis if there is no Oxygen, increases fitness under certain environmental conditions but doesn't actually make ATP, and cellular respiration resumes once O2 is available as pyruvate can now be processed to feed the citric acid cycle and ETC.
Inputs and Outputs:
- 2 ATP created per glucose molecule
- 2 NAD+ is reduced to NADH in glycolysis
- NADH is oxidized back to NAD+ so glycolysis can continue bc NADH can't go into ETC
-Ethanol fermentation produces CO2
Main function of Oxygen in Respiration
Main function is to accept electrons released by glucose oxidation, forming H2O
Negative Feedback
High concentration of a metabolic product can INHIBIT action of an enzyme in the pathway
Positive Feedback
High concentration of a metabolic product can ACTIVATE an enzyme in another pathway, diverting raw materials away from synthesis of the first products
Glycolysis Control Points
- inhibited by excess ATP- blocking arrow
- inhibited by excess citrate (from citric acid cycle)
- activated by excess ADP
have a lot of energy= decrease cellular respiration
low energy = increase cellular respiration
Citric Acid Cycle Control Point
Step 1: inhibited by excess NADH and ATP
Step 3: inhibited by excess NADH and ATP; activated by excess NAD+ and ADP
Photosynthesis Reaction
CO2 + H2O + light energy ---> carbohydrate + O2
Visible Spectrum
Set of wavelengths that are visible to humans and ranges from shorter. Shorter wavelengths contain more energy than longer wavelengths.
Replacement Electrons
Essentially fuel the electrical circuit that runs photosynthesis-comes from water
PSII "splits" water
Pigment molecules become so oxidized that they pull electrons out of the O-H bonds in water, releasing protons (H+) and molecular oxygen (O2)
Photosynthesis Summary
1. Electrons are taken from water and excited to a high energy state when a pigment in PSII absorbs photons
2. The excited electrons are used to reduce an electron acceptor, then passed by PQ to ETC and that pumps photons
3. The resulting photon gradient drives productions of ATP by ATP synthase
Plant outer skin cells wax
Layer that blocks movement of water away from the plant and keeps stems and leaves from drying out. Also prevents movement of gases into or out of the plant.
Guard Cells
Open and close the pores in response to changes in environmental conditions. Regulates the size of the openings.
Photon
"Packet" of energy
When a photon hits a molecule:
Bounce off: scattered or reflected
Pass through: transmitted
Be absorbed: adding energy to the molecule (excited state)
Photosystem
A protein complex with a light absorbing pigments that absorb photons
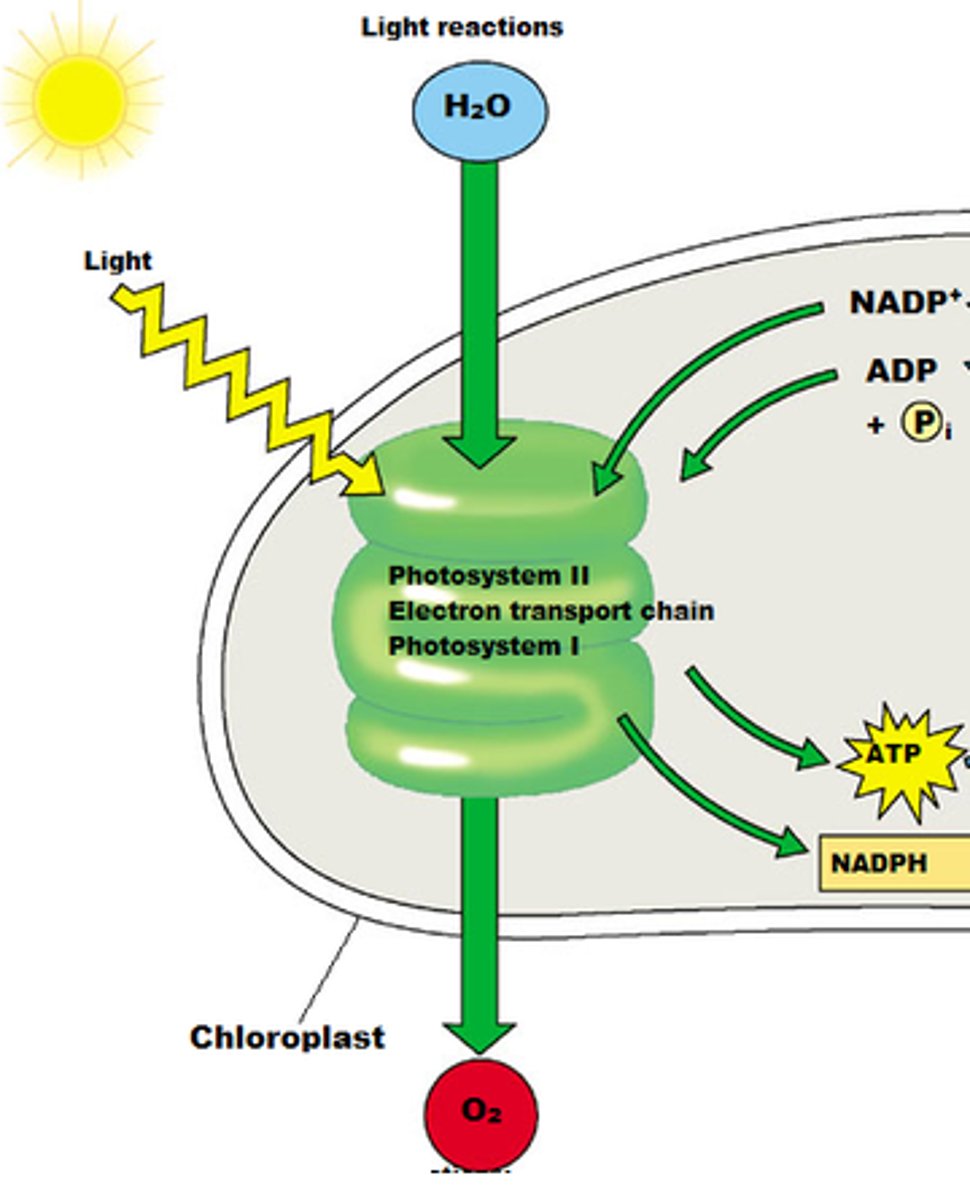
Light Reactions
Convert light to chemical energy (ATP and NADPH). Energy capture and electron transport.
1) light energy is absorbed
2) electrons passed through ETC
