Transition metal complexes/ coordinate compounds
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
what are ligands
An ion or molecule that donates a pair of electrons to a transition metal ion, forming a dative covalent bond (a coordinate bond.)
what is a transition metal complex
2,4 or 6 ligands often surround the TM ion
Altogether, the TM ion and the ligands= transition metal complex
what is coordinate number
The total number of dative covalent bonds (coordinate bonds) around the metal ion
why do transition metals form complexes
Transition metals form complexes due to their small size- this allows them to attract electron-rich species more strongly
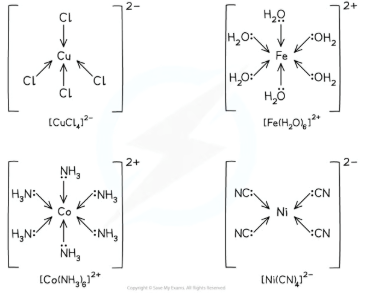
what are monodentate ligands
If a ligand donates one pair of e- to a TM ion/only forms one dative bond to the central metal ion
Examples of monodentate ligands are:
Water (H2O) molecules
Ammonia (NH3) molecules
Chloride (Cl–) ions
Cyanide (CN–) ions
what are bidentate ligands
Can donate two lone pairs of electrons (from two seperate atoms) forming two dative bonds to the central metal ion
Examples of bidentate ligands are:
1,2-diaminoethane (H2NCH2CH2NH2) which is also written as ‘en’
Ethanedioate ion (C2O42- ) which is sometimes written as ‘ox’
what are multidentate ligands?
These make more than two coordinate bonds with a TM ion-contain more than two atoms with lone pairs of electrons
An example of a multidentate ligand is EDTA4-, which is a hexadentate ligand as it forms 6 dative covalent bonds to the central metal ion
why is EDTA4- called a chelating agent
EDTA4- binds to metal ions reducing their concentration in solutions.
Uses of EDTA4-
Detergents (remove Ca2+ / Mg2+ from hard water)
Foodstuffs- as a stabiliser to remove ions that catalyse
Medicines- to prevent blood clotting or to remove heavy metal ions and prevent poisoning eg Hg/Pb
Shape of complex with 2 ligands
Linear
180 degrees
shape of complex with 2 ligands/coordinate bonds
Have tetradedral shape
Complexes with four chloride ions most commonly adopt this geometry
Chloride ligands are large, so only four will fit around the central metal ion
The bond angles in tetrahedral complexes are 109.5o
what shape do compelxes with 6 ligands make
Octahedral
This could be six coordinate bonds with six small, monodentate ligands
As there are six ligands, these complexes are sometimes described as having six-fold coordination
It could be six-coordinate bonds with three bidentate ligands
Each bidentate ligand will form two coordinate bonds, meaning six-coordinate bonds in total
It could be six-coordinate bonds with one multidentate ligand
Bond angle: 90 and 180, usually H2O. NH3 or OH-
how many bonds do chlroide ions usually make
Chlroide ions much greater in size, so fewer surround the central ion. Usually TM-chloride compelxes have 4 Cl-
can transition elements exhibit stereosiomerism?
Transition element complexes can exhibit stereoisomerism
Even though transition element complexes do not have a double bond, they can still have geometrical isomers
Square planar and octahedral complexes with two pairs of different ligands exhibit cis-trans isomerism
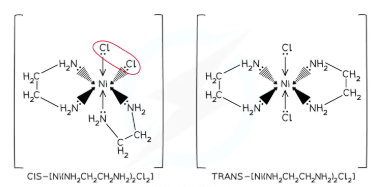
what is cis isomer and trans isomer in TM compelxes
if the two ‘different’ ligands are next to each other then that is the ‘cis’ isomer, and if the two ‘different’ ligands are opposite each other then this is the ‘trans’ isomer
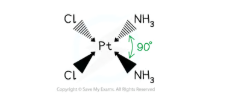
what are square planar complexes
Sometimes, complexes with four coordinate bonds may adopt a square planar geometry instead of a tetrahedral one
Cyanide ions (CN-) are the most common ligands to adopt this geometry
An example of a square planar complex is cisplatin
The bond angles in a square planar complex are 90o
Use of cis and trans platin
Cis-platin is a square planar molecule that has a geometric isomer with the side groups in different positions
Used as a cancer treatment drug
It binds to nitrogen atoms on bases in DNA in fast-growing cancer cells
It passses through cell membrane and undergoes ligand exchange where chlorine are replaced by water molecules
The nitrogen is a better ligand than water and forms dative covalent bonds with the cis-platin
The cis-platin distorts the shape of the DNA and prevents the DNA from replicating
Trans-platin is less effective and is toxic, so cis-platin is administered as a single isomer
Adverse effects of cis-platin
Cis-platin binds to healthy cells as well as cancerous cells, but affects cancer cells more as they are replicating faster
Unfortunately, this means that other healthy cells which replicate quickly, such as hair follicles, are also affected by cis-platin
This is why hair loss is a side-effect of people undergoing cancer treatment
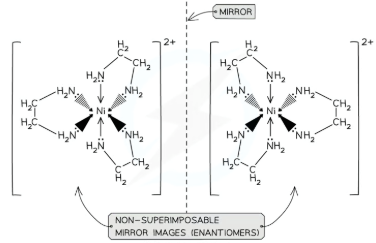
what type of TM complex can form optical isomers?
Octahedral complexes with bidentate ligands also have optical isomers
This means that the two forms are non-superimposable mirror images of each other
They have no plane of symmetry, and one image cannot be placed directly on top of the other
The optical isomers only differ in their ability to rotate the plane of polarised light in opposite directions
what is haem group in hameoglobin
The haem molecule is a complex with iron(II) at its centre
The haemoglobin complex contains a multidentate ligand made up of four haem groups
These consist of mostly carbon and hydrogen atoms
Each haem group has a nitrogen atom forming a dative covalent bond to the Fe2+ ion in a square planar complex
There is a fifth dative bond from the protein (globin) to the Fe2+ ion
Oxygen atoms form a dative covalent bond with the iron(II) which enables oxygen molecules to be transported
job of haemoglboin
Hb is a pigment foudn in rbc. They transport oxygen from the lungs to every cell in body, where O2 is used for respiraiton.
Oxygen molecules are not very good ligands and bond weakly to the iron(II)
The weak bonds allows them to break off easily and be transported into cells
why is carbon monoxide toxic?
Carbon monoxide binds irreversibly with haemoglobin
This shows that the coordinate/dative bond formed from CO to Hb/ Iron II is more stronger /stable than the Hb-O2 dative covalent bond
Prevents oxygen being carried to cells and leads to oxygen starvation and death by suffocation
If oxygen attached to the haemoglobin (oxyhaemoglobin) is replaced by carbon monoxide (carboxyhaemoglobin), a darker red colour is produced in the haem complex
when does anaemia occur
The condition anaemia occurs when a person does not have enough haemoglobin in their blood due to a loss of blood or deficiency in iron
Deficiency in iron can be restored by taking iron sulfate tables in the diet