OCR A Chemistry A Level Paper 1
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
Amount of substance equations
mass = mr x moles
mol = vol x conc
vol(gas) = mol x 24 (dm)
PV=nRT
(pressure, Pa, volume, m3, moles, -, 8.314, temp, k)
Empirical formula
The simplest whole number ratio of atoms of each element in a compound
Molecular formula
The number and type of atoms of each element in a molecule
Anhydrous
Contains no water molecules
Hydrated
Crystalline compound containing water molecules
Celsius to Kelvin
K=C+273
% yield and atom economy
% yield = (actual/ theoretical) x 100
atom economy = (desired products/all reactants) x 100
Cu2+ in aq solution
[Cu(H2O)6]2+
Pale blue
Fe2+ in aq solution
[Fe(H2O)6]2+
Pale green
Fe3+ in aq solution
[Fe(H2O)6]3+
Yellow
Mn2+ in aq solution
[Mn(H2O)6]2+
Pale pink
Cr3+ in aq solution
[Cr(H2O)6]3+
Green
Cu2+ and NaOH (aq)
Initially:
[Cu(OH)2(H2O)4] (s)
Blue precipitate
Excess:
No change
Fe2+ and NaOH (aq)
Initially:
[Fe(OH)2(H2O)4] (s)
Green precipitate
Excess:
No change
Fe3+ and NaOH (aq)
Initially:
[Fe(OH)3(H2O)3] (s)
Orange precipitate
Excess:
No change
Mn2+ and NaOH (aq)
Initially:
[Mn(OH)2(H2O)4] (s)
Pink precipitate
Excess:
No change
Cr3+ and NaOH (aq)
Initially:
[Cr(OH)3(H2O)3] (s)
Grey-green precipitate
Excess:
[Cr(OH)6]3- (aq)
Dark Green
Cu2+ and NH3 (aq)
Initially:
[Cu(OH)2(H2O)4] (s)
Blue precipitate
Excess:
[Cu(NH3)4(H2O)2]2+ (aq)
Dark blue
Fe2+ and NH3 (aq)
Initially:
[Fe(OH)2(H2O)4] (s)
Green precipitate
Excess:
No change
Fe3+ and NH3 (aq)
Initially:
[Fe(OH)3(H2O)3] (s)
Orange precipitate
Excess:
No change
Mn2+ and NH3 (aq)
Initially:
[Mn(OH)2(H2O)4] (s)
Pink precipitate
Excess:
No change
Cr3+ and NH3 (aq)
Initially:
[Cr(OH)3(H2O)3] (s)
