Definitions of endothermic and exothermic
0.0(0)
Studied by 0 peopleCard Sorting
1/16
There's no tags or description
Looks like no tags are added yet.
Last updated 12:59 AM on 2/20/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
1
New cards
reactants + energy = products
endothermic
2
New cards
ΔH > 0
endothermic
3
New cards
energy is transferred INTO the system
endothermic
4
New cards
ΔPE of the system > 0
endothermic
5
New cards
ΔPE of the surroundings < 0
endothermic
6
New cards
bonds are broken
endothermic
7
New cards
net energy of bonds formed is WEAKER than net energy of bonds broken
endothermic
8
New cards
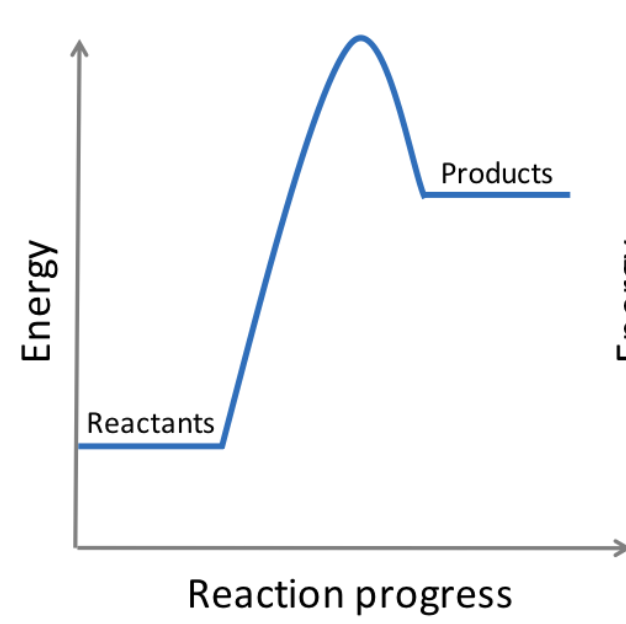
endothermic
9
New cards
combustion
exothermic
10
New cards
reactants = products + energy
exothermic
11
New cards
ΔH < 0
exothermic
12
New cards
energy is transferred OUT of the system
exothermic
13
New cards
ΔPE of the system < 0
exothermic
14
New cards
ΔPE of the surroundings > 0
exothermic
15
New cards
bonds are being formed
exothermicn
16
New cards
net energy of bonds formed is GREATER than net energy of bonds broken
exothermic
17
New cards
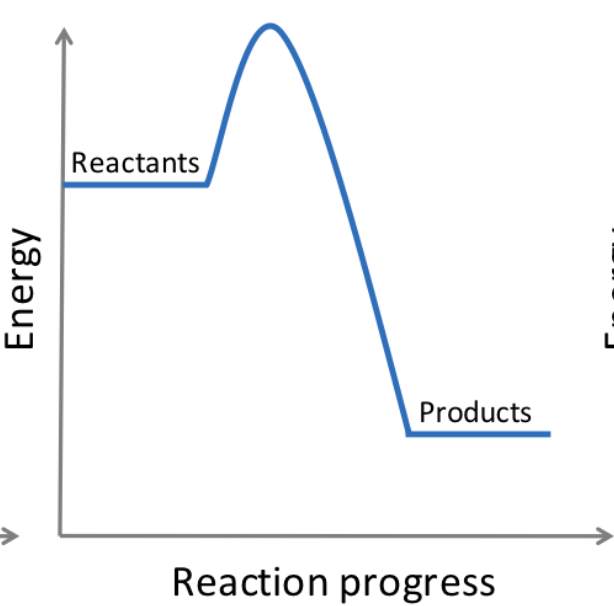
exothermic