-proton gradient; ATP synthase then uses proton gradient to phosphorylate ADP
- electron carrier is the final electron acceptor (NADP+ --> NADPH)
Inputs (ETC):
Photons (from light), H2O, NADP+, ADP + Pi
Outputs:
O2 (and H+), proton gradient, NADPH, ATP
-protons move from high concentration thylakoid space down the gradient through ATP Synthase
-capturing energy to move it to Calvin Cycle
Carbon Fixation
CO2 is reduced to carbohydrates, occurs in stroma, and energy needed is ATP and NADPH.
Chemical energy is converted to energy stored in carbohydrates
Calvin Cycle P1: Fixation
CO2 fixed by rubisco into RuBP to form 3-PGA
- By "fixing" rubisco makes sure carbon stays in the plant
Calvin Cycle P2: Reduction
3-PGA is reduced to GBP, uses ATP and NADPH, (NADPH oxidized to NADP+)
Calvin Cycle P3: Regeneration
Some G3P is used to regenerate RuBP for the next turn of the cycle, uses ATP
Inputs: no light required, CO2, ATP, NADPH
Outputs: G3P (to make glucose and other compounds), ADP, Pi
Carbohydrates
Provide the glucose required to fuel cellular respiration and produce the ATP needed to keep plant cells alive and thriving.
Cellular respiration only
Glycolysis, pyruvate processing, citric acid cycle
- overall is a transfer of chemical energy from glucose to ATP
Photosynthesis only
Calvin Cycle, rubisco, photosystems containing the pigment chlorophyll
- overall is about transforming light energy to chemical energy
Both CR and Photosynthesis
ATP synthase, and an electron transport chain (not the same molecules but pass electrons through redox reactions)
Cellular respiration only
-Carbon atoms are oxidized
-Oxygen atoms are reduced
Photosynthesis only
-Carbon atoms are reduced
-Oxygen atoms are oxidized
Both CR and Photosynthesis
-Electron carriers are reduced
-Electron carriers are oxidized
Cellular respiration only
Chemical energy in the forms of C-C and C-H bonds in carbohydrates is transferred to chemical energy in ATP.
Photosynthesis only
Light energy is transformed into chemical energy on the form of C-C and C-H bonds in carbohydrates
Both CR and Photosynthesis
Mechanical energy in the form of ATP Synthase spinning is transformed to chemical energy by addition of a phosphate group to ADP to form ATP
Both CR and Photosynthesis
Chemical energy in the form of electrons being passed through an ETC is transformed to mechanical energy as proton pumps in the ETC create a proton gradient
Both CR and Photosynthesis
Electrical energy in the form of protons flowing down an electrochemical gradient is transformed to mechanical energy as ATP synthase spins
Gluscose
Main input of cellular respiration
Catabolic interconversions
ATP is synthesized using chemical bond energy from macromolecules
Anabolic interconversions
Macromolecules are synthesized from their monomers using condensation reactions. Energy input: ATP or NADH