cell bio exam 3
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
metabolic pathways
aka cell respiration, includes anabolic and catabolic pathways
anabolic pathways, entropy effects, energy requirement
build up pathways (often polymers like starch/glycogen), decrease entropy, endergonic
catabolic pathways, entropy effects, energy requirement
breaking down pathways (ex hydrolysis of glucose), increase entropy, exergonic
high chemical energy molecule examples that can convert to ATP
GTP (higher in energy), creatine phosphate, NADH (reduced coenzyme, chem potential energy)
energy rich properties of ATP
adenine base, 5c ribose (linked to P groups by phosphoester bond), chain of 3 phosphate groups linked to each other by phosphoanhdyride bonds
where is the missing H that balances the hydrolysis of ATP to ADP reaction?
it is donated to solution as a proton
when is energy released in atp
when phosphoanhydride bonds are hydrolyzed, because Pi is much more stable than reactant
why is atp hydrolysis exergonic (3)
charge repulsion between p groups, resonance stabilization of products, inc entropy/ solubility of products
why is amp less energy than adp and atp?
no charge repulsion btwn p groups
what is delta g prime standard based on
25 degrees C, adp and atp in equal concentration,
what does delta g standard prime actually end up being
-10 to -14 kca/mol in cell, atp concentration is 5x adp driving the hydrolysis
biological oxidation involves
loss of H (dehydrogenation)
what enzymes remove H atoms
dehydrogenases
where do the removed e go from oxidation reaction
to another molecule, which is considered reduced
reduction
addition of e, endergonic process
hydrogenation
reaction involved in reduction becuase transfered e- are accompanied by protons (overall will be neutral H)
typical e- and H acceptors during biological oxidations
conenzymes, carry energetic electrons
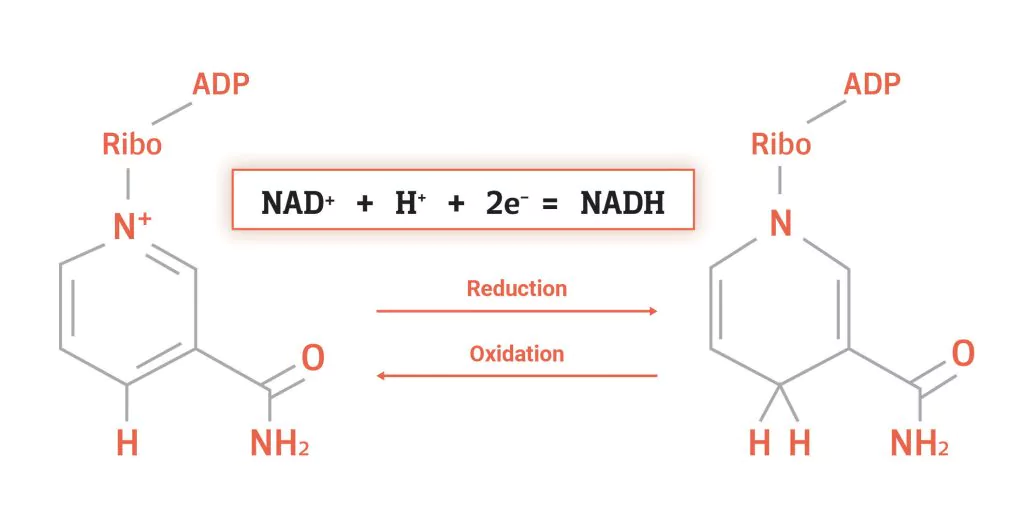
coenzyme involved in energy metabolism (oxidized form)
NAD+ , will becomes NADH + H+ when 2H are added
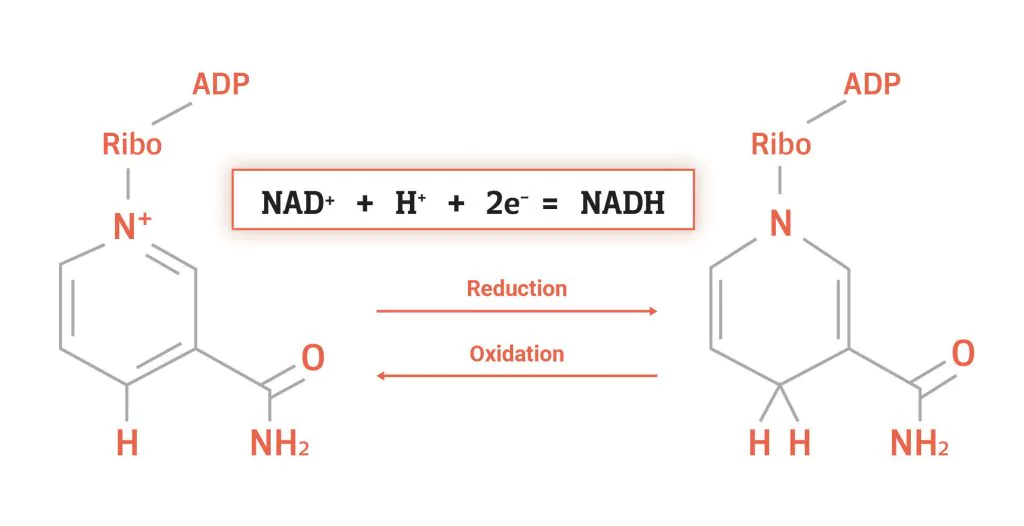
observe each molecule and how it changes in redox rxns
what produces energy from organic compounds (carbs, proteins, fats) and in what form?
oxidation of organic compounds, in form of ATP and reduced coenzymes
oxidation of glucose, what is roughly the delta g, end products, and final e- acceptor
highly exergonic, -686kcal/mol for complete conversion of glucose to CO2 and H2O; O2 is final electron acceptor
do cells obtain the full delta g of glucose oxidation