Grey-green precipitate
Excess:
[Cr(NH3)6]3+ (aq)
Purple
Cu2+ and Cl-
[CuCl-(4)]2-
yellow
![<p>[CuCl-(4)]2-</p><p>yellow</p>](https://knowt-user-attachments.s3.amazonaws.com/c9177298-3934-45ce-b4c5-ab615df7d821.png)
Cu2+ and iodide
off-white copper (I) iodide precipitate
Transition element
A d-block element that can form at least one stable ion with a partially filled d-orbital
have similar physical properties (high densities + mp)
have variable oxidation states cause 4s and 3d orbitals are very close in energy levels
Cu and Cr are exceptions to rule that 4s subshell is filled before 3d subshell
Complex ion
a metal ion which is surrounded by coordinately bonded ligands
Ligand
an atom/ion/molecule that donates a pair of electrons to a central metal ion
Monodentate ligands
Form one coordinate bond to a metal ion
e.g. H2O, NH3, Cl-, CN-
Multidentate ligands
Form more than one coordinate bond each
Bidentate= two
Octahedral
6 ligands
e.g. [Mn(H2O)6]2+
![<p>6 ligands</p><p>e.g. [Mn(H2O)6]2+</p>](https://knowt-user-attachments.s3.amazonaws.com/6c6adfb8-f522-46aa-bf79-67820089f07e.jpg)
Tetrahedral
4 ligands
e.g. [CuCL4]2-
![<p>4 ligands</p><p>e.g. [CuCL4]2-</p>](https://knowt-user-attachments.s3.amazonaws.com/0ada50b2-e4a1-4731-8b6c-f5c7cf07b089.jpg)
Cis-platin
[Pt(NH3)2Cl2]
used in cancer treatment (chemo)
- loses the 2 coordinate cl- ions and coordinating with 2 Nitrogen atoms in a DNA strand
-prevents dna from replicating and cell is unable to divide
![<p>[Pt(NH3)2Cl2]</p><p>used in cancer treatment (chemo)</p><p>- loses the 2 coordinate cl- ions and coordinating with 2 Nitrogen atoms in a DNA strand</p><p>-prevents dna from replicating and cell is unable to divide</p>](https://knowt-user-attachments.s3.amazonaws.com/6f6e3149-a85e-46c7-b4cb-cfdf65eaeb00.png)
