2.2 nomenclature and isomerism in organic compounds
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Homologous series
Same general formula
Similar chemical properties
Successive members differ by CH2
Alkanes General formula, functional group example
CnH2n+2 -ane
C-H e.g ethane C2H6
Alkenes General formula, functional group example
CnH2n
-ene C= C e.g ethene C
Alchoho’s General formula, functional group example
CnH2n+1OH
-ol , hydroxyl-
-OH E.g ethanol or hydroxyethane CH3CH2OH
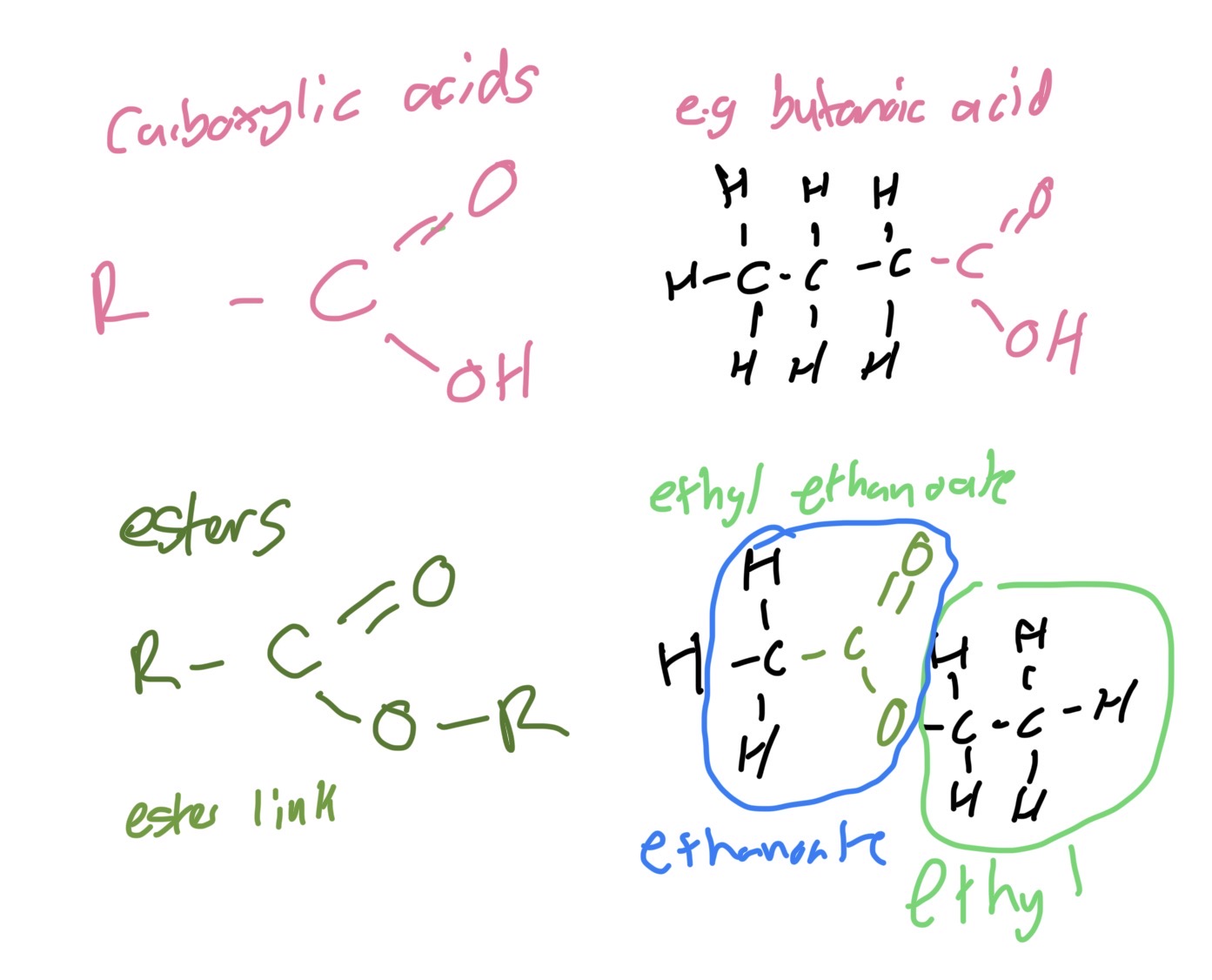
Carboxylic acid General formula, functional group example
CnH2n+1COOH
oic acid H-C=O
E.g ethanoic acid CH3COOH
Haloalkanes , functional group example
Halo- -
Cl - Br - I
e.g chloroethane CH3CH2Cl
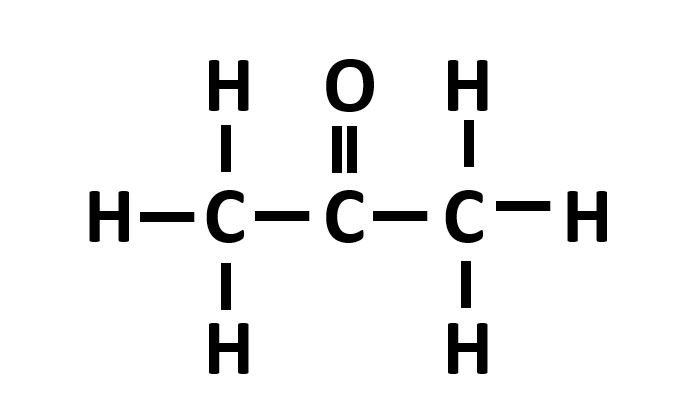
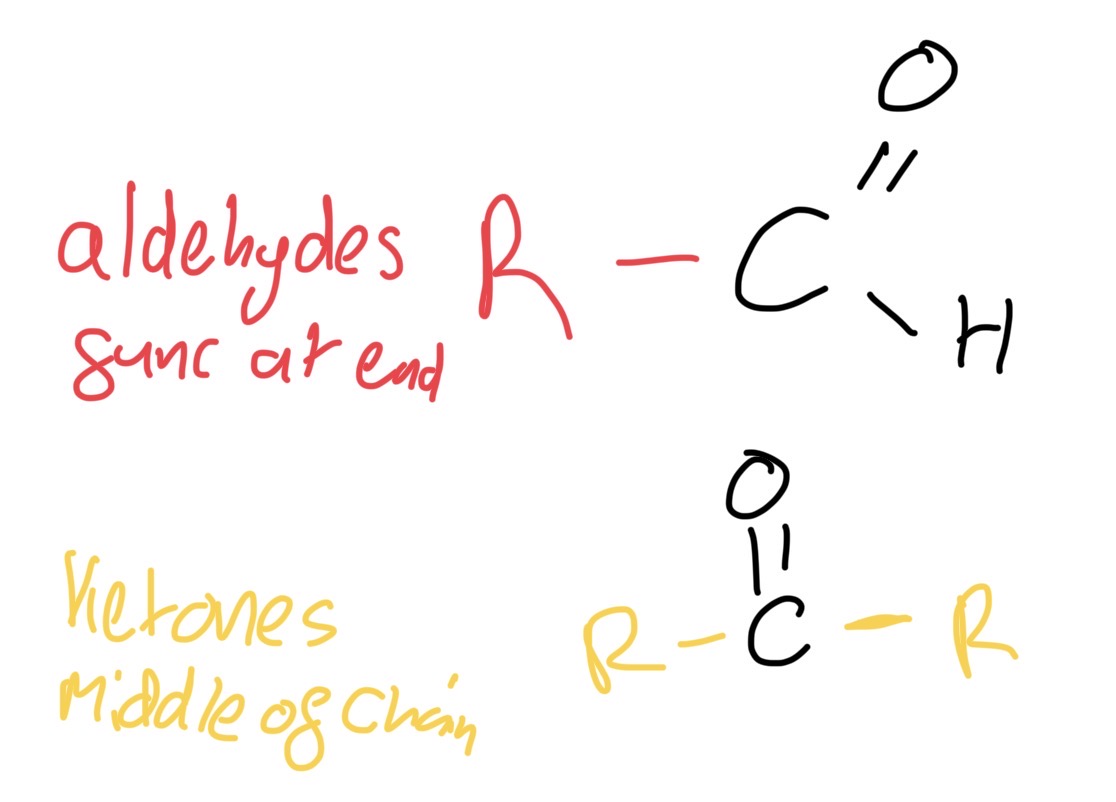
Aldehydes functional group example
-al. - H-C=O
E.G ethanal CH3COH3
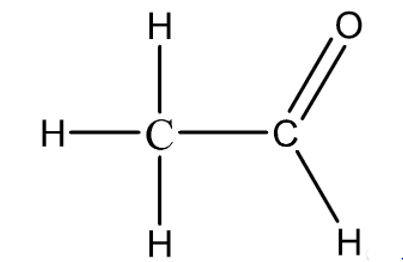
Ketones functional group example
-one C=O
E.g propanone CH3COCH3
1-10 prefix’s