no, energy conversion is not 100% efficient (entropy)
are coenzymes consumed
no they are recycled
overall reaction of glucose
glucose→ 6 co2
glycolysis and its yield
glucose→ 2 pyruvates; 2 atp, 2nadh
pyruvate oxidation and its yield
each pyruvate → acetyl coA (2 total) ; 2NADH
krebs cycle and its yield
2 acetyl coA→ 4 CO2 ; 2atp and 2 fadh2
summary of reactions for phase 1 of glycolysis
glucose is phosphorylated
glucose→ fructose
another phosphorylation
split in half
2 g3p isomers produced
summary of enzymes for phase 1 of glycolysis
kinase
isomerase
kinase
unimoprtant
isomerase
why can phase 2 of glycolysis be done twice
because phase 1 produces 2 g3p isomers
phase 2 of glycolysis reactions summary
redox phosphorylation (energy step)
remove Pi that was just added, add it to adp to make atp
enzymes for phase 2 of glycolysis
dehydrogenase
kinase
phase 3 of glycolysis summary of reactions
move P group to another carbon
removal of h2o
remove P group from C, add it to adp to make atp
enzymes for phase 3 of glycolysis
mutase (isomerase family)
kinase
what is substrate level phosphorylation and which steps of glycolysis use this
direct synthesis of atp by transfer of P from a high energy substrate to ADP; steps 7 and 10 of glycolysis (both involve kinases)
summarize atp input and output for glycolysis
input 2atp, produce 2 atp in phase 2, produce 2 atp phase 3 (net 2 atp)
does pyruvate formation require oxygen
NO
brief overview of 3 phases of glycolysis
preparation and cleavage
oxidation
pyruvate formation
what must fermentation regenerate in the oxidized form so glycolysis can continue?
Nad+
2 types of anaerobic metabolism of glucose
to lactate, and to ethanol + CO2
why isnt there nad+ nadh or h+ shown in reaction of glucose to lactate
no net change in amounts of those molecules
proprionate fermentation
pyruvate → reduced to proprionate
glycol fermentation
butylene glycol formation
fermentation pathway common theme
reoxidizing nadh to nad+ by transfer of e- to an organic acceptor
essential features of fermentation process (2)
no external e- acceptor
no net oxidation
what does it mean that cancer cells exhibit aerobic glycolysis
they ferment glucose to lactate even in the presence of oxygen, allows them to outgrow normal cells
what can detect these cancer cells
pet scans via flurodeoxyglucose which will accumulate in cancer cells
radiolabeled biochemical precursors serve what purpose
label different C in glucose with the isotope c14, allowing tracing of individual molecules