Aromatics Ochem
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
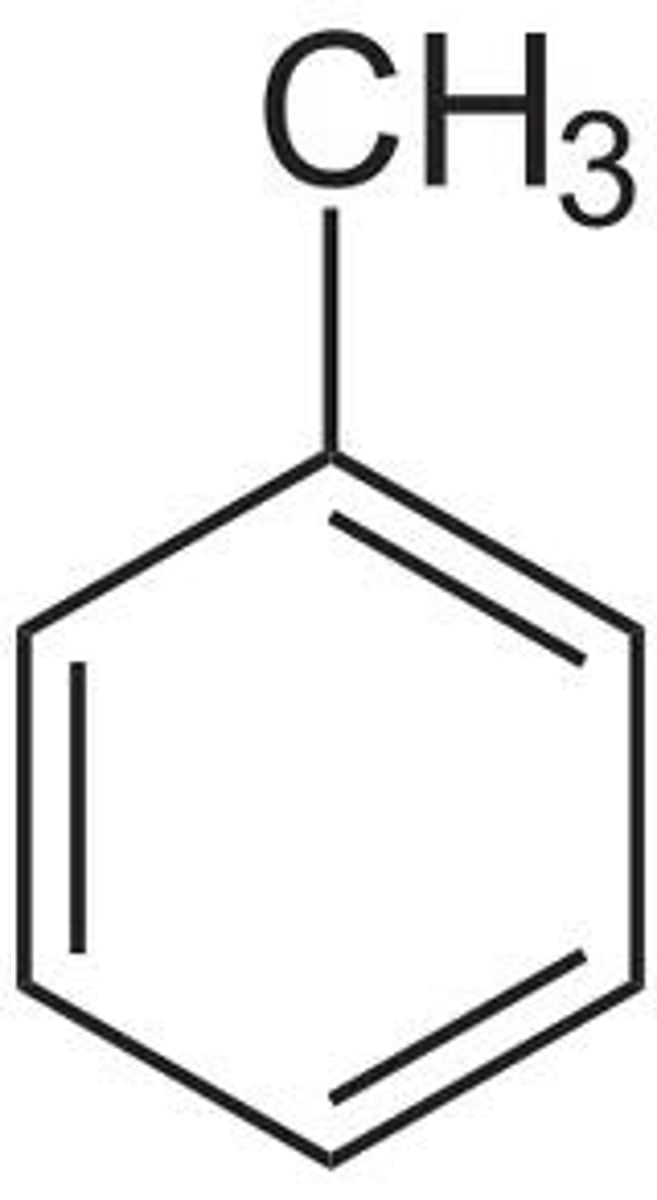
Toluene
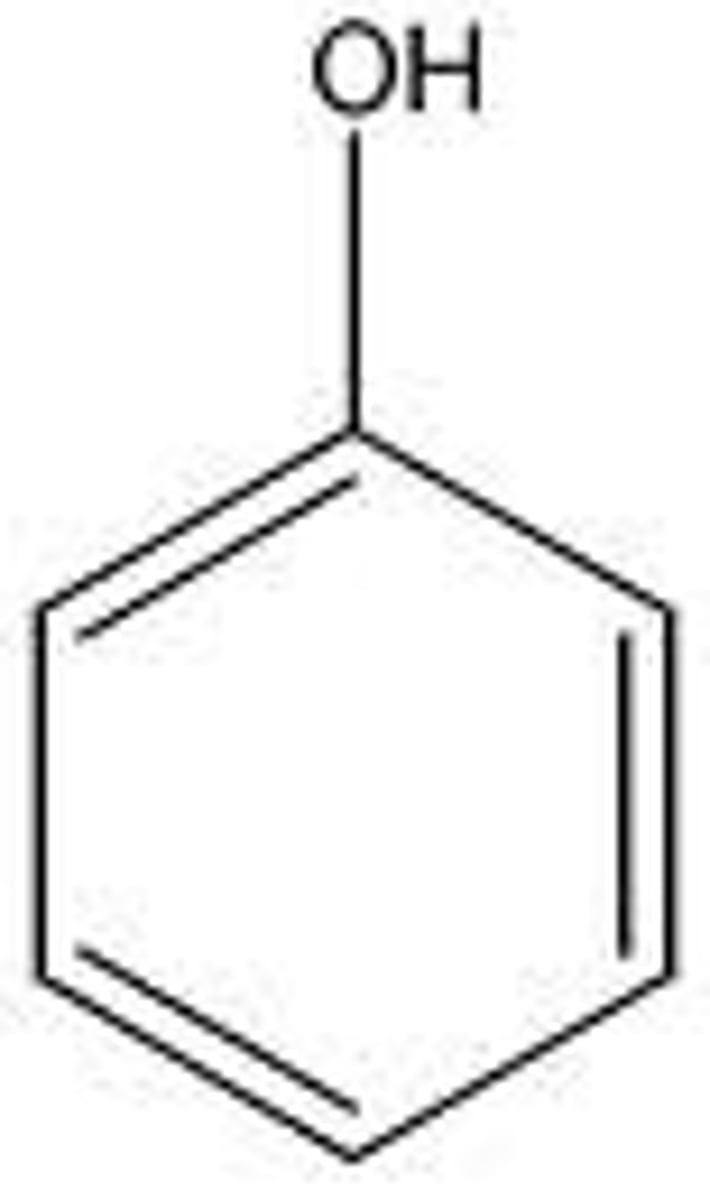
phenol
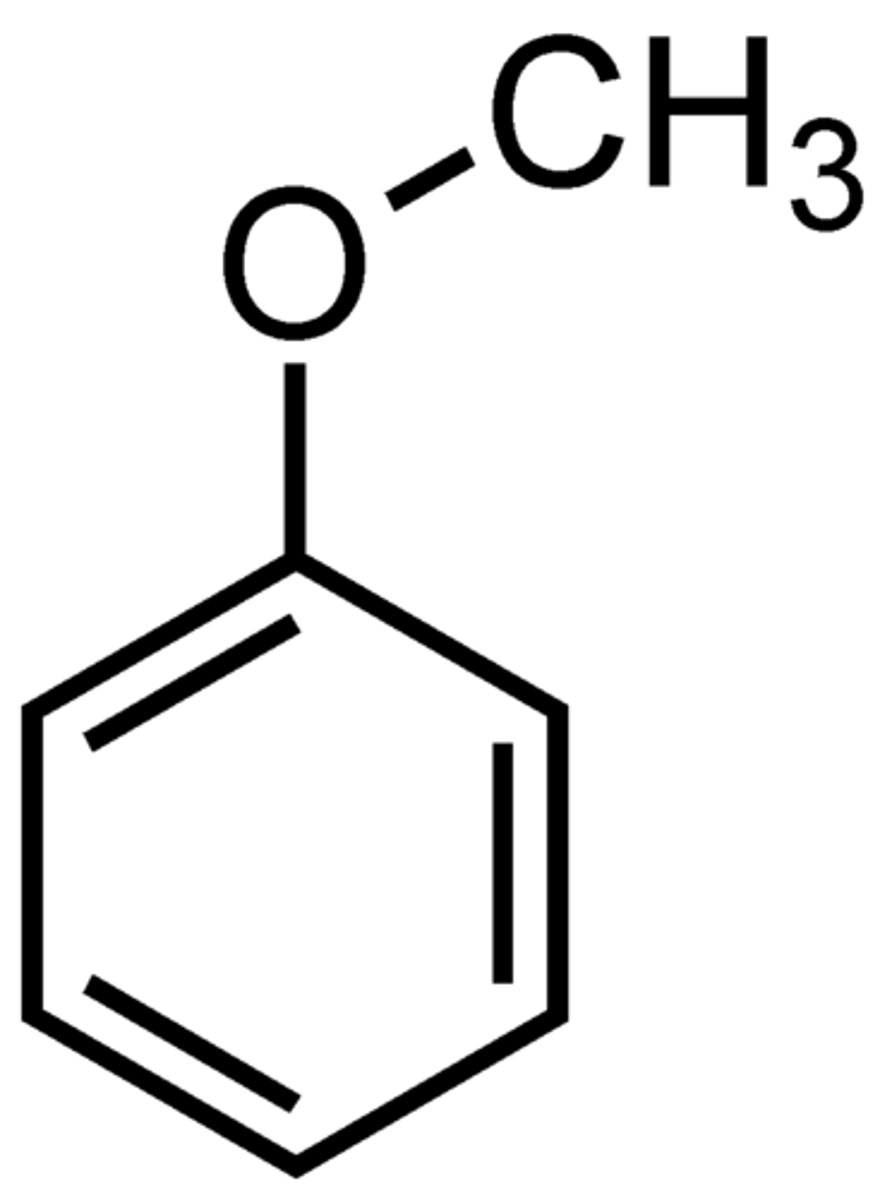
anisole