1- Meth. My
Eth- Evil
Prop- parents
But- buy
Pent- parents
Hex- horribly
Hept-
Oct-
Non-
Dec-
Molecular formula definition
The actual number of atoms of each element in a molecules
E.g ethane C2H6, ethanol C2H6O, ethanoic acid C2H3O2
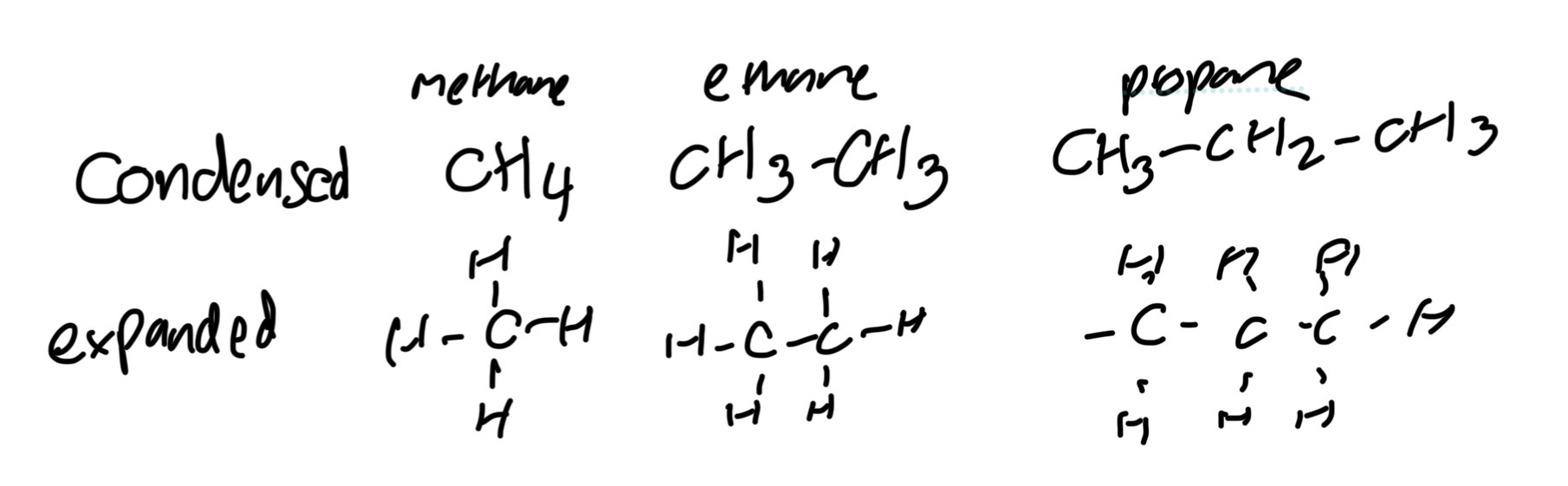
Structural formula def
Shows all atoms and covalent bonds
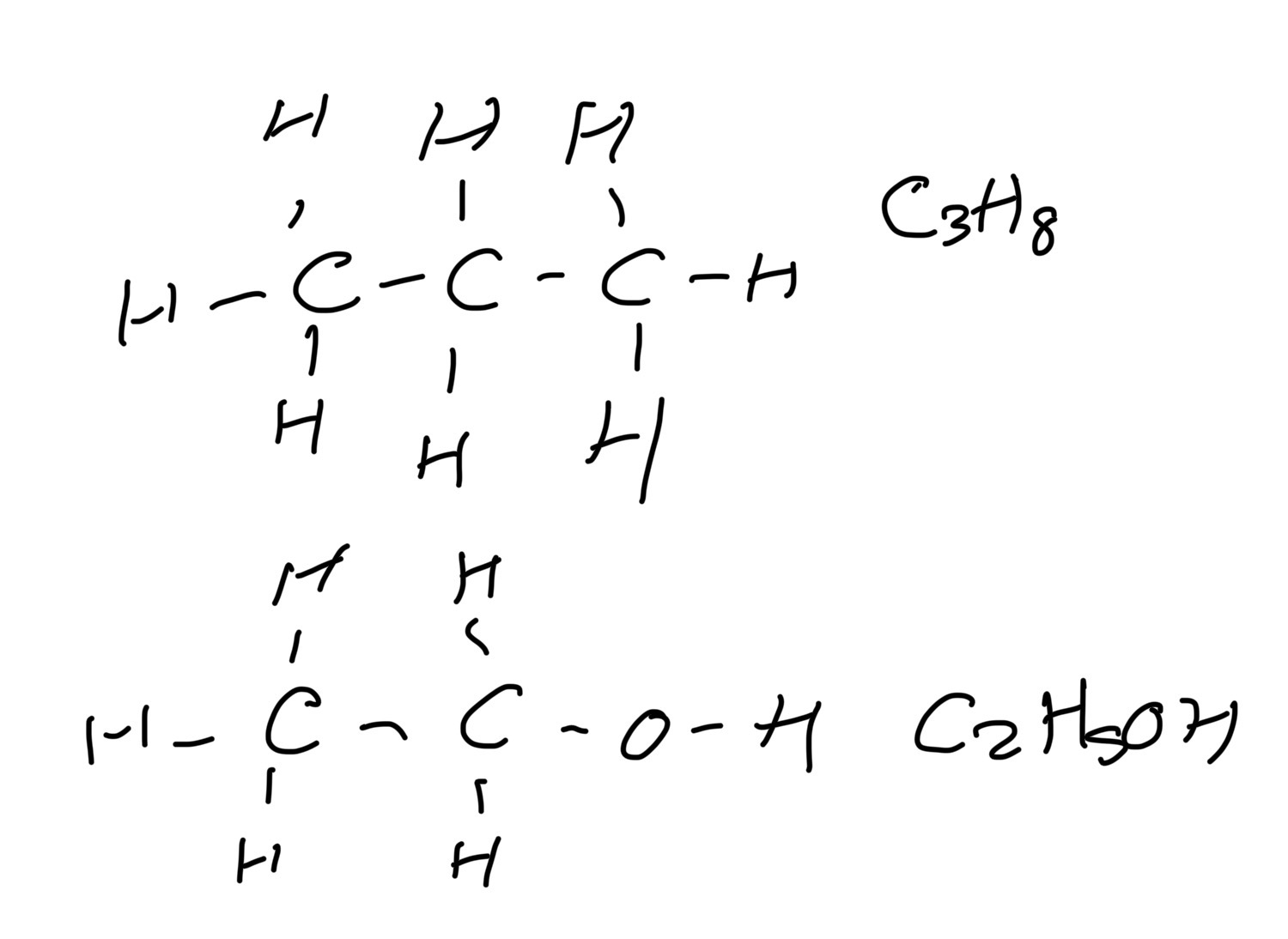
Condensed structural formula
- arrangement of atoms and groups at each carbon atom, (brackets if two different groups attached.
Empirical formula
The simplest ratio of the atoms of the different elements in an organic compound
E.g ethanoic acid CH3COOH - CH2O
Skeletal formula
Carbon structure zig zag - omitting hydrogen atoms
Rules naming compounds
longest continuous carbon chain prefix
No methyl groups on end or start of chain
C atoms number in lowest number possible
Two substituent given prefix- di-, tri-, tetra-
Tw diff substituents given in alphabetical order e.g 2,4- dichloro-3-methylpentane
