Orgo Radicals Quiz
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Heat or light
Main conditions needed for homolytic cleavage (creation of radicals)
T
T/F: The stability of a radical follows the same pattern as carbocation stability
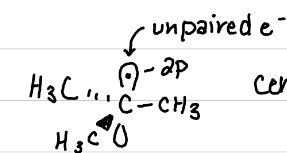
Hyperconjugation
Radicals are stabilized by ….

Benzylic Radical (Draw out!)
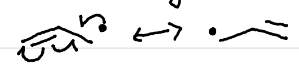
Allylic Radical
NBS/NCS
Common bromide/chloride radical source
AIBN
Source of this radical
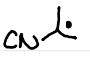
Radical Coupling
Abstraction
Addition (to alkene)
3 types of radical reactivity
Radical coupling
What is this type of radical reactivity?
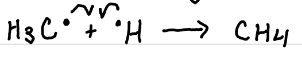
Abstraction
What is this type of radical reactivity?

Sigma
Abstraction is often a reaction between a radical and a [pi/sigma] bond
Radical Addition to Alkene
What is this type of radical reactivity?

Initiation
Forms the 1st radicals in a reaction
Propogation
Stage when the desired reaction occurs + regenerated the reactive species
Termination
Quenching of the radical species
Regeneration of reagents
Creation of product
New side product
Ways for a radical rxn to be quenched/terminated
I2 < Br2 < Cl2< F2
Order of rate of reaction with halogen gases
An iodide radical is relatively stable due to its larger e- cloud —> lower reactivity —> Slower rxn
Why is a reaction with iodide so slow?
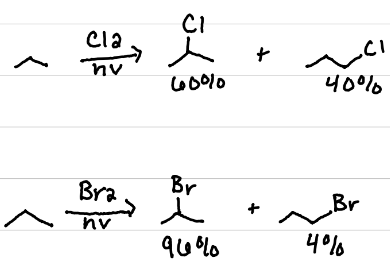
Br2
Which is more selective?: Cl2 or Br2
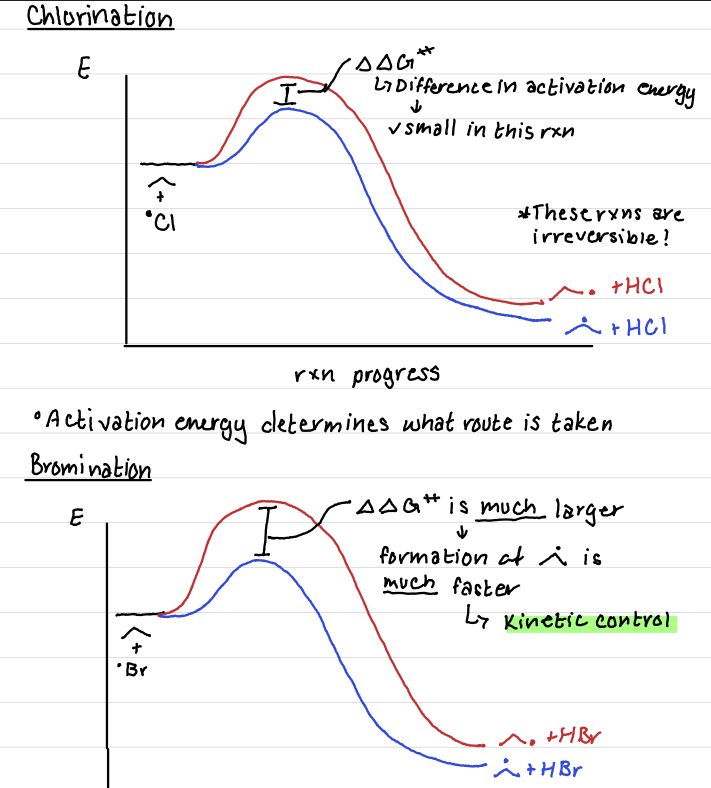
Chlorination
Is chlorination or bromination faster?
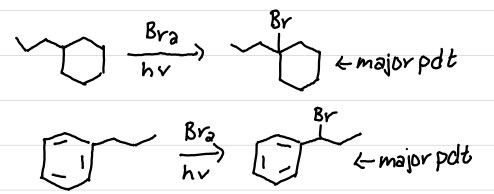
1 carbon away
A benzylic alkyl group will add a halide…
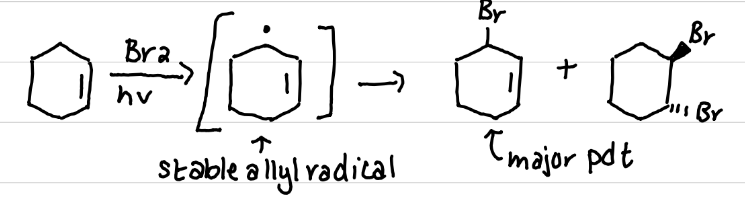
Draw the product(s) of this reaction
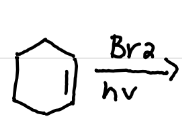
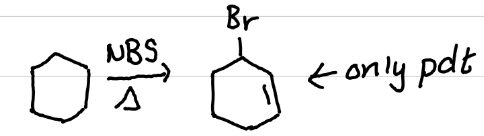
Draw the product(s) of this reaction
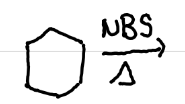
Anti-markovnikov
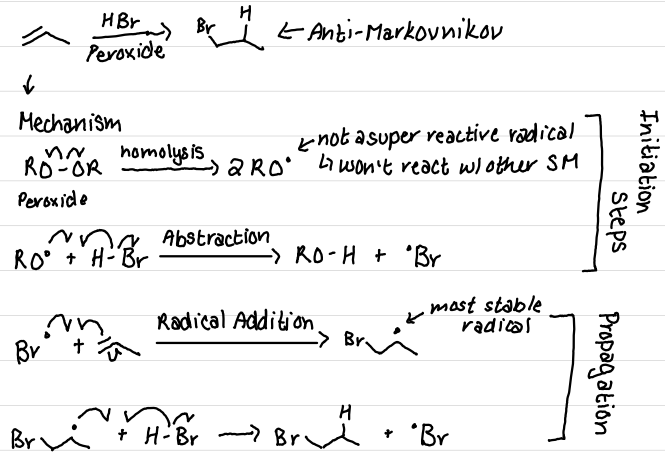
Draw the mechanism + product(s) of this reaction. What is this product called?