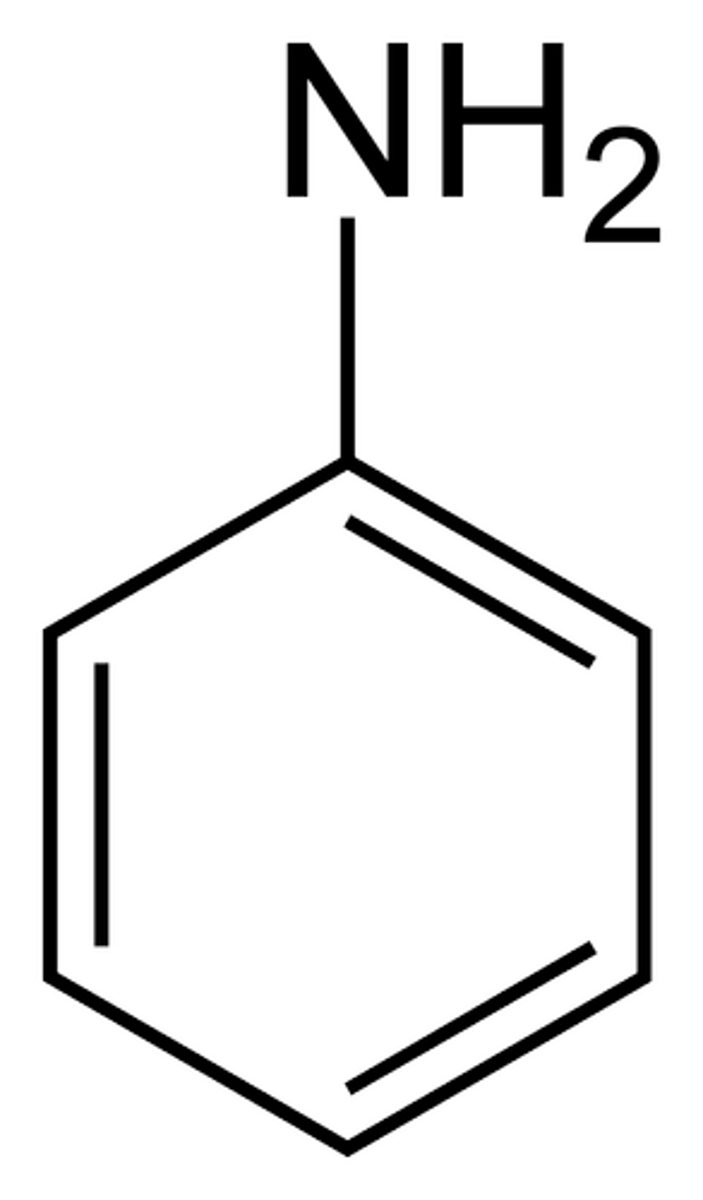
Aniline
benzoic acid
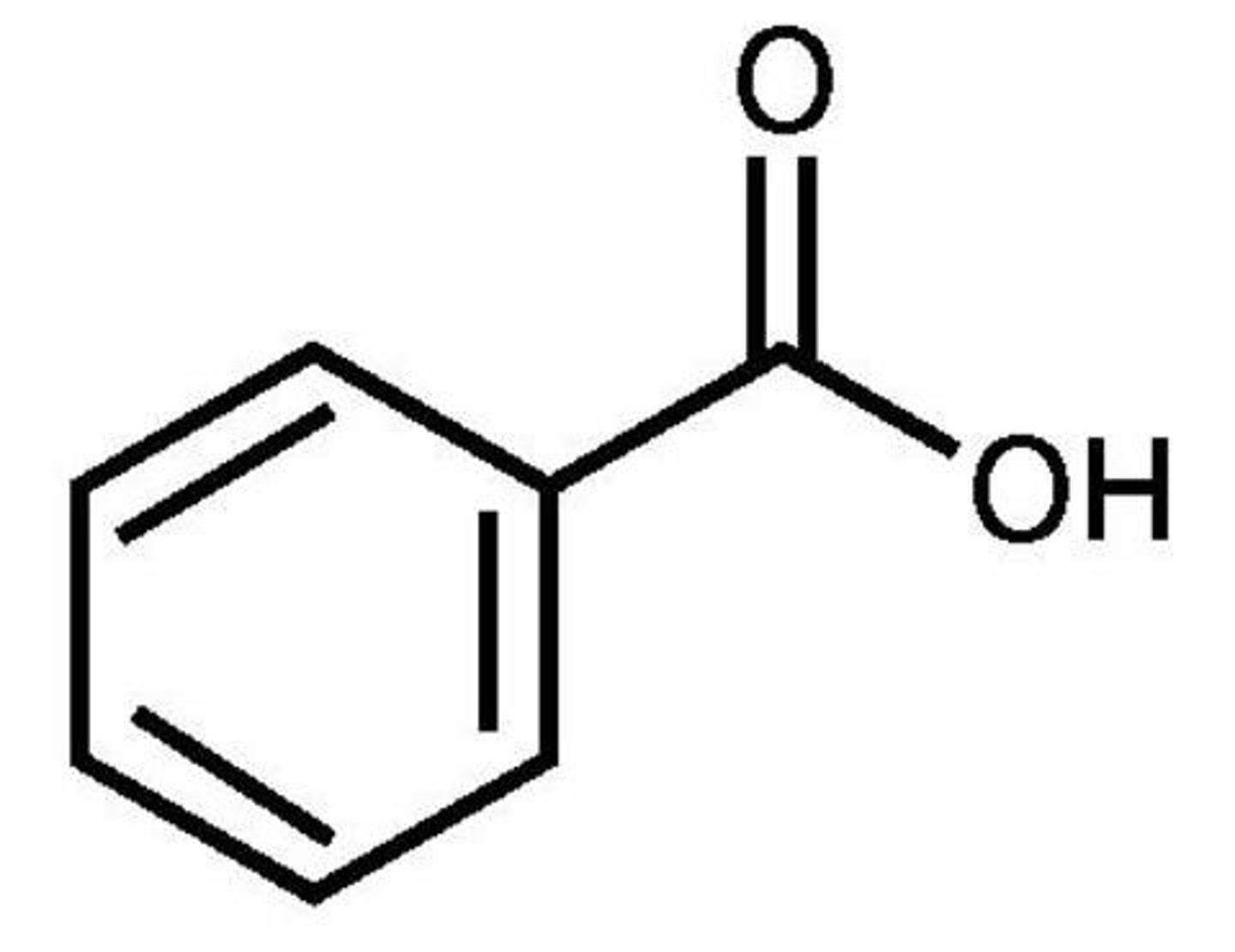
Benzaldehyde
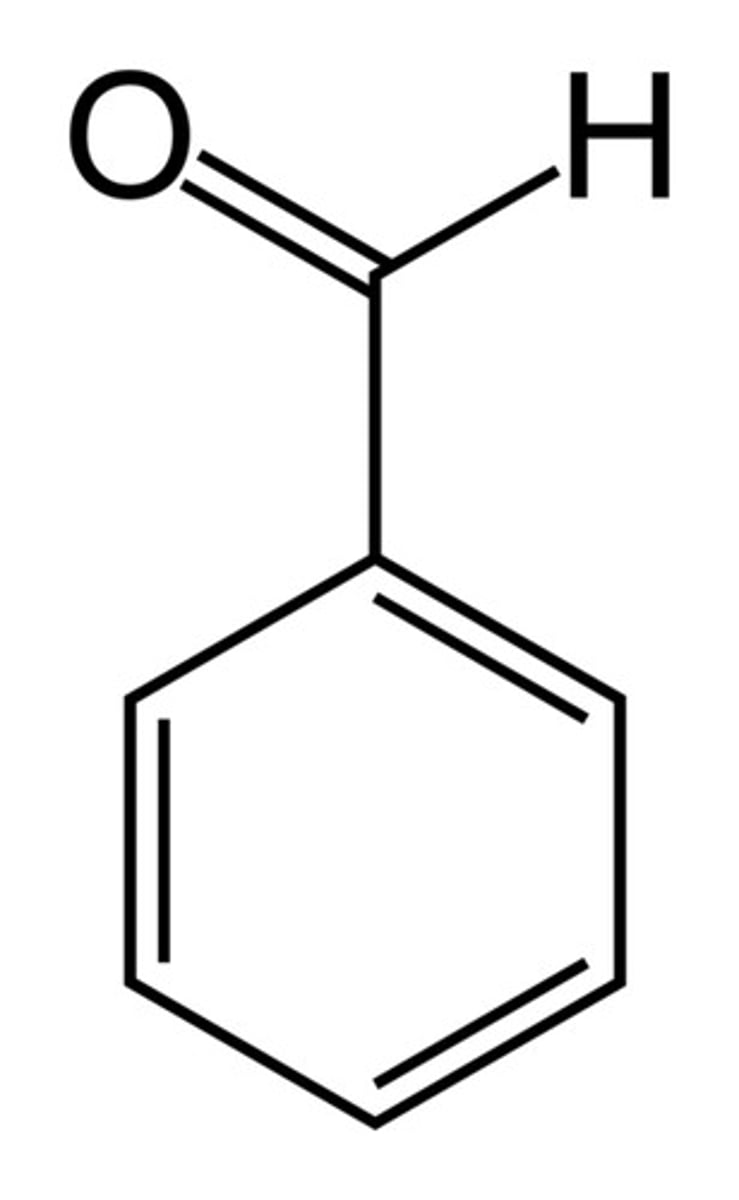
Acetophenone
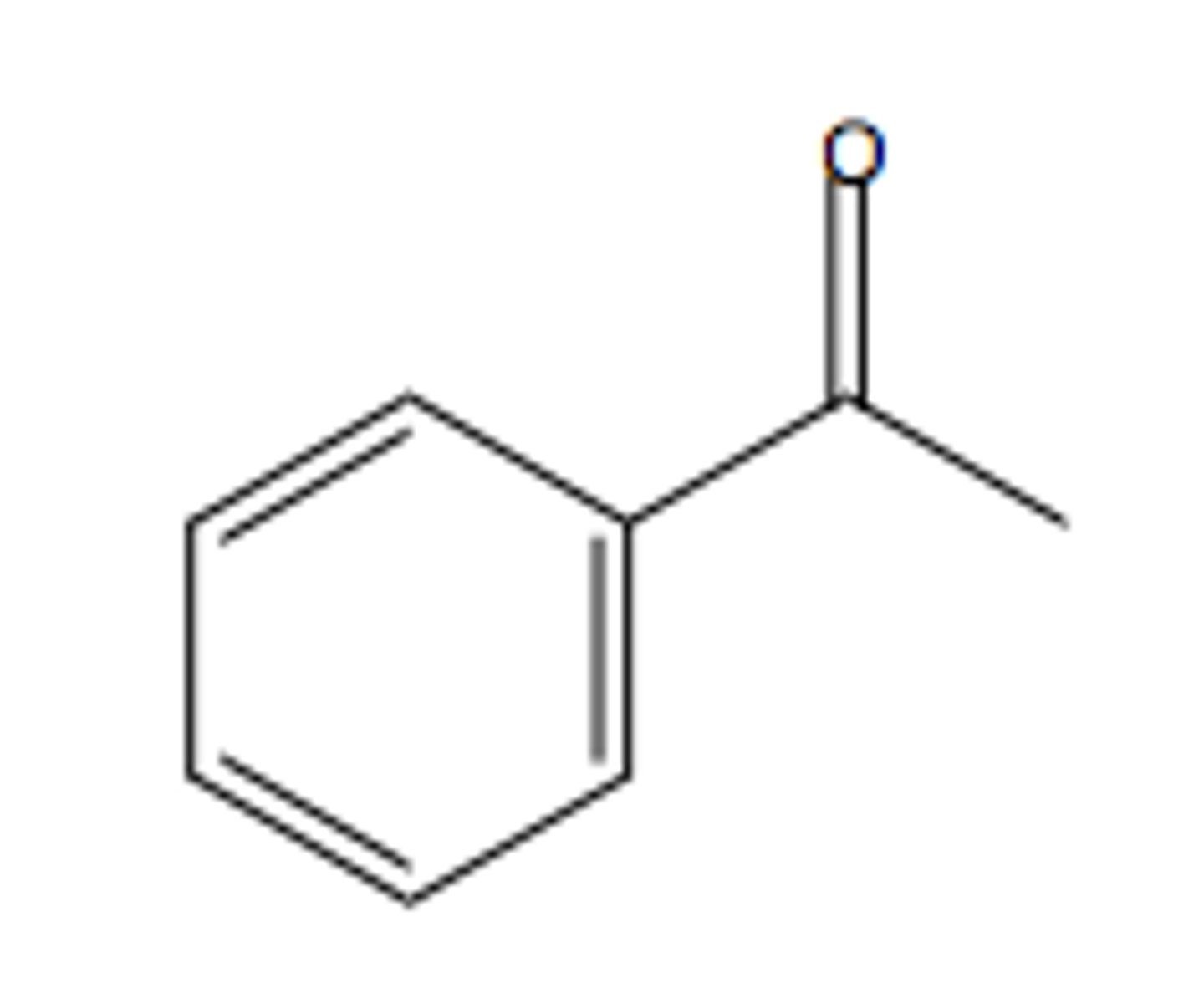
stylene