Bronsted-Lowry acid
proton donor
e.g. HCl, H2SO4, H3PO4
Bronsted-Lowry base
proton acceptor
e.g. NaOH, NH3, KOH
Amphoteric
a substance that can act as both an acid and a base
acid + metal
acid + metal -> salt + hydrogen
e.g. 2H+ + Mg --> Mg2+ + H2
acid + carbonate
acid + carbonate -> salt + water + carbon dioxide
e.g. 2H+ + CuCO3 --> Cu2+ + H2O + CO2
acid + base
acid + base -> salt + water
base is solid metal oxide or hydroxide
e.g. 2H+ + MgO --> Mg2+ + H2O
Sorensen's pH scale
pH = -log[H+]
[H+] = 10^(-pH)
- low value of [H+] = high pH value
- high value of [H+] = low pH value
Strong acid
completely dissociates in a solution
HA (aq) --> H+ + A-
∴ [H+] = [HA]
Ka
Ka = [H+][A-]/[HA]
-changes with temp (values usually standardised at 298K)
Larger Ka = eqm is further right = greater acid strength
Weak acid
partially dissociates in solution
Ka for weak acids
Ka=[H+]^2/[HA]
Approximations:
1) [H+]eqm = [A-]eqm
dissociation of H+ in water will be small so neglected
approcimation breaks down for very weak/dilute acids (pH>6)
2)[HA]start > [H+]eqm
therefore [HA]eqm = [HA]start
dissociation of weak acids is small, conc. of acid is much greater than conc. of H+ ions at eqm
not justified for stronger weak/dilute acids (Ka> 10^-2))
Ionic product of water
Kw = [H+][OH−]
Strong base
e.g. NaOH --> Na+ + OH-
[OH-] = [NaOH]
Weak base
pOH = -log[OH-]
[OH-] = 10^(-pOH)
pH + pOH = 14
Buffer solution
A system that minimises pH changes when small amounts of an acid or a base are added
Contains weak acid and conjugate base
Methods for preparing weak acid buffer solutions
1) CH3COOH(aq) --> H+(aq) + CH3COO-(aq)
CH3COONa(s) + aq --> CH3COO-(aq) + Na+(aq)