Substituent group names
-CH3- methyl, C2H5- ethyl, C37 propyl, C4H9 butyl
-F Fluor CL chloro, -Br broom, I- Iodo
Isomer definition
Isomers are molecules with the same molecular formula but different arrangement of atoms
Structural isomerism definition
Molecules which have the same molecular formula but a different structural formula
Chain isomerism
Same molecular formula - diff arrangement of atoms in carbon chain e.g butane , 2-methylpropne
E.g C5H12 pentane, 2-methylbutane, 2,2-dimethylpropane
Position isomerism
Isomers compounds in which same functional group but different arrangement positions in carbon chain
Propan-1-ol propan-2-ol
Functional group isomerism definition
Molecules same molecular formula but different functional group
Functional group isomerism Aldehydes and ketones
Aldehydes - func group at end
Ketones - func in middle
Functional group isomerism carboxylic acids and esters
What are stereoisomerism
Molecules which have the same molecular and structural formula but different 3D spatial arrangements.
Geometric isomerism
Geometric isomerism - compounds have the same structural formula but different arrangements of atoms due to the presence of one or more C=C bonds.
What causes geometric isomerism
C=C bond energy barrier to free rotation so restricted rotation around carbon carbon double bonds

Why does 3-methylpent-2-ene exists as geometric isomerism,
Restricted rotation around C=C
Both carbons in C=C have 2 different atoms/groups attached
Two types of geometric. Isomerism
Cis-trans isomerism
E-Z isomerism
When is cis-trans notation used.
Both carbons of C=C bonds have hydrogen atom and non-hydrogen atoms directly attached to them
Cisn and trans isomers. definitions
CIS - HYDROGEN SAME SIDE. OF C=C
TRANS- HYDROGEN OPPOSITE-SIDES OF C=C
E-Z isomerism
Assigning priority , both substituents of higher. Priority same side = Z while opposite is E
Assign- atomic number higher priority, next atom or sequence of atoms in subsequent group