Explain Huckel's Rule
A structure can only be an aromatic if the number of electrson of the ring is 2, 6, 10, 14, 18.
What are the two criteria that need to be meat to be an aromatic
1.) Compound must have a ring comprised of continuously overlapping p orbitals
2.) Huckel's rule needs to be followed
If a compound does not have overlapping p orbitals what is it called
Nonaromatic
If a compound has overlapping p orbitals but does not follow Huckel's rule what is it called?
Antiaromatic
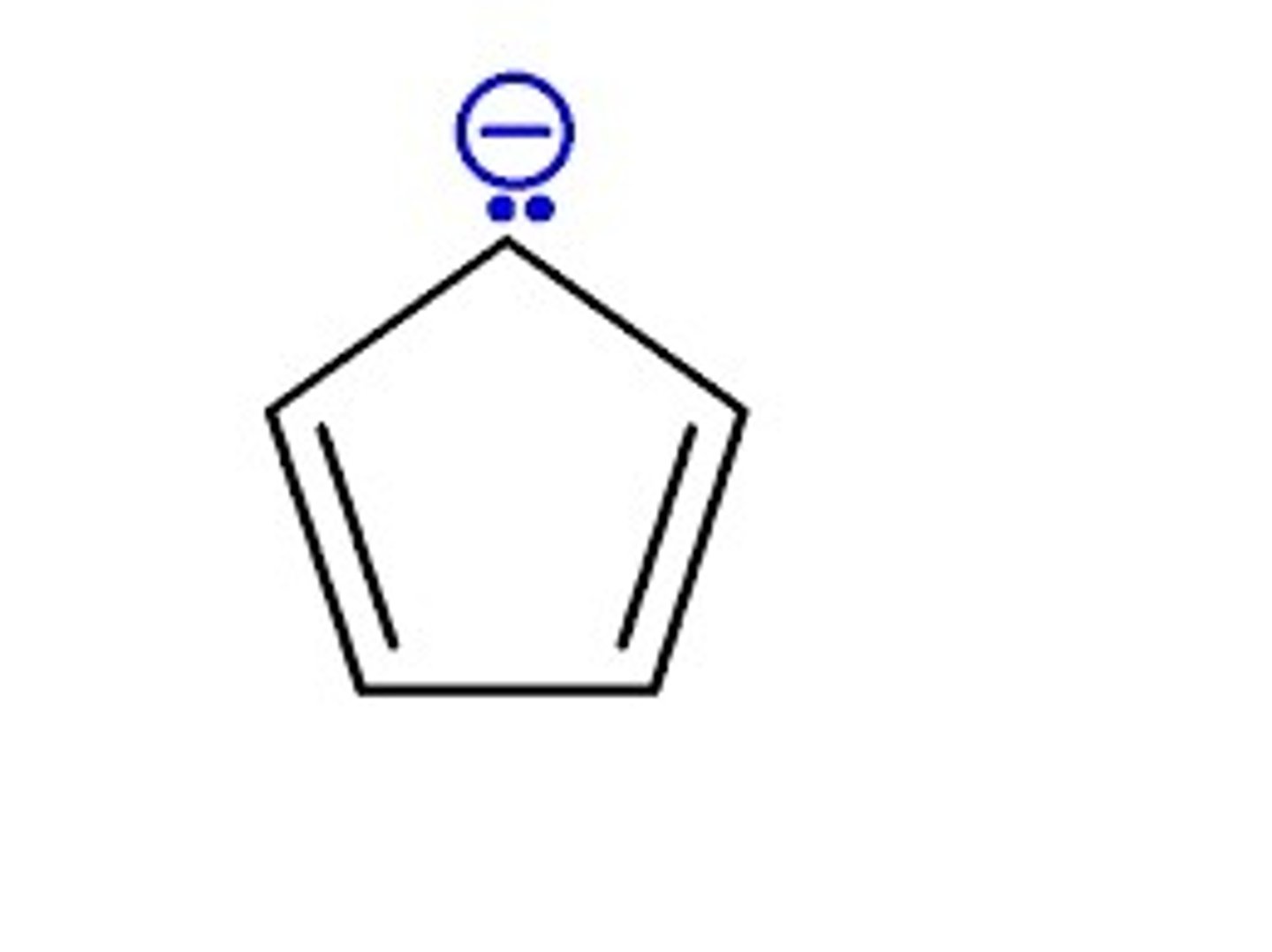
cyclopentadienyl anion (aromatic)
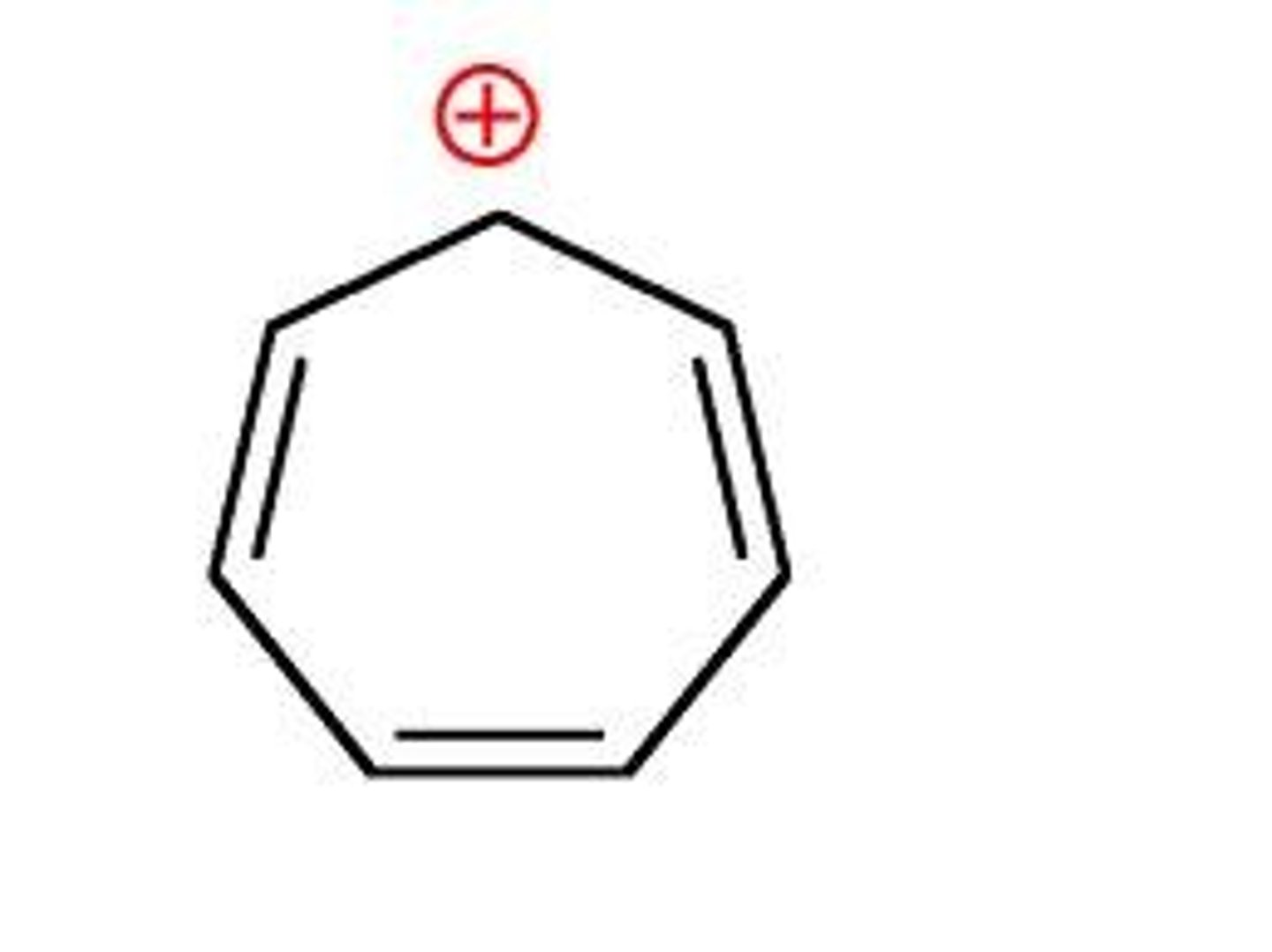
Tropylium cation (aromatic)
Pyridine
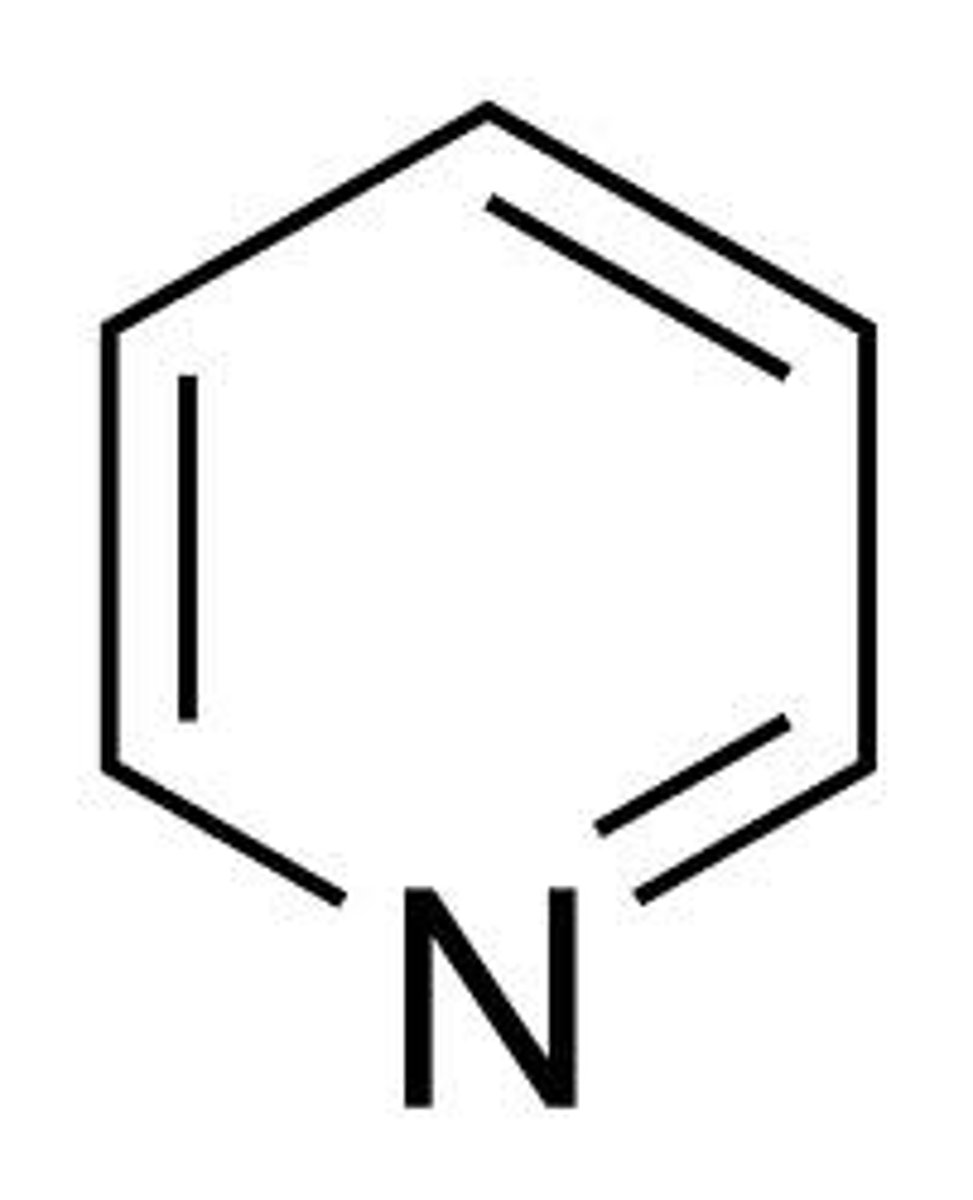