2) partial neutralisation of a weak acid
Excess CH3COOH(aq) + NaOH(aq) --> CH3COONa(s) +H2O(l)
How buffers work: Ha --> H+ + A- (reversible)
Adding small amount of acid, H+ :
H+ conc increases
H+ reacts w/ conjugate base A-
eqm shifts left
reduces H+ conc, pH maintained
Adding small amount of alkali, A- :
OH- conc increases
OH- reacts with H+ to form water
HA dissociates to form more H+ ions
eqm shifts right
increases H+ conc, pH maintained
pH titration curve
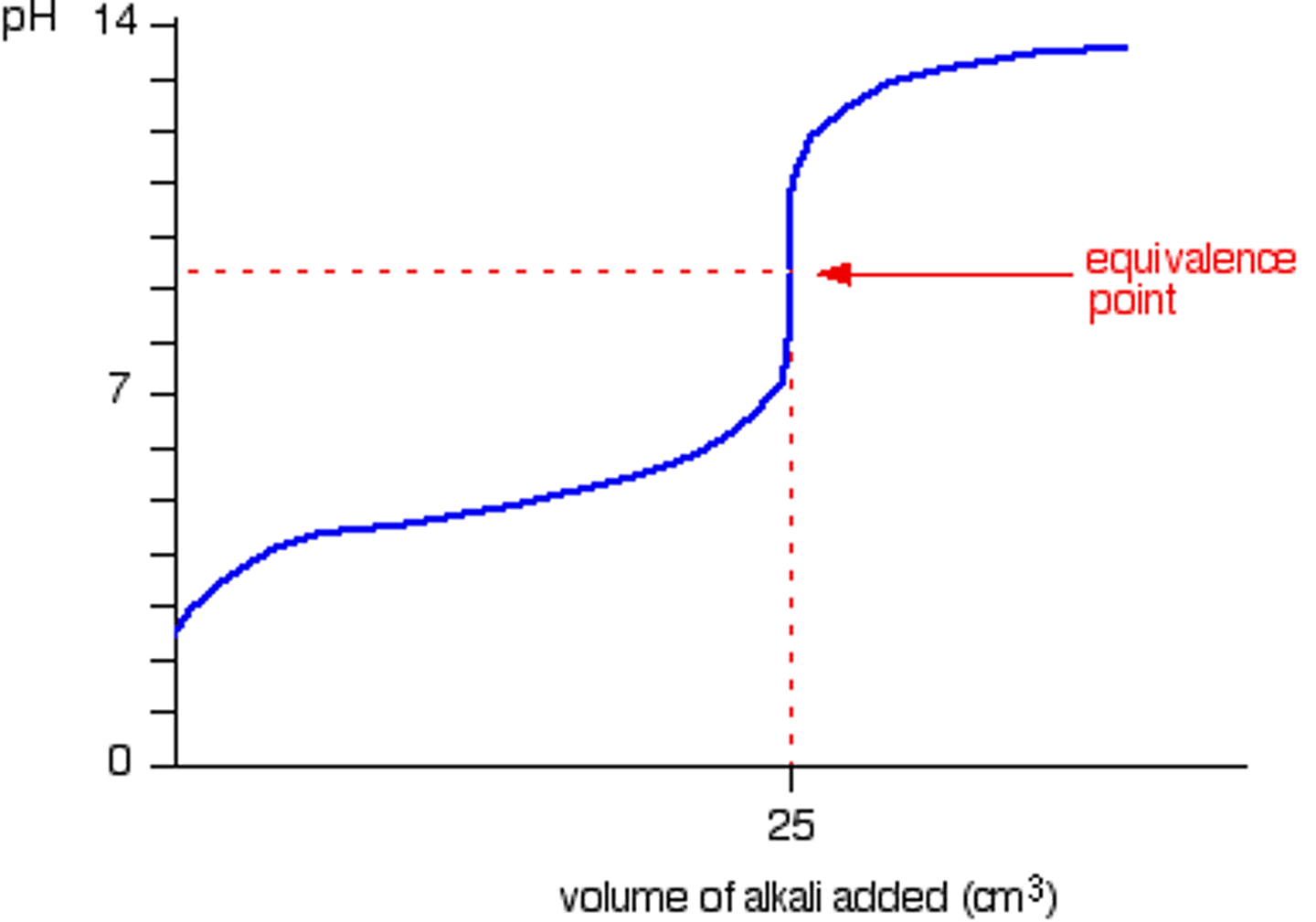
Periodicity
repeating pattern of trends in physical and chemical properties (across period)
Mendeleev's arrangement
elements arranged according to:
-relative atomic mass
-properties
-left gaps for undiscovered elements
-predicted properties of elements that would fill gaps
Arrangment now
elements arranged according to:
-increasing proton number
-elements in period show repeating trends in physical + chemical properties
-elements in groups show repeating/similar chemical properties
Atomic number
number of protons in the nucleus of an atom
Comparing Beryllium and Boron
2p subshell in boron has higher energy that 2s subshell in beryllium
2p electron easier to remove
1st I.E lower for boron
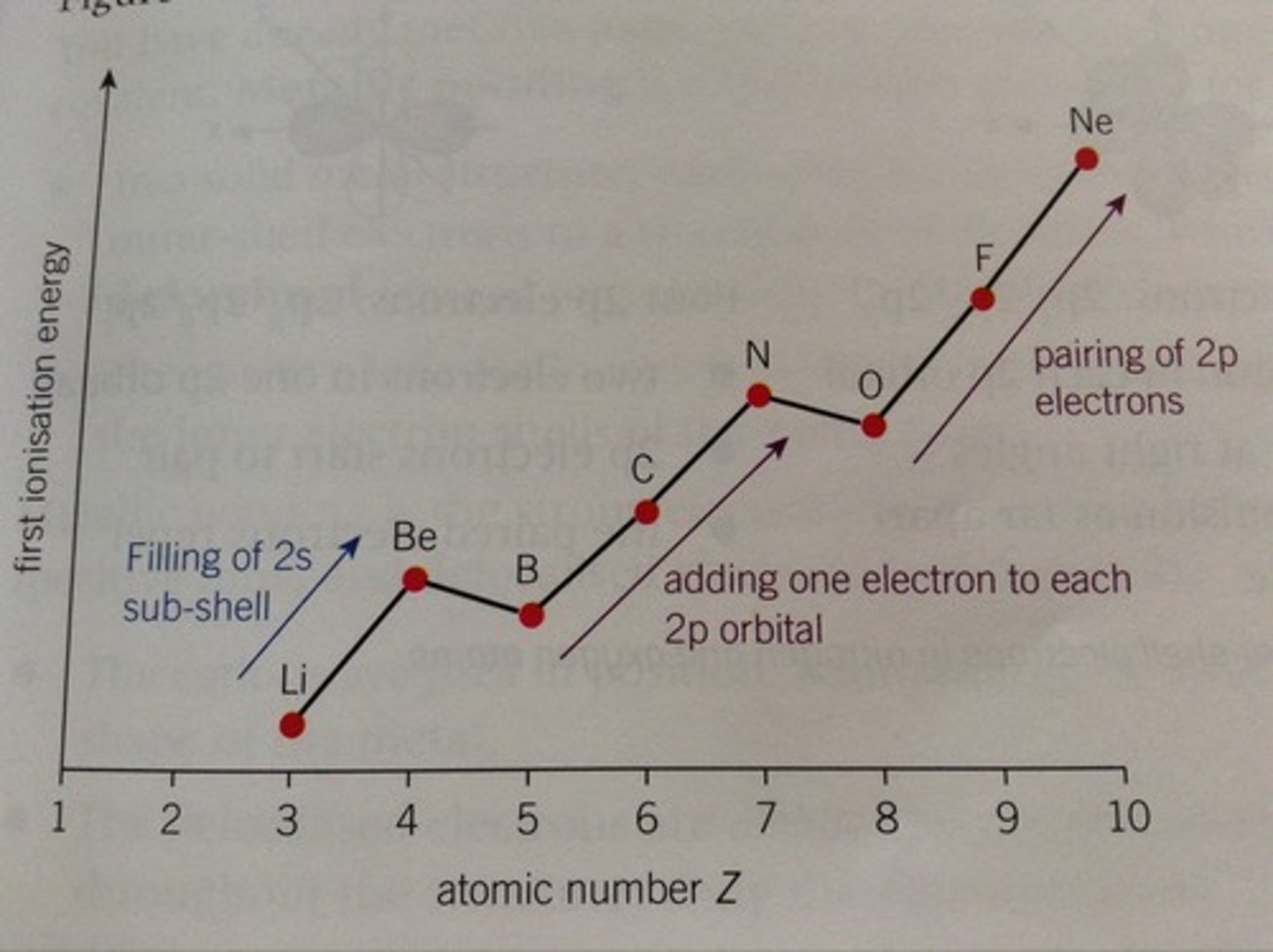
Comparing Nitrogen and Oxygen
In oxygen, the paired electrons in one of the 2p orbitals repel each other (like-charges)
Easier to remove electron from oxygen
1st I.E lower for oxygen
1st Ionisation energy (I.E)
Amount of energy required to remove one mole of electrons from one mole of atoms in a gaseous state
1st: X(g) --> X+(g) + e-
2nd: X+(g) --> X2+(g) + 2e-
Ionisation energy down a group
atomic radius increases
shielding increases
nuclear charge decreases
nuclear attraction to outer electrons decreases