Radical initiator
Peroxide is considered a …
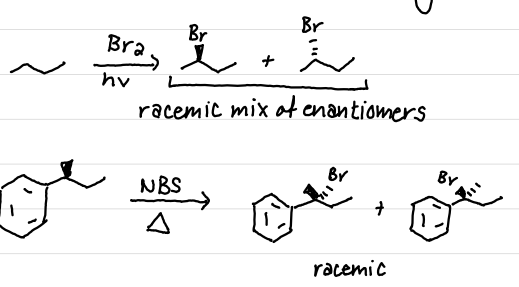
sp²; planar; loss of stereochemical information
The carbon with the radical is sp^# hybridized and …, leading to….

Draw the 2 types of peroxides
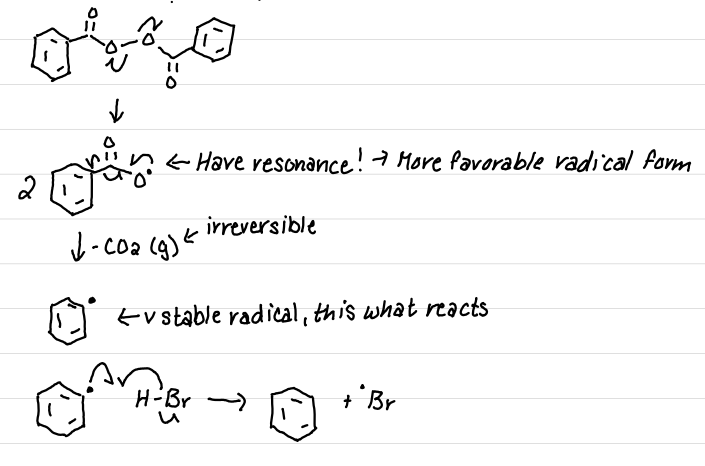
Draw the reaction between (PhCO2)2 with HBr
Hint: Produces Br radical - how?
Possibility of e1 reactions
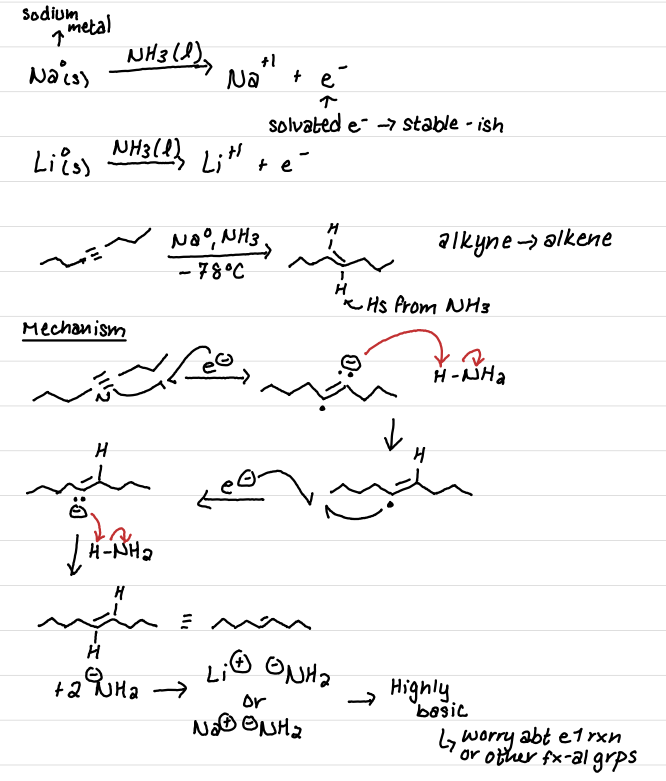
Draw the reaction: Radical hydrogenation of alkynes using Na or Li metal w/ HN3
What do we need to worry about with this reaction?
Hint: Results in highly basic byproducts
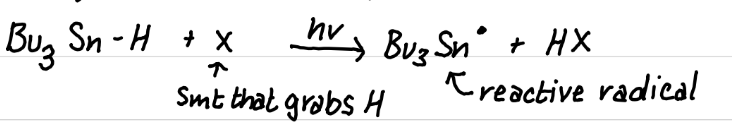
Draw the reaction of HSnBu3 (organotin) with light