Is pyridine a _____
Basic because the lone pair is not part of the conjugated p orbitals
Pyrrole
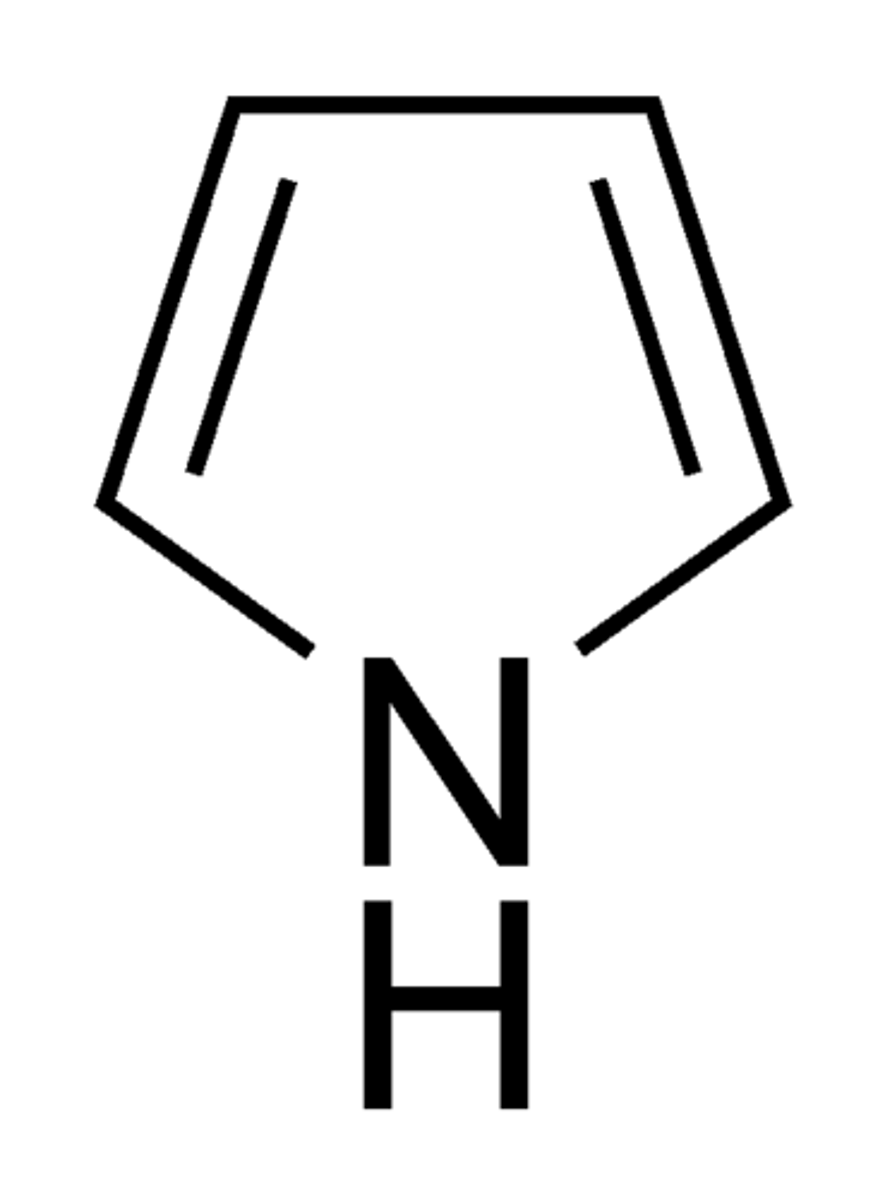
Pyrrole____
Is going to be an acid because it is it needs its one pair to have conjugated p orbitals