1st I.E decreases
Ionisation energy across a period
atomic radius decreases
similar shielding
nuclear charge increases
nuclear attraction to outer electrons increases
1st I.E increases
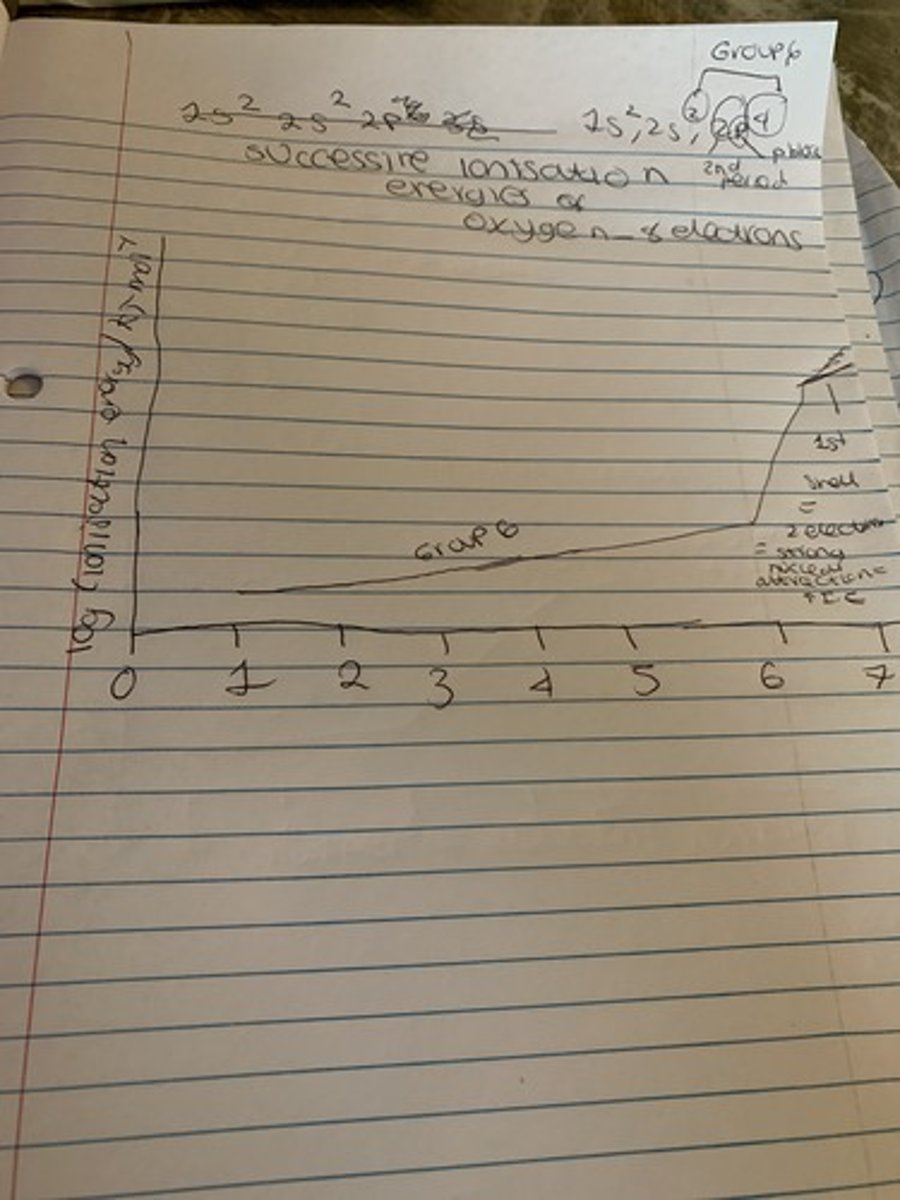
Ionisation graphs
Large jump = new shell
e.g. oxygen
large increase from 6-7th I.E
therefore must have 6 electrons in outer shell
therefore must be in group 6
Ionic bonding
Strong electrostatic attraction between oppositely charged ions (between metals and non-metals)
Melting/boiling point:
-high
-large amount of energy required to over come electrostatic attraction between oppositely charged ions
Electrical conductivity:
solid- no, fixed lattice
molten/aq- yes, mobile ions
Simple Covalent bonding
The strong electrostatic attraction between a shared pair of electrons and the nuclei of the bonded atoms (between 2 non-metals)
Melting/boiling point:
-low
-only london forces
Electrical conductivity:
no, no mobile charge carriers
Giant covalent bonding
Many atoms joined covalently and arranged in a giant regular lattice
Melting/boiling point:
-high
-lots of strong covalent bonds
Electrical conductivity:
diamond-no
silicon-no
graphene- yes
Solubility:
-insoluble
-covalent bonds holding atoms in lattice are too strong to be broken by interactions with solvents
diamond + silicon are tetrahderal, 109°
Metallic bonding
Strong electrostatic attraction between cations and delocalised electrons
Melting/boiling point:
-high
-many electrostatic forces to be broken, lots of energy needed
-giant lattice structure
Electrical conductivity:
yes- delocalised electrons carry charge
Solubility:
-insolubly
-any interactions lead to a reaction, not dissolving
Enthalpy change (△H)