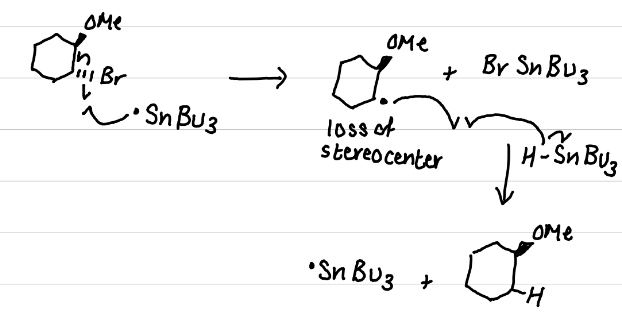
Draw the product(s) of this reaction
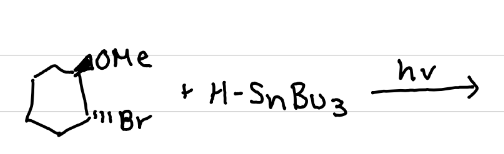
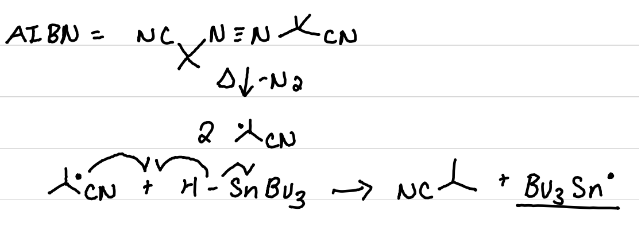
AIBN, Higher temps (60oC)
What can be used to activate HSnBu3 to form a radical? What is that reaction? What conditions does it require?
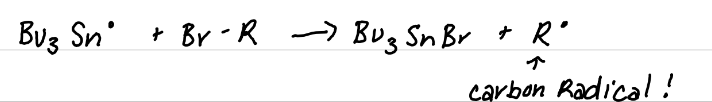
Forming C-C bonds
What is/are the product(s) of this reaction? What are the products useful for?
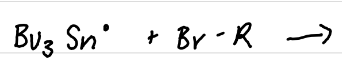
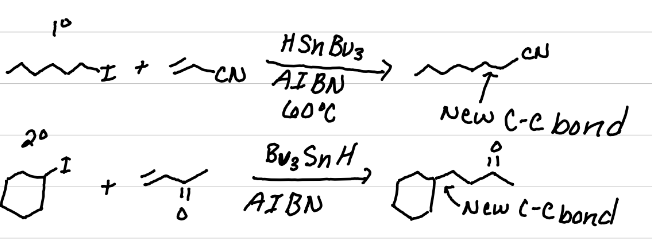
Draw the products of these reactions
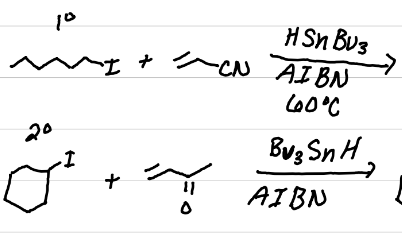
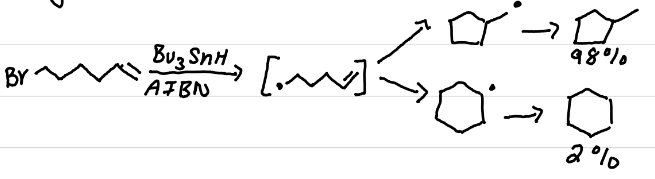
Draw the product(s) of this reaction
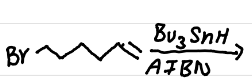
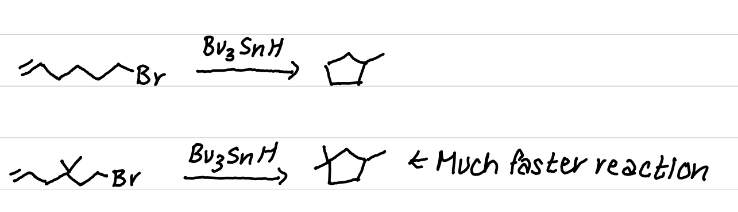
The addition of gem-dimethyls results in a faster ring closure reaction
due to higher steric hinderance favoring ring closure (& other intramolecular reactions)
What is the Thorpe-Infold Effect?