How Does an aromatic with a carbon chain become a benzoic acid?
Na2Cr2O7, H2SO4, H2O
Birch reaction
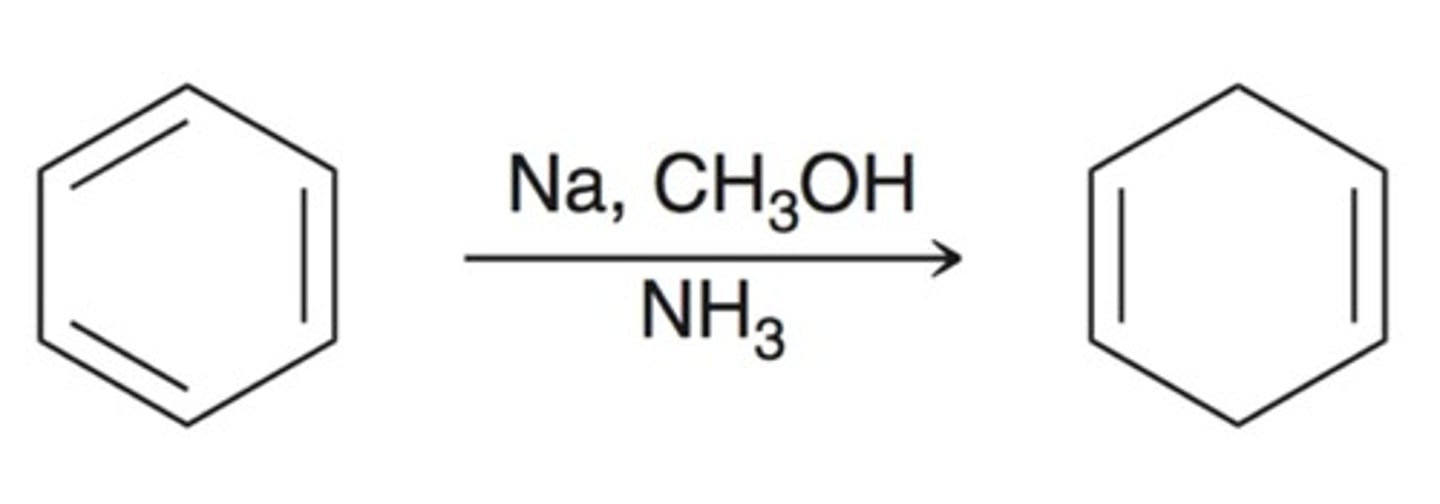
What reagents do you need for a birch reaction?
Na, CH3OH, NH3 (l)
how do EWG and EDG effect birch reactions
The double bond will go on the EDG and the not on the EWG
When is 1,4 adduction favored
Under thermodynamic conditions
When is 1,2 adduction favored
Under kinetic conditions