Amount of heat evolved or absorbed in a reaction carried out at constant pressure
Exothermic
system--> surrounding
△H is -ve
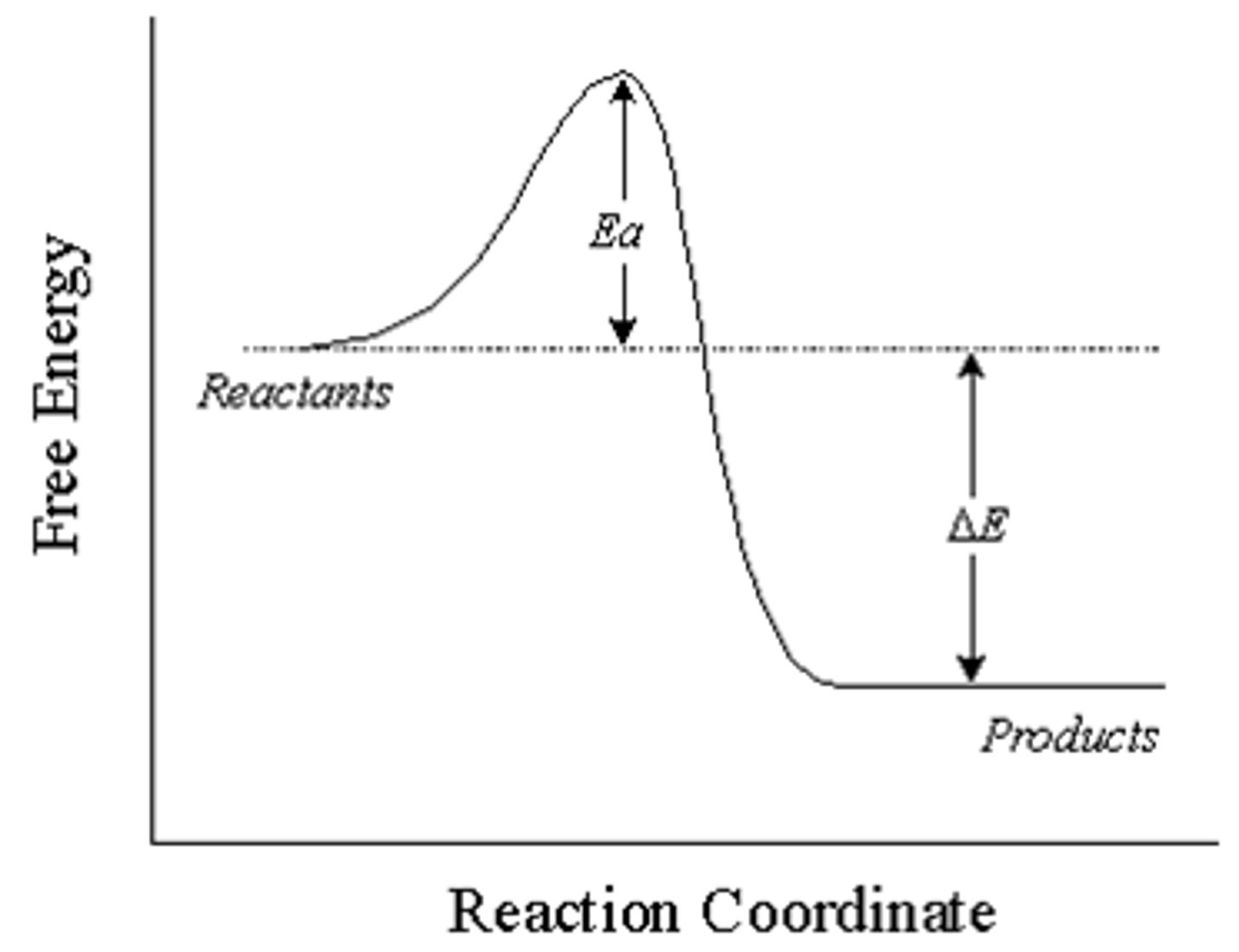
Endothermic
surrounding--> system
△H is +ve
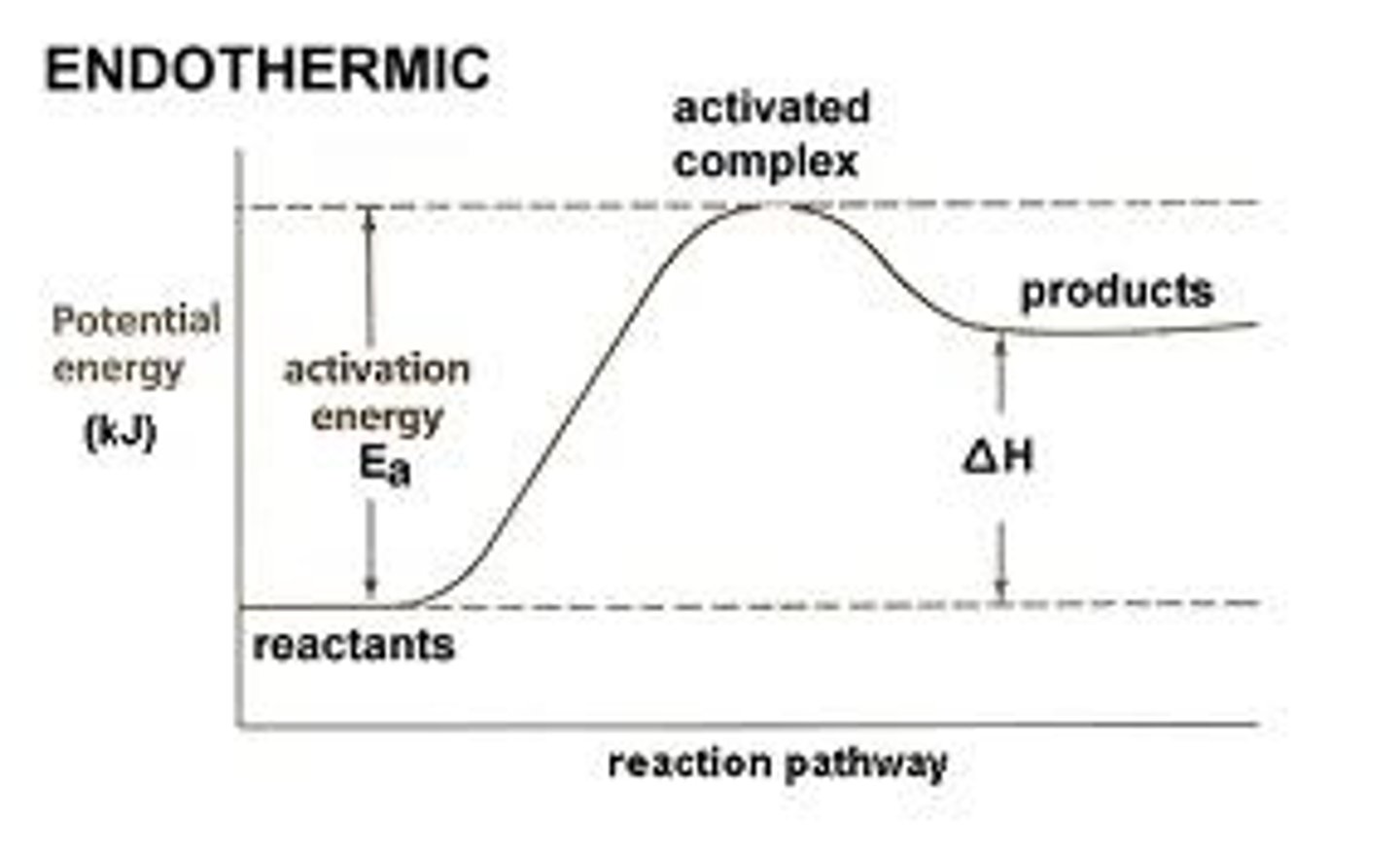
Activation energy (Ea)
Minimum energy required for a reaction to take place/break bonds
Standard conditions
Standard pressure= 100 kPa
Standard temp= 298K (25°C)
Standard conc= 1 mol dm-3
Standard state
Standard enthalpy change of reaction (△rHθ)
Enthalpy change that accompanies a reaction in the molar quantities shown in the chemical equation, under standard conditions + standard states
(if molar quantity is divided by 2, △rHθ is divided by 2)
△H (KJ mol-1) = sum bond enthalpies in reactants- sum bond enthalpies in products
Standard enthalpy change of formation (△fHθ)
Enthalpy change when one mole of a compound is formed from its elements in their standard states + under standard standard conditions
△fHθ for any element in standard state in 0
Standard enthalpy change of combustion (△cHθ)
Enthalpy change when one mole of a substance reacts completely with oxygen under standard conditions + standard states
Standard enthalpy change of neutralisation (△neutHθ)
Enthalpy change that accompanies the reaction between an acid and base to form one mole of water under standard conditions + standard states
(value is same for all neut reactions, -57)
Measuring enthalpy change
1) q= mc△T
m= mass heated (of water/aq solution)
c= 4.18
T= temp change
2) Calculate moles
3) Calculate △H
△H = q/ mol
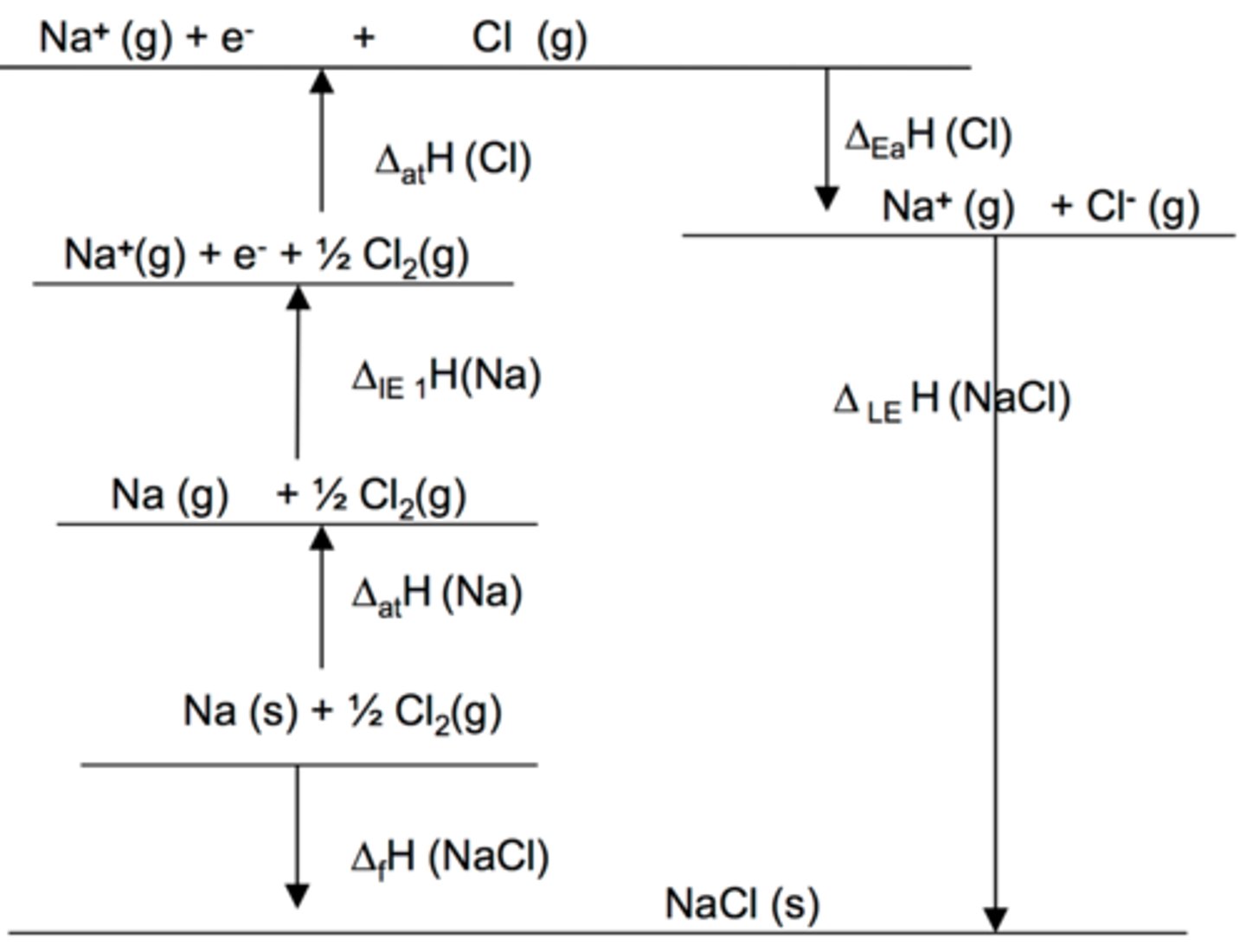
Born-Haber cycle
Rates equation
Rate= conc/time
Rate ∝ [A]^a
Rate= k[A]^a [B]^b
Orders of reactions
Zero order:
Rate∝ [A]^0
conc has no effect on rate
First order:
Rate∝[A]
if conc is doubled, rate is doubled
Second order:
Rate∝[A]^2
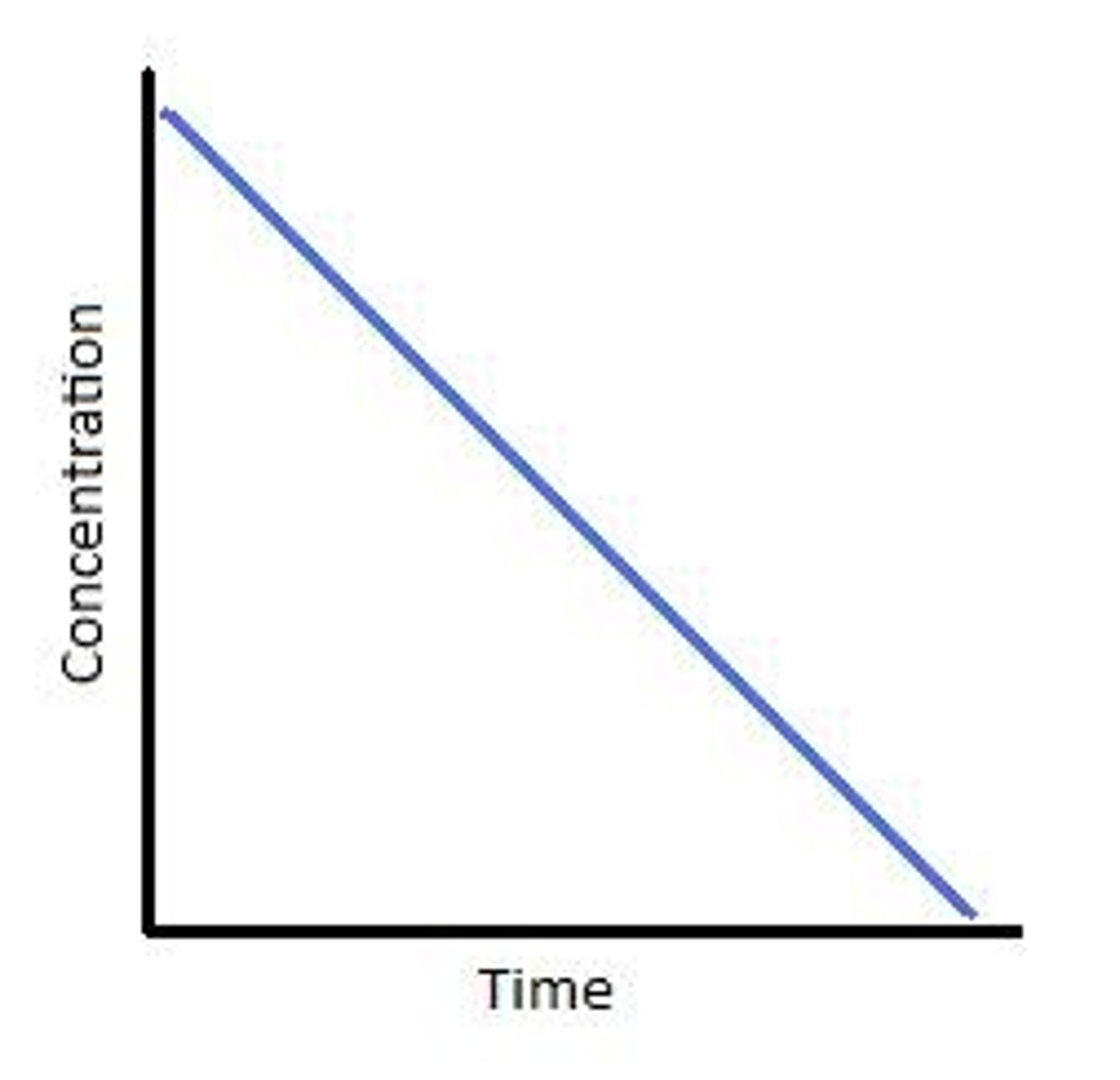
Conc time graph zero order
Straight line with negative gradient
Conc time graph first order
Constant half life
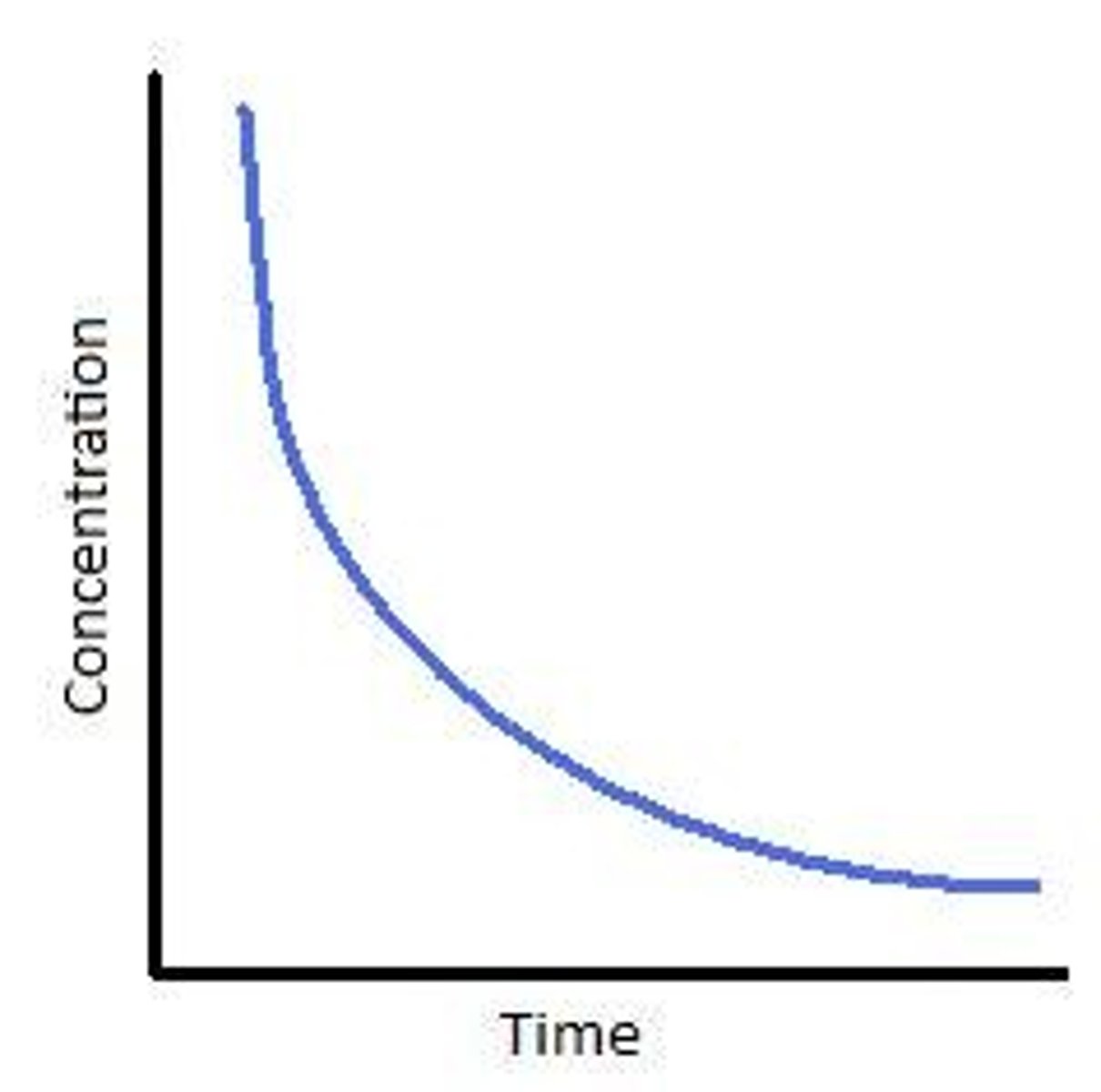
Conc time graph second order
Half life increases as time increases
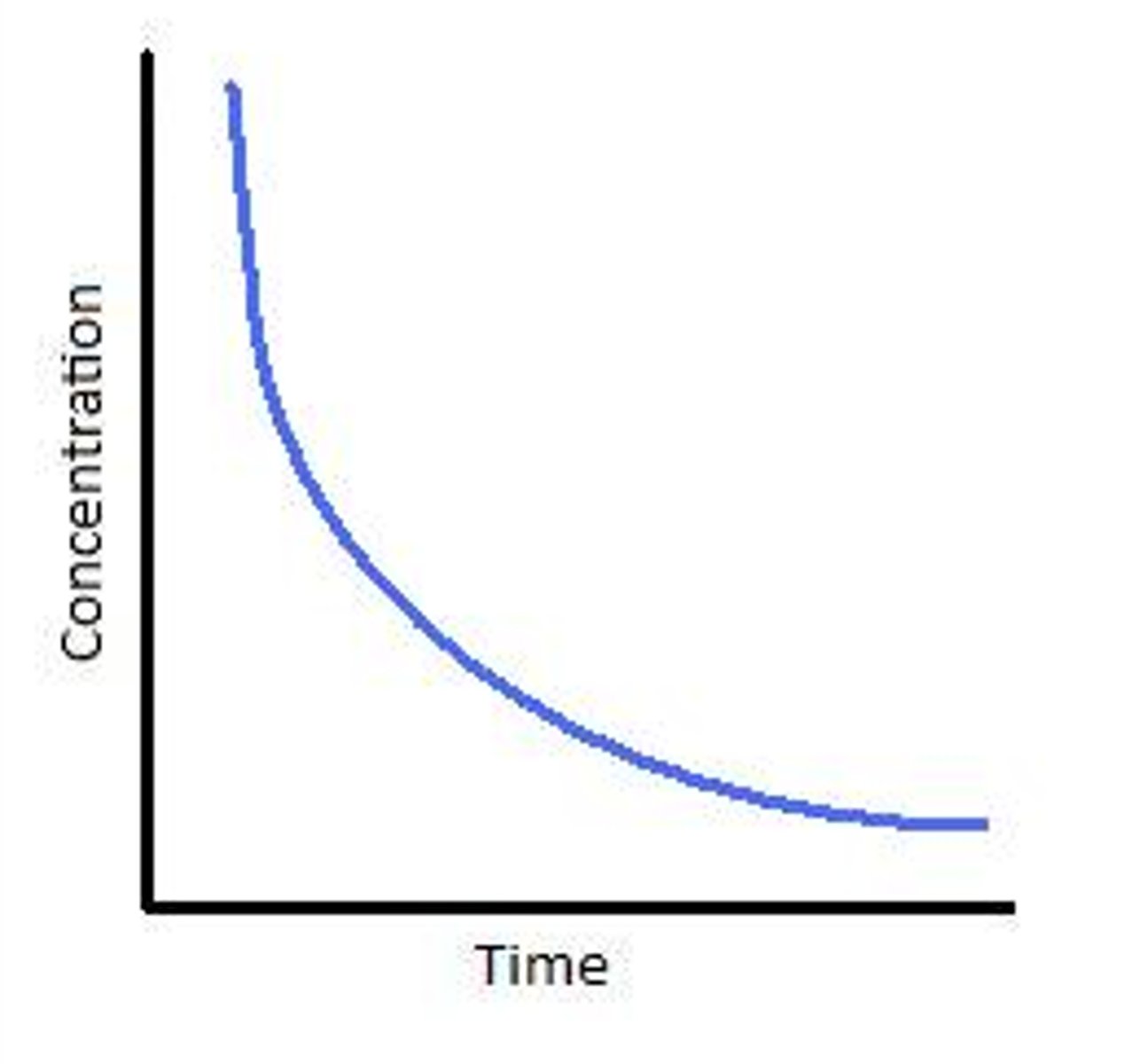
How to work out rate constant for first order reaction
method 1: calculate tangent of curve at half life
method 2: K= ln2/half life
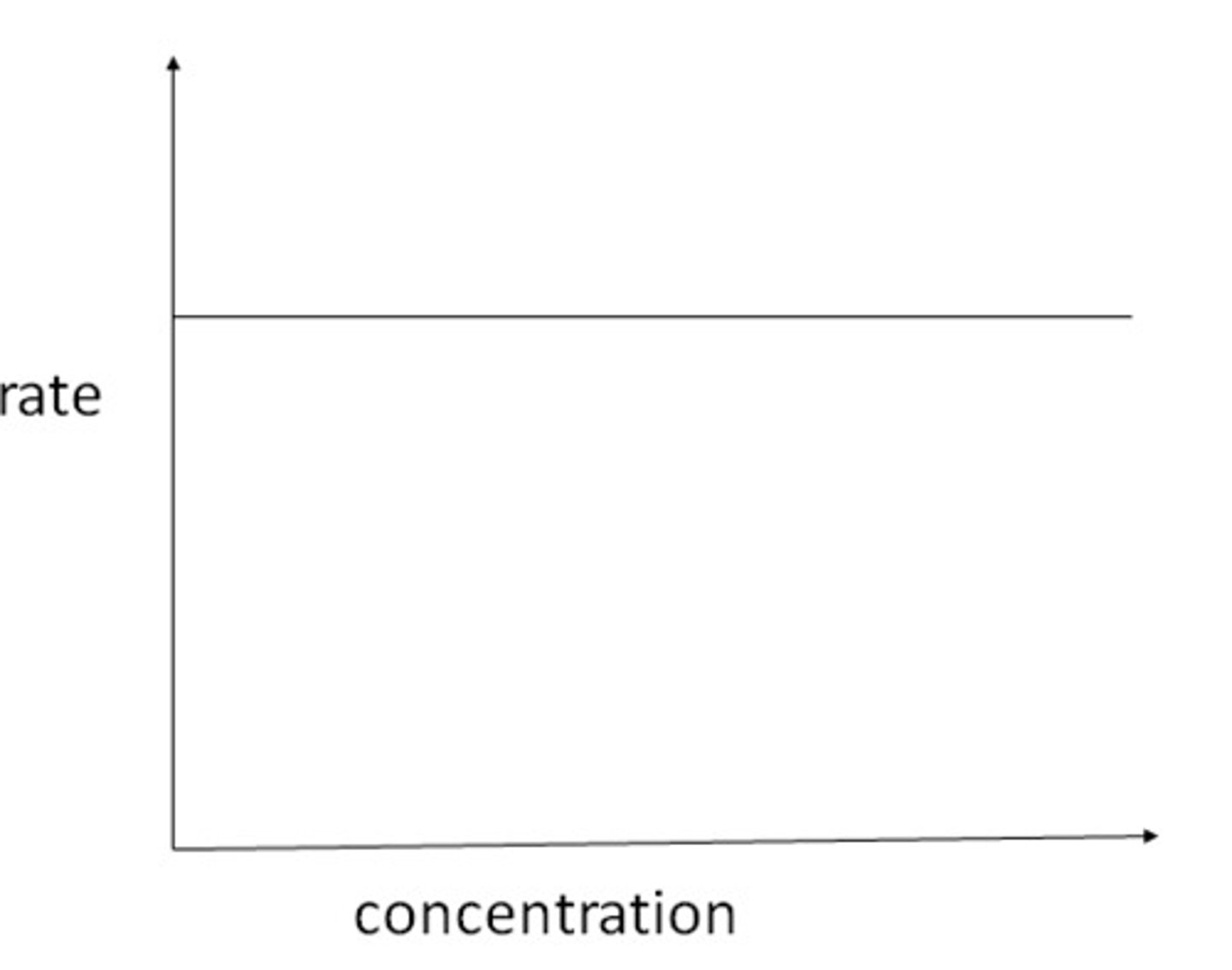
Rate-conc graph zero order
Rate=K
therefore K= y-intercept
Rate-conc graph first order
Rate∝K
Rate= K[A]
therefore K= gradient
Rate-conc graph second order
Rate= k[A]^2
K determined by plotting second graph of rate against conc^2
![<p>Rate= k[A]^2</p><p>K determined by plotting second graph of rate against conc^2</p>](https://knowt-user-attachments.s3.amazonaws.com/ac206ca9-c268-43b5-ac71-9e25e6b99de9.jpg)
Rate-determining step
the slowest step in a reaction mechanism, the one determining the overall rate
If something shows in rate equation but not overall equation, its a catalyst
Arrhenius equation
k = Ae^(-Ea/RT)
R= gas constant (8.314)
T=temp (k)
Ea= activation energy
Increase in temp= rate increases and rate constant increases
Catalyst= Ea lowers = rate increases
Logarithmic form of Arrhenius equation
lnk = -Ea/RT + lnA
A plot of LnK against 1/T gives downswards linear graph of type y=mx+c, where m= -Ea/R and c=lnA
Average bond enthalpy
The energy required to break one mole of a specified type of bond in a gaseous molecule
measure of average strength on covalent bond
always endothermic ( positive value)
BENDO MEXO
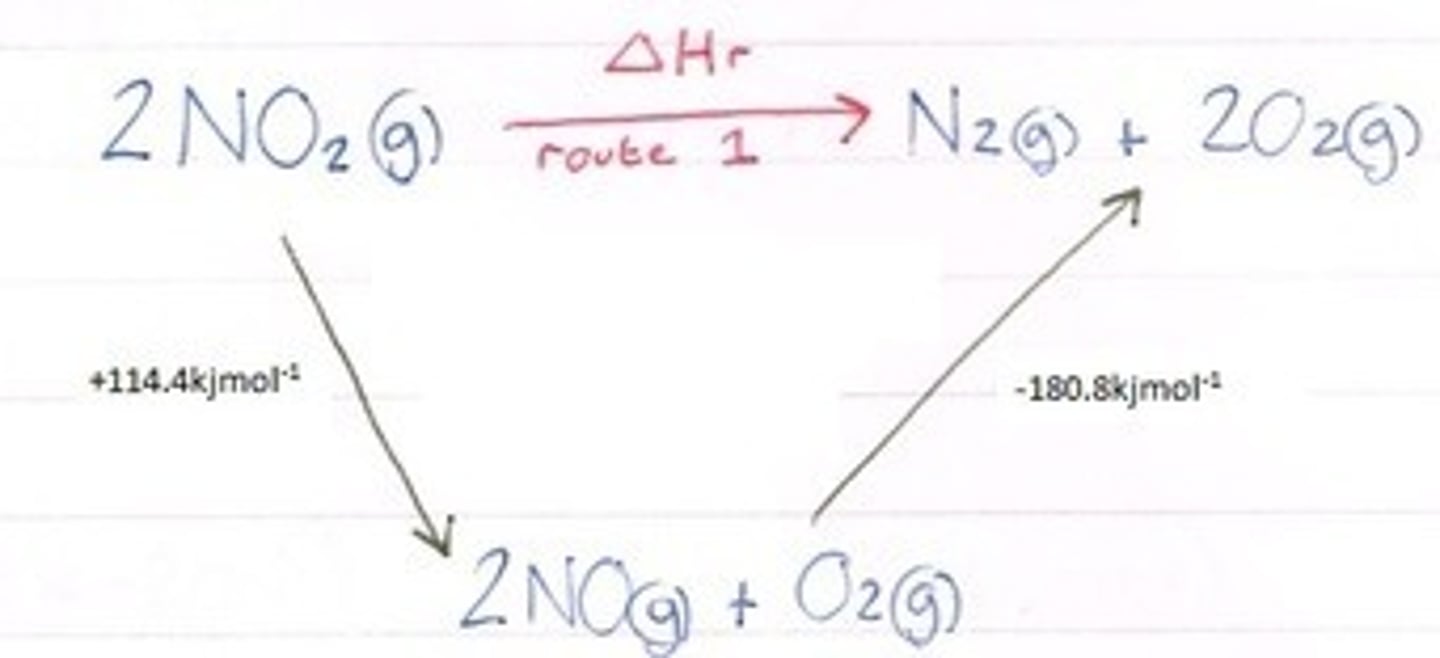
Hess' Law and enthalpy cycles
FUCD- formation up, combustion down
Entropy
dispersal of energy and disorder within the chemicals making up a chemical system (JK-1 mol-1)
Predicting entropy changes
1) At 0k there is no energy and all substrates have an entropy of 0
2) Entropy increases with changes in state to give more random particles
More random system (e.g. gas) = more positive entropy change
3) Changes in number of gas molecules
e.g. more gas molecules in reactants = entropy decreases = entropy is negative
4) Dissolving in crystalline solid (ionic)
solid is highly ordered, solution is disordered --> entropy increases
Standard entropy (Sθ)
The entropy of one mole of a substance under standard conditions
△Sθ = sum of Sθ products - sum of Sθ reactants
Gibbs free energy
overall change in energy during a chemical reaction
△G = △H -T△S
convert △S to kilo by dividing by 1000
Feasibility of a reaction △G
△G > 0 then reaction isnt feasible
△G <0 then reaction is feasible
note: may be feasible, the reaction may not actually happen in any sensible time
△G as a graph
△G= y axis
△H= y intercept
△S= gradient
T= x axis
Mean titre
sum of concordant titres / number of concordant titres
concordant titres are within 0.1 of each other
% uncertainty
Uncertainty x 2 /mean titire x 100
Test for ammonium (NH4 +)
Dilute NaOH + gently heat
may be able to smell ammonia
moist pH indicator --> blue
NH4 +(aq) + OH-(aq) --> NH3(g) + H2O(l)
Test for Carbonate (CO3 2-)
Dilute acid
Efforvesence
Gas (CO2) turns limewater milky
CO3 2-(aq) + 2H+(aq) --> CO2(g) + H2O(l)
Test for sulphate (SO4 2-)
Dilute HCl + Barium chloride
white precipitate (barium sulfate) formed
Ba2+(aq) + SO4 2-(aq) --> BaSO4(s)